Abstract
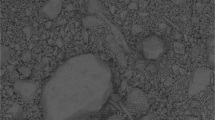
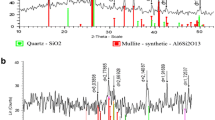
The current study aimed to investigate the efficiencies and mechanisms of slag filter media for removing phosphorus from synthetic wastewater. The steel slag with high ferric oxides (Fe2O3) was subjected for the electric arc furnace (EAF) and selected as the filter media (HFe). The chemical characteristics of HFe were determined using pH, point of zero charge (PZC) and XRF. The phosphorus removal efficiency was studied in a designed vertical steel slag column rock filters in unaerated HFe (UEF) and aerated HFe (AEF) system. The microstructure of HFe was analyzed by FTIR, XRD and SEM-EDX analysis. The results of XRF revealed that ferric oxide (Fe2O3) ranged from 26.1 to 38.2%. PZC for Filter HFe was recorded at pH 10.55 ± 0.27. The highest efficiencies were recorded by UEF and AEF systems at pH 3 and pH 5 (89.97 ± 4.02% and 79.95 ± 6.25% at pH 3 and 72.97 ± 8.38% and 66.00 ± 12.85% at pH 5 for UEF and AEF, respectively). These findings indicated that AEF exhibiting higher removal than UEF systems might be due to presence high Fe concentration in AEF which play important role in the phosphorus removal. The main elements available on the surface of HFe included carbon, oxygen, iron, calcium, magnesium, silicon, platinum, sulphur, manganese, titanium and aluminium. The XRD analysis indicated that the precipitation of orthophosphate as calcium and iron-phosphates was the removal mechanism as confirmed using FT-IR analysis. These findings demonstrated the efficiency of HFe in removing of phosphorus from wastewater.









Similar content being viewed by others
References
Afnizan WM, Hamdan R, Othman N (2015) Study of the maximum uptake capacity on various sizes of electric arc furnace slag in phosphorus aqueous solutions. In Soft Soil Engineering International Conference 2015 136
APHA (2005) Standard Methods for the Examination of Water and Wastewater. 21st Edition. Washington DC: American Public Health Association/American Water Works Association/Water Environment Federation.
APHA, 2012. Standard methods for the examination of water and wastewater, 22nd edition edited by E. W. Rice, R. B. Baird, A. D. Eaton and L. S. Clesceri. American Public Health Association (APHA), American Water Works Association (AWWA) and Water Environment Federation (WEF), Washington, D.C., USA.
Appel C, Ma LQ, Rhue RD (2003) Point of zero charge determination in soils and minerals via traditional methods and detection of electroacoustic mobility. Geoderma 113:77–93
Babayemi A, Onukwuli O (2016) Adsorption isotherms, thermodynamics and statistical modeling of phosphate removal from aqueous solution by locally prepared bio-sorbent. Journal of Applied Chemistry 9(6):46–50
Barca C, Gérente C, Meyer D, Chazarenc F, Andrès Y (2012) Phosphate removal from synthetic and real wastewater using steel slags produced in Europe. Water Research 46(7):2376–2384
Barca C, Meyer D, Liira M, Drissen P, Comeau Y, Andrès Y, Chazarenc F (2014) Steel slag filters to upgrade phosphorus removal in small wastewater treatment plants: Removal mechanisms and performance. Ecological Engineering 68:214–222
Barca C, Troesch S, Meyer D, Drissen P, Andreìs Y, & Chazarenc F (2013) Steel slag filters to upgrade phosphorus removal in constructed wetlands: Two years of field experiments. Environ Sci Technol 47(1):549–556
Berzina-cimdina, L., & Borodajenko, N. (2012). Research of calcium phosphates using fourier transform infrared spectroscopy. In T. Theophanides (Ed.), Materials Science, Engineering and Technology (pp. 123–128). In TechOpen.
Bowden LILB, Jarvis PAP, Younger PLPL, Johnson KLKL (2009) Phosphorus removal from wastewaters using basic oxygen slag. Environmental Science & Technology 43:2476–2481
Drizo A, Forget C, Chapuis RP, Comeau Y (2006) Phosphorus removal by electric arc furnace steel slag and serpentinite. Water Research 40(8):1547–1554
Hamdan R, Mara D (2013) Study of in-filter phosphorus removal mechanisms in an aerated blast furnace slag. International Journal of Research in Engineeing and Technology 2(8):130–136
Hammer M, Hammer M (2001) Water and wastewater technology. Prentice Hall, New Jersey
Han C, Wang Z, Yang H, Xue X (2015) Removal kinetics of phosphorus from synthetic wastewater using basic oxygen furnace slag. Journal of Environmental Sciences 30:21–29
Han C, Wang Z, Yang W, Wu Q, Yang H, Xue X (2016) Effects of pH on phosphorus removal capacities of basic oxygen furnace slag. Ecological Engineering 89:1–6
Hou B, Han H, Jia S, Zhuang H, Zhao Q, Xu P (2014) Effect of alkalinity on nitrite accumulation in treatment of coal chemical industry wastewater using moving bed biofilm reactor. Journal of Environmental Sciences 26(5):1014–1022
Kirby CS, Dennis A, Kahler A (2009) Applied geochemistry aeration to degas CO2 , increase pH, and increase iron oxidation rates for efficient treatment of net alkaline mine drainage. Applied Geochemistry 24(7):1175–1184
Klimeski A, Chardon WJ, Turtola E, Uusitalo R (2012) Potential and limitations of phosphate retention media in water protection : a process-based review of laboratory and field-scale tests. Agricultural and Food Science 21:206–223
Lee C, Park J, Kim S, Lee C, Park J, Kim S (2012) Phosphate removal from aqueous solutions using slag microspheres. Desalination and Water Treatment 44(1–3):229–236
Morse GK, Brett SW, Guy JA, Lester JNU (1998) Review: Phosphorus removal and recovery technologies. The Science of the Total Environment 212:69–81
Ngah WSW, Hanafiah MAKM (2008) Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents : a review. Bioresource Technology 99:3935–3948
Okochi NC, McMartin DW (2011) Laboratory investigations of stormwater remediation via slag: Effects of metals on phosphorus removal. Journal of Hazardous Materials 187(1–3):250–257
Panasiuk, O. (2010). Phosphorus removal and recovery from wastewater using magnetite. Royal Institute of Technology.
Park T, Ampunan V, Maeng S, Chung E (2017) Application of steel slag coated with sodium hydroxide to enhance precipitation-coagulation for phosphorus removal. Chemosphere 167:91–97
Penn, C., Livingston, S., Shedekar, V., King, K. and Williams, M., 2020. Performance of field-scale phosphorus removal structures utilizing steel slag for treatment of surface and subsurface drainage. Water, 12(2), p.443.
Pratt C, Shilton A, Pratt S, Haverkamp RG, Elmetri I (2007) Effects of redox potential and pH changes on phosphorus retention by melter slag filters treating wastewater. Environmental Science and Technology 41(18):6585–6590
Stumm W, Morgan JJ (1970) Aquatic chemistry. Wiley-Interscience, New York
Wilfert P, Kumar PS, Korving L, Witkamp G-J, van Loosdrecht MCM (2015) The relevance of phosphorus and iron chemistry to the recovery of phosphorus from wastewater: a review. Environmental Science & Technology 49(16):9400–9414
Xue Y, Hou H, Zhu S (2009a) Characteristics and mechanisms of phosphate adsorption onto basic oxygen furnace slag. Journal of Hazardous Materials 162:973–980
Xue Y, Hou H, Zhu S (2009b) Competitive adsorption of copper(II), cadmium(II), lead(II) and zinc(II) onto basic oxygen furnace slag. Journal of Hazardous Materials 162(1):391–401
Zhu L, Li X, Zhang C, Duan Z (2017) Pollutants’ release, redistribution and remediation of black smelly river sediment based on re-suspension and deep aeration of sediment. International Journal of Environmental Research and Public Health 14:374–384
Zuo M, Renman G, Gustafsson JP, Klysubun W (2018) Dual slag filters for enhanced phosphorus removal from domestic waste water: performance and mechanisms. Environmental Science and Pollution Research 25(8):7391–7400
Acknowledgements
The authors acknowledge Master thesis of Siti Zu Nurain Ahmad (Phosphate removal efficiencies and mechanisms of different chemical compositions of steel slag column filter system) from which the manuscript is derived. The authors also acknowledge the tables and figures and confirm that there are no third-party material in the manuscript. The authors wish to thank the Ministry of Higher Education (MOHE) for supporting this research under FRGS 1613 (New Insight into PO43- Removal Mechanism due to the Metal Oxides Concentration’s Variuosness in Steel Slag For Industrial Wastewater Treatment).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ahmad, S.Z.N., Al-Gheethi, A., Hamdan, R. et al. Efficiencies and mechanisms of steel slag with ferric oxides for removing phosphate from wastewater using a column filter system. Environ Sci Pollut Res 27, 35184–35194 (2020). https://doi.org/10.1007/s11356-020-09582-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09582-7