Abstract
Objective
This study aimed to reveal the urinary and serum metabolic pattern of endometrial cancer (EC) and establish diagnostic models to identify EC from controls, high-risk from low-risk EC, and type II from type I EC.
Method
This study included 146 EC patients (comprising 79 low-risk and 67 high-risk patients, including 124 type I and 22 type II) and 59 controls. The serum and urine samples were analyzed using ultraperformance liquid chromatography mass spectrometry. Analysis was used to elucidate the distinct metabolites and altered metabolic pathways. Receiver operating characteristic (ROC) analyses were employed to discover and validate the potential biomarker models.
Results
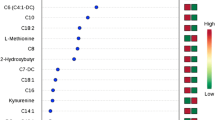
Serum and urine metabolomes displayed significant differences between EC and controls, with metabolites related to amino acid and nicotinamide metabolisms. The serum and urine panels distinguished these two groups with Area Under the Curve (AUC) of 0.821 and 0.902, respectively. The panel consisting of serum and urine metabolites demonstrated the best predictive ability (AUC = 0.953 and 0.976 in discovering and validation group). In comparing high-risk and low risk EC, differential metabolites were enriched in purine and glutamine metabolism. The AUC values for serum and urine panels were 0.818, and 0.843, respectively. The combined panel exhibited better predictive accuracy (0.881 in discovering group and 0.936 in external validation). In the comparison between type I and type II group, altered folic acid metabolism was identified. The serum, urine and combined panels discriminated these two groups with the AUC of 0.829, 0.913 and 0.922, respectively.
Conclusion
The combined urine and serum metabolome effectively revealed the metabolic patterns in EC patients, offering valuable diagnostic models for EC diagnosis and classification.





Similar content being viewed by others
Data availability
Data and materials are available from the corresponding author by reasonable request.
References
Sung, H., Ferlay, J., Siegel, R. L., et al. (2021). Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA., 71(3), 209–249.
McCluggage, W. G. (2006). My approach to the interpretation of endometrial biopsies and curettings. Journal of Clinical Pathology., 59(8), 801–812.
Nicholson, J. K., Lindon, J. C., & Holmes, E. (1999). “Metabonomics”: Understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica, 29(11), 1181–1189.
Bingol, K. (2018). Recent advances in targeted and untargeted metabolomics by NMR and MS/NMR methods. High-Throughput, 7(2), 7.
Kobayashi, T., Nishiumi, S., Ikeda, A., et al. (2013). A novel serum metabolomics-based diagnostic approach to pancreatic cancer. Cancer Epidemiology, Biomarkers & Prevention, 22(4), 571–579.
DeBerardinis, R. J., & Keshari, K. R. (2022). Metabolic analysis as a driver for discovery, diagnosis, and therapy. Cell, 185(15), 2678–2689.
Patti, G. J., Yanes, O., & Siuzdak, G. (2012). Innovation: Metabolomics: The apogee of the omics trilogy. Nature Reviews Molecular Cell Biology, 13(4), 263–269.
Raffone, A., Troisi, J., Boccia, D., et al. (2020). Metabolomics in endometrial cancer diagnosis: A systematic review. Acta Obstetricia Et Gynecologica Scandinavica., 99(9), 1135–1146.
Bahado-Singh, R. O., Lugade, A., Field, J., et al. (2017). Metabolomic prediction of endometrial cancer. Metabolomics, 14(1), 6.
Friedenreich, C. M., Derksen, J. W. G., Speidel, T., et al. (2020). Case-control study of endogenous sex steroid hormones and risk of endometrial cancer. Cancer Causes & Control, 31(2), 161–171.
Shao, X., Wang, K., Liu, X., et al. (2016). Screening and verifying endometrial carcinoma diagnostic biomarkers based on a urine metabolomic profiling study using UPLC-Q-TOF/MS. Clinica Chimica Acta, 463, 200–206.
Njoku, K., Campbell, A. E., Geary, B., et al. (2021). Metabolomic biomarkers for the detection of obesity-driven endometrial cancer. Cancers, 13(4), 8.
Troisi, J., Sarno, L., Landolfi, A., et al. (2018). Metabolomic signature of endometrial cancer. Journal of Proteome Research, 17(2), 804–812.
Knific, T., Vouk, K., Smrkolj, Š, et al. (2018). Models including plasma levels of sphingomyelins and phosphatidylcholines as diagnostic and prognostic biomarkers of endometrial cancer. The Journal of Steroid Biochemistry and Molecular Biology, 178, 312–321.
Paraskevaidi, M., Morais, C. L. M., Ashton, K. M., et al. (2020). Detecting endometrial cancer by blood spectroscopy: A diagnostic cross-sectional study. Cancers, 12(5), 89.
Oaknin, A., Bosse, T. J., Creutzberg, C. L., et al. (2022). Endometrial cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Annals of Oncology, 58, 9.
Abu-Rustum, N. R., Yashar, C. M., Bradley, K., et al. (2021). NCCN Guidelines® insights: Uterine neoplasms, version 3.2021. Journal of the National Comprehensive Cancer Network, 19(8), 888–895.
Zhang, J., Yang, W., Li, S., et al. (2016). An intelligentized strategy for endogenous small molecules characterization and quality evaluation of earthworm from two geographic origins by ultra-high performance HILIC/QTOF MS(E) and Progenesis QI. Analytical and Bioanalytical Chemistry, 408(14), 3881–3890.
Chen, J., Zhao, X., Fritsche, J., et al. (2008). Practical approach for the identification and isomer elucidation of biomarkers detected in a metabonomic study for the discovery of individuals at risk for diabetes by integrating the chromatographic and mass spectrometric information. Analytical Chemistry, 80(4), 1280–1289.
Ananieva, E. A., & Wilkinson, A. C. (2018). Branched-chain amino acid metabolism in cancer. Current Opinion in Clinical Nutrition and Metabolic Care, 21(1), 64–70.
Sivanand, S., & Vander Heiden, M. G. (2020). Emerging roles for branched-chain amino acid metabolism in cancer. Cancer Cell, 37(2), 147–156.
Zhang, L., & Han, J. (2017). Branched-chain amino acid transaminase 1 (BCAT1) promotes the growth of breast cancer cells through improving mTOR-mediated mitochondrial biogenesis and function. Biochemical and Biophysical Research Communications, 486(2), 224–231.
Sheen, J. H., Zoncu, R., Kim, D., et al. (2011). Defective regulation of autophagy upon leucine deprivation reveals a targetable liability of human melanoma cells in vitro and in vivo. Cancer Cell, 19(5), 613–628.
Nishio, Y., Kakizoe, T., Ohtani, M., et al. (1986). L-isoleucine and L-leucine: Tumor promoters of bladder cancer in rats. Science (new York, NY)., 231(4740), 843–845.
Wang, P., Wu, S., Zeng, X., et al. (2018). BCAT1 promotes proliferation of endometrial cancer cells through reprogrammed BCAA metabolism. International Journal of Clinical and Experimental Pathology., 11(12), 5536–5546.
Dossus, L., Kouloura, E., Biessy, C., et al. (2021). Prospective analysis of circulating metabolites and endometrial cancer risk. Gynecologic Oncology., 162(2), 475–481.
Troisi, J., Mollo, A., Lombardi, M., et al. (2022). The metabolomic approach for the screening of endometrial cancer: validation from a large cohort of women scheduled for gynecological surgery. Biomolecules, 12(9), 78.
Warburg, O. (1956). On respiratory impairment in cancer cells. Science (new York, NY)., 124(3215), 269–270.
Pissios, P. (2017). Nicotinamide N-methyltransferase: more than a vitamin B3 clearance enzyme. Trends in Endocrinology and Metabolism, 28(5), 340–353.
Sartini, D., Muzzonigro, G., Milanese, G., et al. (2013). Upregulation of tissue and urinary nicotinamide N-methyltransferase in bladder cancer: Potential for the development of a urine-based diagnostic test. Cell Biochemistry and Biophysics., 65(3), 473–483.
Tomida, M., Mikami, I., Takeuchi, S., et al. (2009). Serum levels of nicotinamide N-methyltransferase in patients with lung cancer. Journal of Cancer Research and Clinical Oncology, 135(9), 1223–1229.
Hu, D., Du, J., Cheng, Y., et al. (2023). Comprehensive analysis of a NAD+ metabolism-derived gene signature to predict the prognosis and immune landscape in endometrial cancer. Biomolecules & Biomedicine, 5, 78.
Pedley, A. M., & Benkovic, S. J. (2017). A new view into the regulation of purine metabolism: The purinosome. Trends in Biochemical Sciences., 42(2), 141–154.
Jové, M., Gatius, S., Yeramian, A., et al. (2016). Metabotyping human endometrioid endometrial adenocarcinoma reveals an implication of endocannabinoid metabolism. Oncotarget, 7(32), 52364–52374.
Griffin, J. L., Pole, J. C., Nicholson, J. K., et al. (2003). Cellular environment of metabolites and a metabonomic study of tamoxifen in endometrial cells using gradient high resolution magic angle spinning 1H NMR spectroscopy. Biochimica Et Biophysica Acta, 1619(2), 151–158.
Altman, B. J., Stine, Z. E., & Dang, C. V. (2016). From Krebs to clinic: Glutamine metabolism to cancer therapy. Nature Reviews Cancer, 16(11), 749.
Moses, M. A., & Neckers, L. (2015). The GLU that holds cancer together: Targeting GLUtamine transporters in breast cancer. Cancer Cell, 27(3), 317–319.
Hensley, C. T., Wasti, A. T., & DeBerardinis, R. J. (2013). Glutamine and cancer: Cell biology, physiology, and clinical opportunities. The Journal of Clinical Investigation, 123(9), 3678–3684.
Zhai, L., Yang, X., Cheng, Y., et al. (2023). Glutamine and amino acid metabolism as a prognostic signature and therapeutic target in endometrial cancer. Cancer Medicine, 12(15), 16337–16358.
Skorupa, A., Poński, M., Ciszek, M., et al. (2021). Grading of endometrial cancer using (1)H HR-MAS NMR-based metabolomics. Scientific Reports, 11(1), 18160.
Raffone, A., Travaglino, A., Mascolo, M., et al. (2020). Histopathological characterization of ProMisE molecular groups of endometrial cancer. Gynecologic Oncology, 157(1), 252–259.
Bokhman, J. V. (1983). Two pathogenetic types of endometrial carcinoma. Gynecologic Oncology, 15(1), 10–17.
Song, J., Medline, A., Mason, J. B., et al. (2000). Effects of dietary folate on intestinal tumorigenesis in the apcMin mouse. Cancer Research, 60(19), 5434–5440.
Smith, A. D., Kim, Y. I., & Refsum, H. (2008). Is folic acid good for everyone? The American Journal of Clinical Nutrition, 87(3), 517–533.
Zhao, Y., Guo, C., Hu, H., et al. (2017). Folate intake, serum folate levels and esophageal cancer risk: An overall and dose-response meta-analysis. Oncotarget, 8(6), 10458–10469.
Galeone, C., Edefonti, V., Parpinel, M., et al. (2015). Folate intake and the risk of oral cavity and pharyngeal cancer: A pooled analysis within the International Head and Neck Cancer Epidemiology Consortium. International Journal of Cancer., 136(4), 904–914.
Wang, R., Zheng, Y., Huang, J. Y., et al. (2014). Folate intake, serum folate levels, and prostate cancer risk: A meta-analysis of prospective studies. BMC Public Health, 14, 1326.
Albertini, A. F., Devouassoux-Shisheboran, M., & Genestie, C. (2012). Pathology of endometrioid carcinoma. Bulletin Du Cancer., 99(1), 7–12.
Du, L., Wang, Y., Zhang, H., et al. (2016). Folate intake and the risk of endometrial cancer: A meta-analysis. Oncotarget, 7(51), 85176–85184.
Liu, J. J., Hazra, A., Giovannucci, E., et al. (2013). One-carbon metabolism factors and endometrial cancer risk. British Journal of Cancer, 108(1), 183–187.
Wang, Z., Liu, X., Liu, X., et al. (2019). UPLC-MS based urine untargeted metabolomic analyses to differentiate bladder cancer from renal cell carcinoma. BMC Cancer, 19(1), 1195.
Gao, Y. (2021). On research and translation of urinary biomarkers. Advances in Experimental Medicine and Biology., 1306, 101–108.
Gao, Y. (2013). Urine-an untapped goldmine for biomarker discovery? Science China Life Sciences, 56(12), 1145–1146.
Acknowledgements
The authors express their gratitude to the entire faculty, nursing staff, and personnel at the Department of Obstetrics & Gynecology in PUMCH for their outstanding patient care. Additionally, the authors extend their heartfelt thanks to all the patients and their families for their invaluable contributions to this research.
Funding
National High Level Hospital Clinical Research Funding (Grant no. 2022-PUMCH-B-082) and The Natural Science Foundation of Shandong Province (Grant no. ZR2023QH426).
Author information
Authors and Affiliations
Contributions
Conceptualization: JC, DC, and WS; Data curvation: JC, HL, JS, FQ, XL, JL, DC, JY, MY, HZ, JW, YZ, NC, PP, and KS; Formal analysis: JC, HL, DC, and WS; Software: JC and HL; Writing—original draft: JC; Writing—review and editing: DC, and WS. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
This study was approved by the Ethics Committee of Peking Union Medical College Hospital (PUMCH) (ZS-2666).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11306_2023_2085_MOESM2_ESM.tif
Metabolic analysis of Endometrial cancers and controls. A. Serum metabolic analysis of PCA; B. Urine metabolic analysis of PCA; C. 100 permutation test of OPLS-DA model in serum samples; D. 100 permutation test of the OPLS-DA model in urine samples; E. ROC curve of external validation of the serum metabolites; F. ROC curve of external validation of the urine metabolites; G. ROC curve of external validation of the biomarker panel Supplementary file2 (TIF 28000 kb)
11306_2023_2085_MOESM3_ESM.tif
Metabolic analysis of Endometrial cancers and controls. A. Serum metabolic analysis of PCA; B. Urine metabolic analysis of PCA; C. 100 permutation test of OPLS-DA model in serum samples; D. 100 permutation test of the OPLS-DA model in urine samples; E. ROC curve of external validation of the serum metabolites; F. ROC curve of external validation of the urine metabolites; G. ROC curve of external validation of the biomarker panel. Supplementary file3 (TIF 10800 kb)
11306_2023_2085_MOESM4_ESM.tif
Analysis of metabolic analysis between type I endometrial cancers and type II endometrial cancers. A. Serum metabolic analysis of PCA; B. Urine metabolic analysis of PCA; C. 100 permutation test of OPLS-DA model in serum samples; D. 100 permutation test of the OPLS-DA model in urine samples; E. ROC curve of 10-fold cross-validation of the serum metabolites; F. ROC curve of 10-fold cross-validation of the urine metabolites; G. ROC curve of 10-fold cross-validation of the biomarker panel. Supplementary file4 (TIF 10800 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Lu, H., Cao, D. et al. Urine and serum metabolomic analysis of endometrial cancer diagnosis and classification based on ultra-performance liquid chromatography mass spectrometry. Metabolomics 20, 18 (2024). https://doi.org/10.1007/s11306-023-02085-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-02085-9