Abstract
Purpose
p-Chloroamphetamine (PCA) is a neurotoxin that selectively degenerates the serotonin (5-HT) axon terminals. In order to study the brain metabolic consequences induced by serotonergic denervation, a single dose of PCA (2.5 or 10 mg/kg i.p.) was administered to male adult rats.
Procedures
In vivo regional brain metabolism was evaluated 3 and 21 days after PCA (2.5 or 10 mg/kg; i.p.) injection by 2-deoxy-2-[18F] fluoro-d-glucose ([18F] FDG) positron emission tomography (PET). At day 22, the following markers of neurotoxicity were determined: (a) 5-HT axon terminal lesion by 5-HT transporter (SERT) autoradiography, (b) reactive gliosis by glial fibrillary acidic protein immunohistochemistry, and (c) eventual neurodegeneration by DAPI/Fluoro-Jade C labeling.
Results
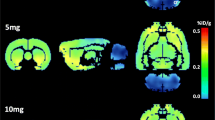
An average of 20 % reduction of [18F] FDG uptake in most brain areas was observed at day 21 under 10 mg/kg PCA treatment. Instead, 2.5 mg/kg PCA only reduced metabolic activity in neocortex. Likewise, the high dose of PCA exerted a strong decrease (>30 %) in SERT density in several 5-HT innervated regions, but no effect was found in midbrain raphe nuclei, the main source of serotonergic neurons. Although PCA induced astroglial activation both in hippocampus and cortex in response to axotomy, no signs of neuronal death in these areas were detected.
Conclusions
Overall, [18F] FDG PET revealed that the reduction of the brain metabolic activity induced by PCA is related to 5-HT axon terminal lesion, with no apparent affectation of neuronal viability.






Similar content being viewed by others
References
Brown P, Molliver ME (2000) Dual serotonin (5-HT) projections to the nucleus accumbens core and shell: relation of the 5-HT transporter to amphetamine-induced neurotoxicity. J Neurosci 20:1952–1963
Fuller RW (1992) Effects of p-chloroamphetamine on brain serotonin neurons. Neurochem Res 17:449–456
Colado MI, Murray TK, Green AR (1993) 5-HT loss in rat brain following 3,4-methylenedioxymethamphetamine (MDMA), p-chloroamphetamine and fenfluramine administration and effects of chlormethiazole and dizocilpine. Br J Pharmacol 108:583–589
Fernandez F, Aguerre S, Mormede P, Chaouloff F (2001) Differential sensitivities to the lethal, but not the neurotoxic, effects of p-chloroamphetamine in inbred rat strains. Neurosci Lett 297:53–57
Sugimoto Y, Ohkura M, Inoue K, Yamada J (2001) Involvement of serotonergic and dopaminergic mechanisms in hyperthermia induced by a serotonin-releasing drug, p-chloroamphetamine in mice. Eur J Pharmacol 430:265–268
Hsieh MT, Kuo LH, Tsai FH et al (2002) Effects of puerarin on scopolamine-, mecamylamine-, p-chloroamphetamine- and dizocilpine-induced inhibitory avoidance performance impairment in rats. Planta Med 68:901–905
Solana-Figueroa R, Salado-Castillo R, Quirarte GL et al (2002) Enhanced inhibitory avoidance training protects against the amnesic effect of p-chloroamphetamine. Life Sci 71:391–399
Maneepak M, le Grand S, Srikiatkhachorn A (2009) Serotonin depletion increases nociception-evoked trigeminal NMDA receptor phosphorylation. Headache 49:375–382
Kanno H, Sekiguchi K, Yamaguchi T et al (2009) Effect of yokukansan, a traditional Japanese medicine, on social and aggressive behaviour of para-chloroamphetamine-injected rats. J Pharm Pharmacol 61:1249–1256
Trulson ME, Jacobs BL (1976) Behavioral evidence for the rapid release of CNS serotonin by PCA and fenfluramine. Eur J Pharmacol 36:149–154
Sanders-Bush E, Sulser F (1970) P-chloroamphetamine: in vivo investigations on the mechanism of action of the selective depletion of cerebral serotonin. J Pharmacol Exp Ther 175:419–426
Lauzurica N, Garcia-Garcia L, Pinto S et al (2010) Changes in NPY and POMC, but not serotonin transporter, following a restricted feeding/repletion protocol in rats. Brain Res 1313:103–112
Rothman RB, Jayanthi S, Wang X et al (2003) High-dose fenfluramine administration decreases serotonin transporter binding, but not serotonin transporter protein levels, in rat forebrain. Synapse 50:233–239
Green AR, Mechan AO, Elliott JM et al (2003) The pharmacology and clinical pharmacology of 3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”). Pharmacol Rev 55:463–508
Hatzidimitriou G, McCann UD, Ricaurte GA (1999) Altered serotonin innervation patterns in the forebrain of monkeys treated with (+/−) 3,4-methylenedioxymethamphetamine seven years previously: factors influencing abnormal recovery. J Neurosci 19:5096–5107
McLane MW, McCann U, Ricaurte G (2011) Identifying the serotonin transporter signal in Western blot studies of the neurotoxic potential of MDMA and related drugs. Synapse 65:1368–1372
Thomas DM, Dowgiert J, Geddes TJ et al (2004) Microglial activation is a pharmacologically specific marker for the neurotoxic amphetamines. Neurosci Lett 367:349–354
Xie T, Tong L, McLane MW et al (2006) Loss of serotonin transporter protein after MDMA and other ring-substituted amphetamines. Neuropsychopharmacology 31:2639–2651
Nielsen K, Brask D, Knudsen GM, Aznar S (2006) Immunodetection of the serotonin transporter protein is a more valid marker for serotonergic fibers than serotonin. Synapse 59:270–276
Capela JP, Araujo SD, Costa VM et al (2013) The neurotoxicity of hallucinogenic amphetamines in primary cultures of hippocampal neurons. Neurotoxicology 34:254–263
Fukumura M, Cappon GD, Pu C, Broening HW, Vorhees CV (1998) A single dose model of methamphetamine-induced neurotoxicity in rats: effects on neostriatal monoamines and glial fibrillary acidic protein. Brain Res 806:1–7
de Cristóbal J, García-García L, Delgado M et al (2014) A longitudinal FDG-PET study of transgenic mice overexpressing GSK-3β in the brain. Curr Alzheim Res 11:175–181
Jupp B, O’Brien TJ (2007) Application of coregistration for imaging of animal models of epilepsy. Epilepsia 48(Suppl 4):82–89
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates. Elsevier Academic, San Diego
Rehavi M, Roz N, Weizman A (2002) Chronic clozapine, but not haloperidol, treatment affects rat brain vesicular monoamine transporter 2. Eur Neuropsychopharmacol 12:261–268
Schmued LC, Stowers CC, Scallet AC, Xu L (2005) Fluoro-Jade C results in ultra high resolution and contrast labeling of degenerating neurons. Brain Res 1035:24–31
Jacobs BL, Azmitia EC (1992) Structure and function of the brain serotonin system. Physiol Rev 72:165–229
Kanarik M, Matrov D, Kõiv K et al (2008) Changes in regional long-term oxidative metabolism induced by partial serotonergic denervation and chronic variable stress in rat brain. Neurochem Int 52:432–437
Tõnissaar M, Mällo T, Eller M et al (2008) Rat behavior after chronic variable stress and partial lesioning of 5-HT-ergic neurotransmission: effects of citalopram. Prog Neuropsychopharmacol Biol Psychiatry 32:164–177
Deleye S, Verhaeghe J, Wyffels L et al (2014) Towards a reproducible protocol for repetitive and semi-quantitative rat brain imaging with (18) F-FDG: exemplified in a memantine pharmacological challenge. Neuroimage 96:276–287
Biezonski DK, Meyer JS (2011) The nature of 3, 4-methylenedioxymethamphetamine (MDMA)-induced serotonergic dysfunction: evidence for and against the neurodegeneration hypothesis. Curr Neuropharmacol 9:84–90
Freo U, Larson DM, Tolliver T et al (1991) Parachloroamphetamine selectively alters regional cerebral metabolic responses to the serotonergic agonist metachlorophenylpiperazine in rats. Brain Res 544:17–25
Ridet JL, Malhotra SK, Privat A, Gage FH (1997) Reactive astrocytes: cellular and molecular cues to biological function. Trends Neurosci 23:570–577
Martínez-Clemente J, López-Arnau R, Abad S et al (2014) Dose and time-dependent selective neurotoxicity induced by mephedrone in mice. PLoS One 9:e99002. doi:10.1371/journal.pone.0099002
Thomas DM, Kuhn DM (2005) Cyclooxygenase-2 is an obligatory factor in methamphetamine-induced neurotoxicity. J Pharmacol Exp Ther 313:870–876
Gao Z, Zhu Q, Zhang Y et al (2013) Reciprocal modulation between microglia and astrocyte in reactive gliosis following the CNS injury. Mol Neurobiol 48:690–701
Mamounas LA, Mullen CA, O’Hearn E, Molliver ME (1991) Dual serotoninergic projections to forebrain in the rat: morphologically distinct 5-HT axon terminals exhibit differential vulnerability to neurotoxic amphetamine derivatives. J Comp Neurol 314:558–586
Riezzo I, Cerretani D, Fiore C et al (2010) Enzymatic–nonenzymatic cellular antioxidant defense systems response and immunohistochemical detection of MDMA, VMAT2, HSP70, and apoptosis as biomarkers for MDMA (Ecstasy) neurotoxicity. J Neurosci Res 88:905–916
Mahar I, Bambico FR, Mechawar N, Nobrega JN (2014) Stress, serotonin, and hippocampal neurogenesis in relation to depression and antidepressant effects. Neurosci Biobehav Rev 38:173–192
Dooley AE, Pappas IS, Parnavelas JG (1997) Serotonin promotes the survival of cortical glutamatergic neurons in vitro. Exp Neurol 148:205–214
Stankovski L, Alvarez C, Ouimet T et al (2007) Developmental cell death is enhanced in the cerebral cortex of mice lacking the brain vesicular monoamine transporter. J Neurosci 27:1315–1324
Acknowledgments
We thank Dr. José Luis López-Lacomba, director of the Instituto de Estudios Biofuncionales UCM, and Dr. Enrique Martínez-Campos for their assistance in the use of the fluorescence microscope.
This work was financially supported by grants from the Spanish Ministerio de Ciencia e Innovación (SAF2009-09020) and Comunidad de Madrid (I2M2, S2010/BMD-2349).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
García-García, L., Delgado, M., Al-Sayed, A.A. et al. In Vivo [18F] FDG PET Imaging Reveals that p-Chloroamphetamine Neurotoxicity is Associated with Long-Term Cortical and Hippocampal Hypometabolism. Mol Imaging Biol 17, 239–247 (2015). https://doi.org/10.1007/s11307-014-0794-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-014-0794-4