Abstract
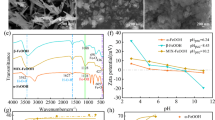
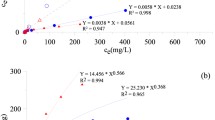
Arsenic in groundwater for human consumption has negative effects on human’s health worldwide. Due to the above, it is essential to invest in the development of new materials and more efficient technology for the elimination of such priority contaminants as arsenic. Therefore, in the present work, it was synthesized an amorphous hybrid material ZrOx-FeOx with a high density of OH groups, to improve the arsenic adsorption capacity of iron (FeOx) and zirconium (ZrOx) that makes up the bimetallic oxyhydroxide. The spectra of FT-IR and pKa’s distribution suggest that in the synthesized binary oxides, a new union between the two metallic elements is formed by means of an oxygen (metal-O-metal). In addition, TEM profiles suggest that there are chemical interactions between both metals since no individual particles of iron oxide and zirconium oxide were found. According to the results, the adsorption capacity of the ZrOx-FeOx material increases 4.5 and 1.4 times with respect to FeOx and ZrOx, respectively. At pH 6, the maximum adsorption capacity was 27 mg g−1, but at pH greater than 7, the arsenic adsorption capacity onto ZrOx-FeOx decreased 66%.

Graphical Abstract












Similar content being viewed by others
References
ALOthman ZA (2012) A review: fundamental aspects of silicate mesoporous materials. Materials 5:2874–2902. https://doi.org/10.3390/ma5122874
ALOthman ZA, Inamuddin, Mu N (2013) Recent developments in the synthesis, characterization and applications of zirconium (IV) based composite ion exchangers. J Inorg Organomet Polym Mater 23:257–269. https://doi.org/10.1007/s10904-012-9797-2
Al-Othman ZA, Naushad M, Inamuddin (2011) Organic–inorganic type composite cation exchanger poly-o-toluidine Zr (IV) tungstate: preparation, physicochemical characterization and its analytical application in separation of heavy metals. Chem Eng J 172:369–375. https://doi.org/10.1016/j.cej.2011.06.018
Arcibar-Orozco JA, Josue D-B, Rios-Hurtado JC, Rangel-Mendez JR (2014) Influence of iron content, surface area and charge distribution in the arsenic removal by activated carbons. Chem Eng J 249:201–209. https://doi.org/10.1016/j.cej.2014.03.096
Barrera S, M., Chavez G, M., Soto E, A.M., Velasquez O, C., Garcia S, M.A., Olvera T, L., & Rivera M, T. (2004). Synthesis of amorphous zirconium oxide with luminescent characteristics. 7 International Conference, Mexico. https://www.osti.gov/etdeweb/biblio/20534556. Accessed 22 Apr 2020
Berg M, Tran HC, Nguyen TC et al (2001) Arsenic contamination of groundwater and drinking water in Vietnam: a human health threat. Environ Sci Technol 35:2621–2626. https://doi.org/10.1021/es010027y
Biswas K, Bandhoyapadhyay D, Ghosh UC (2007) Adsorption kinetics of fluoride on iron (III)-zirconium (IV) hybrid oxide. Adsorption 13:83–94. https://doi.org/10.1007/s10450-007-9000-1
Biswas K, Gupta K, Ghosh UC (2009) Adsorption of fluoride by hydrous iron (III)–tin (IV) bimetal mixed oxide from the aqueous solutions. Chem Eng J 149:196–206. https://doi.org/10.1016/j.cej.2008.09.047
Chaudhry SA, Zaidi Z, Siddiqui SI (2017) Isotherm, kinetic and thermodynamics of arsenic adsorption onto Iron-Zirconium Binary Oxide-Coated Sand (IZBOCS): modelling and process optimization. J Mol Liq 229:230–240. https://doi.org/10.1016/j.molliq.2016.12.048
Choong TSY, Chuah TG, Robiah Y et al (2007) Arsenic toxicity, health hazards and removal techniques from water: an overview. Desalination 217:139–166. https://doi.org/10.1016/j.desal.2007.01.015
Cornell RM, Schwertmann U (2003) The iron oxides structure, properties, reactions, occurrences, and uses, 2nd, completely rev. and extended ed edn. Wiley-VCH, Weinheim
Deguillaume L, Leriche M, Desboeufs K et al (2005) Transition metals in atmospheric liquid phases: sources, reactivity, and sensitive parameters. Chem Rev 105:3388–3431. https://doi.org/10.1021/cr040649c
Ferreccio C, González C, Milosavjlevic V et al (2000) Lung cancer and arsenic concentrations in drinking water in Chile. Epidemiology 11:673–679
Gupta K, Ghosh UC (2009) Arsenic removal using hydrous nanostructure iron (III)–titanium (IV) binary mixed oxide from aqueous solution. J Hazard Mater 161:884–892. https://doi.org/10.1016/j.jhazmat.2008.04.034
Gupta K, Basu T, Ghosh UC (2009) Sorption characteristics of arsenic (V) for removal from water using agglomerated nanostructure iron (III)−zirconium (IV) bimetal mixed oxide. J Chem Eng Data 54:2222–2228. https://doi.org/10.1021/je900282m
Han B, Runnells T, Zimbron J, Wickramasinghe R (2002) Arsenic removal from drinking water by flocculation and microfiltration. Desalination 145:293–298. https://doi.org/10.1016/S0011-9164(02)00425-3
Hossain MF (2006) Arsenic contamination in Bangladesh—an overview. Agric Ecosyst Environ 113:1–16. https://doi.org/10.1016/j.agee.2005.08.034
Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133:1–16. https://doi.org/10.1016/S0378-4274(02)00084-X
Jadhav SV, Bringas E, Yadav GD et al (2015) Arsenic and fluoride contaminated groundwaters: a review of current technologies for contaminants removal. J Environ Manag 162:306–325. https://doi.org/10.1016/j.jenvman.2015.07.020
Jing C, Cui J, Huang Y, Li A (2012) Fabrication, characterization, and application of a composite adsorbent for simultaneous removal of arsenic and fluoride. ACS Appl Mater Interfaces 4:714–720. https://doi.org/10.1021/am2013322
Kim S-H, Kim K, Ko K-S et al (2012) Co-contamination of arsenic and fluoride in the groundwater of unconsolidated aquifers under reducing environments. Chemosphere 87:851–856. https://doi.org/10.1016/j.chemosphere.2012.01.025
Kosmulski M (2002) The significance of the points of zero charge of zirconium (hydr) oxide reported in the literature. J Dispers Sci Technol 23:529–538. https://doi.org/10.1081/DIS-120014021
Kumar M, Puri A (2012) A review of permissible limits of drinking water. Indian J Occup Environ Med 16:40–44. https://doi.org/10.4103/0019-5278.99696
Lakshmipathiraj P, Narasimhan BRV, Prabhakar S, Bhaskar Raju G (2006) Adsorption of arsenate on synthetic goethite from aqueous solutions. J Hazard Mater 136:281–287. https://doi.org/10.1016/j.jhazmat.2005.12.015
Mandal BK, Suzuki KT (2002) Arsenic round the world: a review. Talanta 58:201–235. https://doi.org/10.1016/S0039-9140(02)00268-0
Naushad M, Inamuddin, Rangreez TA, ALOthman ZA (2014) A mercury ion selective electrode based on poly-o-toluidine Zr(IV) tungstate composite membrane. J Electroanal Chem 713:125–130. https://doi.org/10.1016/j.jelechem.2013.12.002
Peters SC, Blum JD (2003) The source and transport of arsenic in a bedrock aquifer, New Hampshire, USA. Appl Geochem 18:1773–1787. https://doi.org/10.1016/S0883-2927(03)00109-4
Pierce ML, Moore CB (1982) Adsorption of arsenite and arsenate on amorphous iron hydroxide. Water Res 16:1247–1253. https://doi.org/10.1016/0043-1354(82)90143-9
Ren Z, Zhang G, Paul Chen J (2011) Adsorptive removal of arsenic from water by an iron–zirconium binary oxide adsorbent. J Colloid Interface Sci 358:230–237. https://doi.org/10.1016/j.jcis.2011.01.013
Rodríguez L (2006) Parámetros fisicoquímicos y concentración de flúor y arsénico en el agua de pozos de la ciudad de San Luis Potosí y zona conurbada. Alternativa de tratamiento: Adsorción de fluoruro y arsénico en la interfase Al2O3 activada/solución acuosa. Tesis de Maestría, Facultad de ciencias químicas, ingeniería y medicina. Universidad Autónoma de San Luis Potosí
Smedley PL, Nicolli HB, Macdonald DMJ et al (2002) Hydrogeochemistry of arsenic and other inorganic constituents in groundwaters from La Pampa, Argentina. Appl Geochem 17:259–284. https://doi.org/10.1016/S0883-2927(01)00082-8
Streat M, Hellgardt K, Newton NLR (2008) Hydrous ferric oxide as an adsorbent in water treatment: part 2. Adsorption studies. Process Saf Environ Prot 86:11–20. https://doi.org/10.1016/j.psep.2007.10.008
Velazquez-Jimenez LH, Hurt RH, Matos J, Rangel-Mendez JR (2014) Zirconium–carbon hybrid sorbent for removal of fluoride from water: oxalic acid mediated Zr (IV) assembly and adsorption mechanism. Environ Sci Technol 48:1166–1174. https://doi.org/10.1021/es403929b
Vences-Alvarez E, Velazquez-Jimenez LH, Chazaro-Ruiz LF et al (2015) Fluoride removal in water by a hybrid adsorbent lanthanum–carbon. J Colloid Interface Sci 455:194–202. https://doi.org/10.1016/j.jcis.2015.05.048
Vences-Alvarez E, Flores-Arciniega JL, Flores-Zuñiga H, Rangel-Mendez JR (2019) Fluoride removal from water by ceramic oxides from cerium and manganese solutions. J Mol Liq 286:110880. https://doi.org/10.1016/j.molliq.2019.110880
Yosca TH, Rittle J, Krest CM et al (2013) Iron (IV) hydroxide pKa and the role of thiolate ligation in C-H bond activation by cytochrome P450. Science 342:825–829. https://doi.org/10.1126/science.1244373
Zhang C, Li Y, Wang F et al (2017) Performance of magnetic zirconium-iron oxide nanoparticle in the removal of phosphate from aqueous solution. Appl Surf Sci 396:1783–1792. https://doi.org/10.1016/j.apsusc.2016.11.214
Acknowledgments
The authors appreciate the technical support of Carmen Rocha Medina, Guillermo Vidriales, Beatriz Rivera Escoto, Hector Silva Pereyra, and Dulce Partida. In addition, the authors thank Lucia Aldana Navarro for the assistance in the English writing of this manuscript.
Funding
This work was financially supported by the CONACYT project SEP-CB-2014-01-237118 and FORDECYT-2018-8-297525.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 366 kb)
Rights and permissions
About this article
Cite this article
Vences-Alvarez, E., Lopez-Valdivieso, A., Cházaro-Ruíz, L.F. et al. Enhanced arsenic removal from water by a bimetallic material ZrOx-FeOx with high OH density. Environ Sci Pollut Res 27, 33362–33372 (2020). https://doi.org/10.1007/s11356-020-09492-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09492-8