Abstract
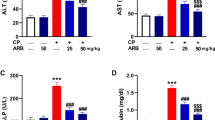
Treatment with anti-neoplastic agents, including cyclophosphamide (CP), is associated with several adverse reactions. Here, we distinguished the potential protective effect of allicin against CP-mediated hepatotoxicity in rats. To assess the effect of allicin, four experimental groups were used, with 7 rats per group, including control, allicin (10 mg/kg), CP (200 mg/kg), and allicin + CP-treated groups. All groups were treated for 10 days. Blood and liver samples were collected for biochemical, molecular, and histological analyses. Treatment with CP led to deformations in the liver tissue that were associated with higher liver function markers (alanine transaminase, aspartate transaminase, and alkaline phosphatase). Additionally, a disturbance in the redox balance was observed after CP exposure, as indicated by increased levels of oxidants, including malondialdehyde and nitric oxide, and the decreased levels of endogenous antioxidants, including glutathione, glutathione peroxidase, glutathione reductase, superoxide dismutase, and catalase. At the molecular level, CP treatment resulted in reduced expression of the Nrf2/ARE pathway and other genes related to this pathway, including NAD(P)H quinone dehydrogenase 1 and glutamate-cysteine ligase catalytic subunit. CP also led to a hyper-inflammatory response in hepatic tissue, with increased production of pro-inflammatory cytokines, including tumor necrosis factor-alpha and interlukin-1beta, and upregulation of nitric oxide synthase 2. CP also enhanced the immunoreactivity of the profibrogenic cytokine, transforming growth factor-beta, in liver tissue. Upregulation of caspase 3 and Bcl-2-associated X protein and downregulation of B-cell lymphoma 2 were also observed in response to CP treatment. Treatment with allicin reversed the molecular, biochemical, and histological changes that occurred with CP exposure. These results suggest that allicin can be used in combination with CP to avoid hepatotoxicity.









Similar content being viewed by others
References
Abdallah HM et al (2019) Protective effect of some natural products against chemotherapy-induced toxicity in rats. Heliyon 5:e01590
Abdel-Daim MM, Abushouk AI, Donia T, Alarifi S, Alkahtani S, Aleya L, Bungau SG (2019) The nephroprotective effects of allicin and ascorbic acid against cisplatin-induced toxicity in rats. Environ Sci Pollut Res 26:13502–13509
Abdel-Daim MM, Khalifa HA, Ahmed AA (2017a) Allicin ameliorates doxorubicin-induced cardiotoxicity in rats via suppression of oxidative stress, inflammation and apoptosis. Cancer Chemother Pharmacol 80:745–753
Abdel-Daim MM, Kilany OE, Khalifa HA, Ahmed AAM (2017b) Allicin ameliorates doxorubicin-induced cardiotoxicity in rats via suppression of oxidative stress, inflammation and apoptosis. Cancer Chemother Pharmacol 80:745–753. https://doi.org/10.1007/s00280-017-3413-7
Adikwu E, Bokolo B (2018) Protective assessment of cimetidine against cyclophosphamide-induced kidney injury. Asian J Med Sci 9:25–30
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Al-Brakati AY, Fouda MS, Tharwat AM, Elmahallawy EK, Kassab RB, Abdel Moneim AE (2019) The protective efficacy of soursop fruit extract against hepatic injury associated with acetaminophen exposure is mediated through antioxidant, anti-inflammatory, and anti-apoptotic activities. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-019-04935-3
Alam RTM, Fawzi EM, Alkhalf MI, Alansari WS, Aleya L, Abdel-Daim MM (2018a) Anti-inflammatory, immunomodulatory, and antioxidant activities of allicin, norfloxacin, or their combination against pasteurella multocida infection in male New Zealand rabbits. Oxidative Med Cell Longev 2018:1780956–1780956. https://doi.org/10.1155/2018/1780956
Alam RTM, Fawzi EM, Alkhalf MI, Alansari WS, Aleya L, Abdel-Daim MM (2018b) Anti-inflammatory, immunomodulatory, and antioxidant activities of allicin, norfloxacin, or their combination against pasteurella multocida infection in male New Zealand rabbits. Oxidative Med Cell Longev 2018:1780956. https://doi.org/10.1155/2018/1780956
Almeer RS et al (2018) Royal jelly abrogates cadmium-induced oxidative challenge in mouse testes: involvement of the nrf2 pathway. Int J Mol Sci 19. https://doi.org/10.3390/ijms19123979
Andersson MC, Tobin G, Giglio D (2008) Cholinergic nitric oxide release from the urinary bladder mucosa in cyclophosphamide-induced cystitis of the anaesthetized rat. Br J Pharmacol 153:1438–1444. https://doi.org/10.1038/bjp.2008.6
Arellano Buendia AS et al (2018) Immunomodulatory effects of the nutraceutical garlic derivative allicin in the progression of diabetic nephropathy. Int J Mol Sci 19. https://doi.org/10.3390/ijms19103107
Arellano Buendía AS, Tostado González M, Sánchez Reyes O, García Arroyo F, Argüello García R, Tapia E, Sánchez Lozada L, Osorio Alonso H (2018) Immunomodulatory effects of the nutraceutical garlic derivative allicin in the progression of diabetic nephropathy. Int J Mol Sci 19:3107. https://doi.org/10.3390/ijms19103107
Borlinghaus J, Albrecht F, Gruhlke MC, Nwachukwu ID, Slusarenko AJ (2014) Allicin: chemistry and biological properties. Molecules 19:12591–12618. https://doi.org/10.3390/molecules190812591
Bruck R, Aeed H, Brazovsky E, Noor T, Hershkoviz R (2005) Allicin, the active component of garlic, prevents immune-mediated, concanavalin A-induced hepatic injury in mice. Liver Int 25:613–621. https://doi.org/10.1111/j.1478-3231.2005.01050.x
Caglayan C, Temel Y, Kandemir FM, Yildirim S, Kucukler S (2018) Naringin protects against cyclophosphamide-induced hepatotoxicity and nephrotoxicity through modulation of oxidative stress, inflammation, apoptosis, autophagy, and DNA damage. Environ Sci Pollut Res Int 25:20968–20984. https://doi.org/10.1007/s11356-018-2242-5
Cao Y, Wang X, Li S, Wang H, Yu L, Wang P (2017) The effects of l-carnitine against cyclophosphamide-induced injuries in mouse testis. Basic Clin Pharmacol Toxicol 120:152–158
Casanova NA, Simoniello MF, Lopez Nigro MM, Carballo MA (2017) Modulator effect of watercress against cyclophosphamide-induced oxidative stress in mice. Medicina (B Aires) 77:201–206
Chen W et al (2014) Neuroprotective effect of allicin against traumatic brain injury via Akt/endothelial nitric oxide synthase pathway-mediated anti-inflammatory and anti-oxidative activities. Neurochem Int 68:28–37. https://doi.org/10.1016/j.neuint.2014.01.015
Clancy RM, Gomez PF, Abramson SB (2004) Nitric oxide sustains nuclear factor kappaB activation in cytokine-stimulated chondrocytes. Osteoarthr Cartil 12:552–558. https://doi.org/10.1016/j.joca.2004.04.003
Clifton K, Barnett C, Martinez A, Karuturi M, Saleem S, Litton J (2018) Severe hyponatremia following cyclophosphamide infusion in breast cancer patients. Breast J 24:691–692. https://doi.org/10.1111/tbj.13026
Deng J et al (2018) Carnosine attenuates cyclophosphamide-induced bone marrow suppression by reducing oxidative DNA damage. Redox Biol 14:1–6. https://doi.org/10.1016/j.redox.2017.08.003
Dkhil MA, Abdel Moneim AE, Hafez TA, Mubaraki MA, Mohamed WF, Thagfan FA, Al-Quraishy S (2019) Myristica fragrans kernels prevent paracetamol-induced hepatotoxicity by inducing anti-apoptotic genes and Nrf2/HO-1 pathway. Int J Mol Sci 20:993
El-Sebaey AM, Abdelhamid FM, Abdalla OA (2019) Protective effects of garlic extract against hematological alterations, immunosuppression, hepatic oxidative stress, and renal damage induced by cyclophosphamide in rats. Environ Sci Pollut Res Int 26:15559–15572. https://doi.org/10.1007/s11356-019-04993-7
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Engdal S, Klepp O, Nilsen OG (2009) Identification and exploration of herb-drug combinations used by cancer patients. Integrat Cancer Ther 8:29–36
Fabregat I, Moreno-Càceres J, Sánchez A, Dooley S, Dewidar B, Giannelli G, ten Dijke P (2016) TGF-β signalling and liver disease. FEBS J 283:2219–2232
Fisher AE, Maxwell SC, Naughton DP (2003) Catalase and superoxide dismutase mimics for the treatment of inflammatory diseases. Inorg Chem Commun 6:1205–1208
Fouad AA, Qutub HO, Al-Melhim WN (2016) Punicalagin alleviates hepatotoxicity in rats challenged with cyclophosphamide. Environ Toxicol Pharmacol 45:158–162. https://doi.org/10.1016/j.etap.2016.05.031
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138
Huang H, Jiang Y, Mao G, Yuan F, Zheng H, Ruan Y, Wu T (2017) Protective effects of allicin on streptozotocin-induced diabetic nephropathy in rats. J Sci Food Agric 97:1359–1366. https://doi.org/10.1002/jsfa.7874
Huehnchen P, van Kampen A, Boehmerle W, Endres M (2020) Cognitive impairment after cytotoxic chemotherapy. Neuro-oncol Pract 7:11–21. https://doi.org/10.1093/nop/npz052
Janero DR (1990) Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9:515–540
Kong X, Gong S, Su L, Li C, Kong Y (2017) Neuroprotective effects of allicin on ischemia-reperfusion brain injury. Oncotarget 8:104492–104507. https://doi.org/10.18632/oncotarget.22355
Kurauchi K, Nishikawa T, Miyahara E, Okamoto Y, Kawano Y (2017) Role of metabolites of cyclophosphamide in cardiotoxicity. BMC Res Notes 10:406. https://doi.org/10.1186/s13104-017-2726-2
Lee SI, Kang KS (2017) Function of capric acid in cyclophosphamide-induced intestinal inflammation, oxidative stress, and barrier function in pigs. Sci Rep 7:16530–16530. https://doi.org/10.1038/s41598-017-16561-5
Lin S et al (2017) Oyster (Ostrea plicatula Gmelin) polysaccharides intervention ameliorates cyclophosphamide-Induced genotoxicity and hepatotoxicity in mice via the Nrf2-ARE pathway. Biomedi Pharmacother Biomed Pharmacother 95:1067–1071. https://doi.org/10.1016/j.biopha.2017.08.058
Luo J et al (2020) Chlorogenic acid attenuates cyclophosphamide-induced rat interstitial cystitis. Life Sci:117590. https://doi.org/10.1016/j.lfs.2020.117590
Lv R et al (2017) Allicin protects against H(2)O(2)-induced apoptosis of PC12 cells via the mitochondrial pathway. Exper Therapeut Med 14:2053–2059. https://doi.org/10.3892/etm.2017.4725
Mahmoud AM, Germoush MO, Alotaibi MF, Hussein OE (2017) Possible involvement of Nrf2 and PPARgamma up-regulation in the protective effect of umbelliferone against cyclophosphamide-induced hepatotoxicity. Biomedi Pharmacother Biomed Pharmacother 86:297–306. https://doi.org/10.1016/j.biopha.2016.12.047
Metwally DM, Al-Olayan EM, Alanazi M, Alzahrany SB, Semlali A (2018) Antischistosomal and anti-inflammatory activity of garlic and allicin compared with that of praziquantel in vivo. BMC Complement Altern Med 18:135. https://doi.org/10.1186/s12906-018-2191-z
Mills KA, Chess-Williams R, McDermott C (2019) Novel insights into the mechanism of cyclophosphamide-induced bladder toxicity: chloroacetaldehyde’s contribution to urothelial dysfunction in vitro. Arch Toxicol 93:3291–3303. https://doi.org/10.1007/s00204-019-02589-1
Ming Z, Yongqiang Z, Zijin Z, Yan X, Di C, Xiaoxin T (2019) Severe and prolonged cyclophosphamide-induced hepatotoxicity in a breast cancer patient carrying a CYP2B6*7 variant. Pharmacogenomics 20:1119–1124. https://doi.org/10.2217/pgs-2019-0093
Mir BA et al (2018) Chemoprotective potential of zingerone (vanillyl acetone) in cyclophosphamide-induced hepatic toxicity. Pharmacogn Mag 14:434
Oboh G, Akomolafe TL, Adefegha SA, Adetuyi AO (2011) Inhibition of cyclophosphamide-induced oxidative stress in rat brain by polar and non-polar extracts of Annatto (Bixa orellana) seeds. Exp Toxicol Pathol 63:257–262
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Pandurangan AK, Ismail S, Saadatdoust Z, Esa NM (2015) Allicin alleviates dextran sodium sulfate- (dss-) induced ulcerative colitis in BALB/c mice. Oxidative Med Cell Longev 13:605208. https://doi.org/10.1155/2015/605208
Panyod S, Wu WK, Ho CT, Lu KH, Liu CT, Chu YL, Lai YS, Chen WC, Lin YE, Lin SH, Sheen LY (2016) Diet supplementation with allicin protects against alcoholic fatty liver disease in mice by improving anti-inflammation and antioxidative functions. J Agric Food Chem 64:7104–7113. https://doi.org/10.1021/acs.jafc.6b02763
Pathak S et al (2018) Benefits of aged garlic extract in modulating toxicity biomarkers against p-dimethylaminoazobenzene and phenobarbital induced liver damage in Rattus norvegicus. Drug Chem Toxicol:1–14. https://doi.org/10.1080/01480545.2018.1499773
Radi R (2018) Oxygen radicals, nitric oxide, and peroxynitrite: redox pathways in molecular medicine. Proc Natl Acad Sci U S A 115:5839–5848. https://doi.org/10.1073/pnas.1804932115
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochim Biophys Acta (BBA) - Mol Cell Res 1863:2977–2992. https://doi.org/10.1016/j.bbamcr.2016.09.012
Reiter J, Levina N, van der Linden M, Gruhlke M, Martin C, Slusarenko AJ (2017) Diallylthiosulfinate (Allicin), a volatile antimicrobial from garlic (Allium sativum), kills human lung pathogenic bacteria, including mdr strains, as a vapor. Molecules (Basel, Switzerland) 22:1711. https://doi.org/10.3390/molecules22101711
Samra YA, Hamed MF, El-Sheakh AR (2020) Hepatoprotective effect of allicin against acetaminophen-induced liver injury: role of inflammasome pathway, apoptosis, and liver regeneration. J Biochem Mol Toxicol
Sherif IO (2018) The effect of natural antioxidants in cyclophosphamide-induced hepatotoxicity: role of Nrf2/HO-1 pathway. Int Immunopharmacol 61:29–36. https://doi.org/10.1016/j.intimp.2018.05.007
Sheth VG, Navik U, Maremanda KP, Jena G (2018) Effect of diethyldithiocarbamate in cyclophosphamide-induced nephrotoxicity: immunohistochemical study of superoxide dismutase 1 in rat. Indian J Pharm 50:4–11. https://doi.org/10.4103/ijp.IJP_850_16
Shokrzadeh M, Ahmadi A, Naghshvar F, Chabra A, Jafarinejhad M (2014) Prophylactic efficacy of melatonin on cyclophosphamide-induced liver toxicity in mice. Biomed Res Int 6:470425. https://doi.org/10.1155/2014/470425
Singh C, Prakash C, Tiwari KN, Mishra SK, Kumar V (2018) Premna integrifolia ameliorates cyclophosphamide-induced hepatotoxicity by modulation of oxidative stress and apoptosis. Biomedi Pharmacother Biomed Pharmacother 107:634–643. https://doi.org/10.1016/j.biopha.2018.08.039
Smith IK, Vierheller TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5,5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 175:408–413
Stojiljkovic N et al (2019) Nanoliposome-encapsulated ellagic acid prevents cyclophosphamide-induced rat liver damage. Mol Cell Biochem. https://doi.org/10.1007/s11010-019-03541-8
Takimoto C, Calvo E (2008) In: Pazdur R, Wagman LD, Camphausen KA, Hoskins WJ (eds) Principles of oncologic pharmacotherapy: in cancer management, a multidisciplinary approach. PRR, Melville
Uskudar Cansu D, Oztas E, Yilmaz E, Korkmaz C (2019) Cyclophosphamide-induced severe acute hepatitis in a rheumatic disease: case-based review. Rheumatol Int 39:377–385. https://doi.org/10.1007/s00296-018-4189-8
Wang ET, Chen DY, Liu HY, Yan HY, Yuan Y (2015) Protective effect of allicin against glycidamide-induced toxicity in male and female mice. Gen Physiol Biophys 34:177–187. https://doi.org/10.4149/gpb_2014038
Wu CC, Chu YL, Sheen LY (2012) Allicin modulates the antioxidation and detoxification capabilities of primary rat hepatocytes. J Tradit Complement Med 2:323–330. https://doi.org/10.1016/s2225-4110(16)30118-3
Wu X et al (2017) Allicin protects against cisplatin-induced vestibular dysfunction by inhibiting the apoptotic pathway. Eur J Pharmacol 805:108–117. https://doi.org/10.1016/j.ejphar.2017.02.052
Yang D et al (2017) Activation of the Nrf2 signaling pathway involving KLF9 plays a critical role in allicin resisting against arsenic trioxide-induced hepatotoxicity in rats. Biol Trace Elem Res 176:192–200. https://doi.org/10.1007/s12011-016-0821-1
Zhai J, Zhang F, Gao S, Chen L, Feng G, Yin J, Chen W (2018) Schisandra chinensis extract decreases chloroacetaldehyde production in rats and attenuates cyclophosphamide toxicity in liver, kidney and brain. J Ethnopharmacol 210:223–231
Zhang L, Zhang H, Miao Y, Wu S, Ye H, Yuan Y (2012) Protective effect of allicin against acrylamide-induced hepatocyte damage in vitro and in vivo. Food Chem Toxicol 50:3306–3312. https://doi.org/10.1016/j.fct.2012.05.060
Zhang M, Pan H, Xu Y, Wang X, Qiu Z, Jiang L (2017) Allicin decreases lipopolysaccharide-induced oxidative stress and inflammation in human umbilical vein endothelial cells through suppression of mitochondrial dysfunction and activation of Nrf2 cellular physiology and biochemistry. Int J Exper Cell Physiol Biochem Pharmacol 41:2255–2267. https://doi.org/10.1159/000475640
Zhuge J, Cederbaum AI (2006) Increased toxicity by transforming growth factor-beta 1 in liver cells overexpressing CYP2E1. Free Radic Biol Med 41:1100–1112
Availability of data and materials
All relevant data are within the paper.
Author information
Authors and Affiliations
Contributions
Hassan Al Sberi, Maha S Lokman, and Rami B. Kassab: animal treatments, biochemical methodology, and histological and immunohistochemistry examinations. Manar S. Fouda and Mohamed S. Othman: molecular analysis. Dongsheng Sun and Chen Sun: resources, visualization, investigation, and software. Gongcai Qiu and Lei Yao: writing—reviewing and editing. Jian Yu and Ahmed E. Abdel Moneim: conceptualization, validation, and supervision.
Corresponding author
Ethics declarations
Ethical approval
The current experimental design was approved by the research ethical committee of the Helwan University (approval no. HU2019/Z/RK0420-09) in accordance with the National Institutes of Health (NIH) Guidelines for the Care and Use of Laboratory Animals, 8th edition (NIH Publication no. 85–23, revised 1985).
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, D., Sun, C., Qiu, G. et al. Allicin mitigates hepatic injury following cyclophosphamide administration via activation of Nrf2/ARE pathways and through inhibition of inflammatory and apoptotic machinery. Environ Sci Pollut Res 28, 39625–39636 (2021). https://doi.org/10.1007/s11356-021-13392-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13392-w