Abstract
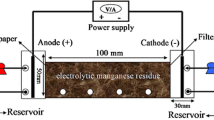
Regional contamination by electrolytic manganese residue (EMR) not only composes a serious environmental problem but also leads to severe valuable resources waste. Directly recovering manganese and ammonium sulfate is a promising way, but it is still challenging to efficiently recover without high water consumption. Herein, a recovery method based on water column leaching under extremely low water consumption was firstly reported. The effect of continuous leaching and intermittent leaching on leaching behaviors, leaching trends, and spatial variations of (NH4)2SO4 and Mn with depth after leaching were fully investigated. Results indicated that some Mn-bearing soluble salts which covered on the surface of SiO2 in the micropores could be fully dissolved and transported out of the micropores in the EMR with the help of rest periods in the method of intermittent leaching, resulting in higher leaching efficiencies with comparison to continuous leaching, 73.50% of Mn and 67.71% of (NH4)2SO4 and 71.57% of Mn and 65.40% of (NH4)2SO4 were recovered by intermittent leaching and continuous leaching, respectively. This work demonstrates a practical approach to recover valuable materials from industrial solid wastes.











Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Al-Sibai M, Adey MA, Rose DA (1997) Movement of solute through a porous medium under intermittent leaching. Eur J Soil Sci 48:711–725
Chen H, Liu R, Shu J, Li W (2015) Simultaneous stripping recovery of ammonia-nitrogen and precipitation of manganese from electrolytic manganese residue by air under calcium oxide assist. J Environ Sci Heal a 50:1282–1290
Chen H, Liu R, Liu Z, Shu J, Tao C (2016) Immobilization of Mn and NH 4+-N from electrolytic manganese residue waste. Environ Sci Pollut Res 23:12352–12361
Chen H, Long Q, Zhang Y, Qin L (2019) Simultaneous immobilization of NH 4+ and Mn 2+ from electrolytic manganese residue using phosphate and magnesium sources. RSC Adv 9:4583–4590
Cheng H (2001) The techno-economic analysis of the extending of tailing pond in Ymdongpo gold mine. Gold 23:32–34
Cote CM, Bristow KL, Ross PJ (2000) Increasing the efficiency of solute leaching: impacts of flow interruption with drainage of the “preferential flow paths.” J Contam Hydrol 43:191–209
Du B, Hou D, Duan N, Zhou C, Wang J, Dan Z (2015) Immobilization of high concentrations of soluble Mn (II) from electrolytic manganese solid waste using inorganic chemicals. Environ Sci Pollut Res 22:7782–7793
Duan N, Zhou C, Chen B, Jiang W, Xin B (2011) Bioleaching of Mn from manganese residues by the mixed culture of Acidithiobacillus and mechanism. J Chem Technol Biotechnol 86:832–837
Ge XX, Cai GP, Zeng GM (2004) Study on harmless disposal and comprehensive utilization of manganese sulfate waste residue. China’s Manganese Ind 22:11–14
He D, Shu J, Wang R, Chen M, Wang R, Gao Y, Wang N (2021) A critical review on approaches for electrolytic manganese residue treatment and disposal technology: reduction, pretreatment, and reuse. J Hazard Mater 418:126235
Huang Y (2007) The technological and economic analyze about Daxing Bukang EMM residue dam project. China’s Manganese Ind 25:28–30
Li D (2008) Principles of Electrochemistry. Beihang University Press, Beijing
López S, Kalbe U, Berger W, Simon F (2010) Effect of contact time on the release of contaminants from granular waste materials during column leaching experiments. Waste Manag 30:565–571
Oster JD, Willardson LS, Hoffman GJ (1972) Sprinkling and ponding techniques for reclaiming saline soils. Trans ASAE 15:1115–1117
Ouyang Y, Li Y, Li H, Li Z, Peng X, Yan W (2008) Recovery of manganese from electrolytic manganese residue by different leaching techniques in the presence of accessory ingredients. Rare Metal Mater Eng 37:603–608
Qiao J, Sun H, Luo X, Zhang W, Mathews S, Yin X (2017) EDTA-assisted leaching of Pb and Cd from contaminated soil. Chemosphere 167:422–428
Rose DA, Adey MA, Al-Sibai M (2000) Laboratory experiments and modelling studies of leaching of intermittently drained columns. Aust J Soil Res 38:891–903
Shu J, Liu R, Liu Z, Chen H, Tao C (2016) Enhanced extraction of manganese from electrolytic manganese residue by electrochemical. J Electroanal Chem 780:32–37
Shu J, Chen M, Wu H, Li B, Wang B, Li B, Liu R, Liu Z (2019) An innovative method for synergistic stabilization/solidification of Mn2+, NH4+-N, PO43− and F− in electrolytic manganese residue and phosphogypsum. J Hazard Mater 376:212–222
SMM (2019) https://news.smm.cn/news/101007541
Tabelin CB, Sasaki R, Igarashi T, Park I, Tamoto S, Arima T, Ito M, Hiroyoshi N (2017) Simultaneous leaching of arsenite, arsenate, selenite and selenate, and their migration in tunnel-excavated sedimentary rocks: I. Column experiments under intermittent and unsaturated flow. Chemosphere 186:558–569
Tan Z, Mei G, Li W, Zeng K, Liang N, Zeng X (2004) Metallurgy of manganese. Press of Central South University of Technology, Changsha
Tian Y, Shu J, Chen M, Wang J, Wang Y, Luo Z, Sun Z (2019) Manganese and ammonia nitrogen recovery from electrolytic manganese residue by electric field enhanced leaching. J Clean Prod 236:117708
Wang Y, Gao S, Liu X, Tang B, Mukiza E, Zhang N (2019) Preparation of non-sintered permeable bricks using electrolytic manganese residue: environmental and NH3-N recovery benefits. J Hazard Mater 378:120768
Xin B, Chen B, Duan N, Zhou C (2011) Extraction of manganese from electrolytic manganese residue by bioleaching. Bioresour Technol 102:1683–1687
Yu D (1991) Application of Eh – pH diagrams to ammonia hydrometallurgy: Cu - NH3 - H2O, Cu - NH3 - H2O, Cu - NH3 - H2O, system at 25 ºC. J Chongqing Teach Coll (Nat. Sci. Ed). 8 25–30
Zhang R, Ma X, Shen X, Zhai Y, Zhang T, Ji C, Hong J (2020) Life cycle assessment of electrolytic manganese metal production. J Clean Prod 253:119951
Zheng F, Li Y, Wang D, Li J, Zhu H (2018) The effects of Zn 2 and polyacrylamide (PAM) on the electrodeposition of manganese from non-selenium sulfate solutions. Int J Electrochem Sci 13:1879–1892
Zheng F, Zhu H, Luo T, Wang H, Hou H (2020) Water leaching soluble manganese from electrolytic manganese residue: leaching kinetics model analysis and characterization. J Environ Chem Eng 103916
Zhou C, Wang J, Wang N (2013) Treating electrolytic manganese residue with alkaline additives for stabilizing manganese and removing ammonia. Korean J Chem Eng 30:2037–2042
Funding
All works in this study are financially supported by the National Key R&D Program of China (2018YFC1801705).
Author information
Authors and Affiliations
Contributions
Fan Zheng carried out the experiment, analyzed the data, and wrote the original draft; Wei Xie carried out the related experiments, drew pictures, and characterization test; Hua Zhu conceived this study, reviewed, and edited the manuscript; Haobo Hou acquired the funding.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible editor: Ioannis A. Katsoyiannis
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zheng, F., Xie, W., Zhu, H. et al. Water column leaching recovery manganese and ammonium sulfate from electrolytic manganese residue: extremely low water consumption toward practical applications. Environ Sci Pollut Res 29, 80323–80335 (2022). https://doi.org/10.1007/s11356-022-21463-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21463-9