Abstract
Introduction
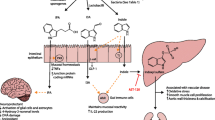
Phytoestrogens found in soy, fruits, peanuts, and other legumes, have been identified as metabolites capable of providing beneficial effects in multiple pathological conditions due to their ability to mimic endogenous estrogen. Interestingly, the health-promoting effects of some phytoestrogens, such as isoflavones, are dependent on the presence of specific gut bacteria. Specifically, gut bacteria can metabolize isoflavones into equol, which has a higher affinity for endogenous estrogen receptors compared to dietary isoflavones. We have previously shown that patients with multiple sclerosis (MS), a neuroinflammatory disease, lack gut bacteria that are able to metabolize phytoestrogen. Further, we have validated the importance of both isoflavones and phytoestrogen-metabolizing gut bacteria in disease protection utilizing an animal model of MS. Specifically, we have shown that an isoflavone-rich diet can protect from neuroinflammatory diseases, and that protection was dependent on the ability of gut bacteria to metabolize isoflavones into equol. Additionally, mice on a diet with isoflavones showed an anti-inflammatory response compared to the mice on a diet lacking isoflavones. However, it is unknown how isoflavones and/or equol mediates their protective effects, especially their effects on host metabolite levels.
Objectives
In this study, we utilized untargeted metabolomics to identify metabolites found in plasma that were modulated by the presence of dietary isoflavones.
Results
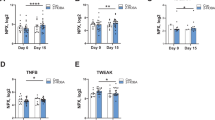
We found that the consumption of isoflavones increased anti-inflammatory monounsaturated fatty acids and beneficial polyunsaturated fatty acids while reducing pro-inflammatory glycerophospholipids, sphingolipids, phenylalanine metabolism, and arachidonic acid derivatives.
Conclusion
Isoflavone consumption alters the systemic metabolic landscape through concurrent increases in monounsaturated fatty acids and beneficial polyunsaturated fatty acids plus reduction in pro-inflammatory metabolites and pathways. This highlights a potential mechanism by which an isoflavone diet may modulate immune-mediated disease.




Similar content being viewed by others
Data Availability
Metabolomic data utilized for analysis is in Table S1.
References
Aboushanab, S. A., Khedr, S. M., Gette, I. F., Danilova, I. G., Kolberg, N. A., Ravishankar, G. A., Ambati, R. R., & Kovaleva, E. G. (2021). Isoflavones derived from plant raw materials: bioavailability, anti-cancer, anti-aging potentials, and microbiome modulation. Critical Reviews in Food Science and Nutrition. Ltd: Bellwether Publishing. https://doi.org/10.1080/10408398.2021.1946006
Akazawa, Y., Morisaki, T., Fukuda, H., Norimatsu, K., Shiota, J., Hashiguchi, K., Tabuchi, M., Kitayama, M., Matsushima, K., Yamaguchi, N., Kondo, H., Fujita, F., Takeshita, H., Nakao, K., & Takeshima, F. (2021). Significance of serum palmitoleic acid levels in inflammatory bowel disease. Scientific Reports, 11(1), https://doi.org/10.1038/s41598-021-95923-6
Bjørnevik, K., Chitnis, T., Ascherio, A., & Munger, K. L. (2017). Polyunsaturated fatty acids and the risk of multiple sclerosis. Multiple Sclerosis, 23(14), 1830–1838. https://doi.org/10.1177/1352458517691150
Bjornevik, K., Myhr, K. M., Beiske, A., Bjerve, K. S., Holmøy, T., Hovdal, H., Midgard, R., Riise, T., Wergeland, S., & Torkildsen, Ø. (2019). α-Linolenic acid is associated with MRI activity in a prospective cohort of multiple sclerosis patients. Multiple Sclerosis Journal, 25(7), 987–993. https://doi.org/10.1177/1352458518779925
Chamras, H., Barsky, S. H., Ardashian, A., Navasartian, D., Heber, D., & Glaspy, J. A. (2005). Novel Interactions of Vitamin E and Estrogen in Breast Cancer. Nutrition and Cancer, 52(1), 43–48. https://doi.org/10.1207/s15327914nc5201_6
Chen, J., Chia, N., Kalari, K. R., Yao, J. Z., Novotna, M., Soldan, M. M. P., Luckey, D. H., Marietta, E., Jeraldo, P. R., Chen, X., Weinshenker, B. G., Rodriguez, M., Kantarci, O. H., Nelson, H., Murray, J. A., & Mangalam, A. K. (2016). Multiple sclerosis patients have a distinct gut microbiota compared to healthy controls. Scientific Reports, 6. https://doi.org/10.1038/srep28484
Coll, T., Eyre, E., Rodríguez-Calvo, R., Palomer, X., Sánchez, R. M., Merlos, M., Laguna, J. C., & Vázquez-Carrera, M. (2008). Oleate reverses palmitate-induced insulin resistance and inflammation in skeletal muscle cells. Journal of Biological Chemistry, 283(17), 11107–11116. https://doi.org/10.1074/jbc.M708700200
Díaz-Rubio, M. E., Pérez-Jiménez, J., Martínez-Bartolomé, M., Álvarez, I., & Saura-Calixto, F. (2015). Regular Consumption of an Antioxidant-rich Juice Improves Oxidative Status and Causes Metabolome Changes in Healthy Adults. Plant Foods for Human Nutrition, 70(1), 9–14. https://doi.org/10.1007/s11130-014-0455-4
el Aidy, S., Burokas, A., Cristiano, C., Marzal, P. L., Pardo, P. P., Zheng, Y., Bek, M. K., Prince, N. Z., Marzal, P., Garssen, L. N., Perez Pardo, J., P., & Kraneveld, A. D. (2021). The Role of Bacterial-Derived Aromatic Amino Acids Metabolites Relevant in Autism Spectrum Disorders: A Comprehensive Review. https://doi.org/10.3389/fnins.2021.738220
François, M., Karpe, A., Liu, J. W., Beale, D., Hor, M., Hecker, J., Faunt, J., Maddison, J., Johns, S., Doecke, J., Rose, S., & Leifert, W. R. (2021). Salivaomics as a Potential Tool for Predicting Alzheimer’s Disease During the Early Stages of Neurodegeneration. Journal of Alzheimer’s Disease, 82, 1301–1313. https://doi.org/10.3233/JAD-210283
Gao, S., Quick, C., Guasch-Ferre, M., Zhuo, Z., Hutchinson, J. M., Su, L., Hu, F., Lin, X., & Christiani, D. (2021). The Association Between Inflammatory and Oxidative Stress Biomarkers and Plasma Metabolites in a Longitudinal Study of Healthy Male Welders. Journal of Inflammation Research, 14, 2825–2839. https://doi.org/10.2147/JIR.S316262
Ghimire, S., Cady, N. M., Lehman, P., Peterson, S. R., Shahi, S. K., Rashid, F., Giri, S., & Mangalam, A. K. (n.d.). Dietary Isoflavones Alter Gut Microbiota and Lipopolysaccharide Biosynthesis to Reduce Inflammation.Gut Microbes, 14(1),2127446. https://doi.org/10.1080/19490976.2022.2127446
Gryp, T., Vanholder, R., Vaneechoutte, M., & Glorieux, G. (2017). p-cresyl sulfate. Toxins, 9(2), https://doi.org/10.3390/TOXINS9020052
Hannun, Y. A., Gault, C. R., & Obeid, L. M. (2010). Sphingolipids as Signaling and Regulatory Molecules An Overview of Sphingolipid Metabolism: From Synthesis to Breakdown
Jana, A., & Pahan, K. (2010). Sphingolipids in Multiple Sclerosis. Neuromolecular Medicine. https://doi.org/10.1007/s12017-010-8128-4
Jensen, S. N., Cady, N. M., Shahi, S. K., Peterson, S. R., Gupta, A., Gibson-Corley, K. N., & Mangalam, A. K. (2021). Isoflavone diet ameliorates experimental autoimmune encephalomyelitis through modulation of gut bacteria depleted in patients with multiple sclerosis. In Sci. Adv (Vol. 7, Issue 9). https://doi.org/https://www.science.org
Jewell, D. E., & Jackson, M. I. (2022). Dietary Betaine and Fatty Acids Change Circulating Single-Carbon Metabolites and Fatty Acids in the Dog. Animals, 12(6), https://doi.org/10.3390/ani12060768
Johnson, E. L., Heaver, S. L., Waters, J. L., Kim, B. I., Bretin, A., Goodman, A. L., Gewirtz, A. T., Worgall, T. S., & Ley, R. E. (2020). Sphingolipids produced by gut bacteria enter host metabolic pathways impacting ceramide levels. Nature Communications, 11(1), 2471. https://doi.org/10.1038/s41467-020-16274-w
Kassambara, A. (2020). ggpubr: “ggplot2” Based Publication Ready Plots. R package version 0.4.0
Lee, J. S., Wang, R. X., Goldberg, M. S., Clifford, G. P., Kao, D. J., & Colgan, S. P. (2020). Microbiota-Sourced Purines Support Wound Healing and Mucous Barrier Function. IScience, 23(6), 101226. https://doi.org/10.1016/j.isci.2020.101226
Li, Z., & Vance, D. E. (2008). Phosphatidylcholine and choline homeostasis. In Journal of Lipid Research (Vol. 49, Issue 6, pp. 1187–1194). https://doi.org/10.1194/jlr.R700019-JLR200
Luo, Q., Cheng, D., Huang, C., Li, Y., Lao, C., Xia, Y., Liu, W., Gong, X., Hu, D., Li, B., He, X., & Chen, Z. (2019). Improvement of Colonic Immune Function with Soy Isoflavones in High-Fat Diet-Induced Obese Rats. Molecules, 24(6), 1139. https://doi.org/10.3390/molecules24061139
Malashree, L., Mudgil, P., Dagar, S. S., Kumar, S., & Puniya, A. K. (2012). β-Glucosidase Activity of Lactobacilli for Biotransformation of Soy Isoflavones. Food Biotechnology, 26(2), 154–163. https://doi.org/10.1080/08905436.2012.670832
Mangalam, A., Poisson, L., Nemutlu, E., Datta, I., Denic, A., Dzeja, P., Rodriguez, M., Rattan, R., & Giri, S. (2013). Profile of Circulatory Metabolites in an Animal Model of Multiple Sclerosis using Global Metabolomics. Journal of Clinical & Cellular Immunology, 04(03), https://doi.org/10.4172/2155-9899.1000150
Mangalam, A., Yadav, M., & Yadav, R. (2021). The emerging world of microbiome in autoimmune disorders: Opportunities and challenges. Indian Journal of Rheumatology (Vol, 16(1), 57–72. https://doi.org/10.4103/injr.injr_210_20. Wolters Kluwer Medknow Publications
Mayo, B., Vázquez, L., & Flórez, A. B. (2019a). Equol: A bacterial metabolite from the Daidzein isoflavone and its presumed beneficial health effects. Nutrients, 11(9), https://doi.org/10.3390/NU11092231
Mayo, B., Vázquez, L., & Flórez, A. B. (2019b). Equol: A Bacterial Metabolite from The Daidzein Isoflavone and Its Presumed Beneficial Health Effects. Nutrients, 11(9), 2231. https://doi.org/10.3390/nu11092231
Mcginley, M. P., & Cohen, J. A. (2021). Therapeutics Sphingosine 1-phosphate receptor modulators in multiple sclerosis and other conditions. The Lancet, 398, 1184–1194. https://doi.org/10.1016/S0140-6736(21)00244-0
Mehta, A. K., Singh, B. P., Arora, N., & Gaur, S. N. (2010). Choline attenuates immune inflammation and suppresses oxidative stress in patients with asthma. Immunobiology, 215(7), 527–534. https://doi.org/10.1016/j.imbio.2009.09.004
Experimental Procedures. Metabolon Help Metabolon Inc, & Center (2022). https://doi.org/https://help.metabolon.com/docs/microbiome-smartpanel/knowledge-base/experimental-procedures/
Mezei, O., Li, Y., Mullen, E., Ross-Viola, J. S., & Shay, N. F. (2006). Dietary isoflavone supplementation modulates lipid metabolism via PPARalpha-dependent and -independent mechanisms. Physiological Genomics, 26(1), 8–14. https://doi.org/10.1152/physiolgenomics.00155.2005
Munger, K., Bjornevik, K., Cortese, M., Edan, G., Freedman, M., Hartung, H. P., Montalban, X., Sandbrink, R., Radue, E. W., Barkhof, F., Wicklein, E. M., Kappos, L., & Ascherio, A. (2022). A Prospective Study of Serum Levels of Polyunsaturated Fatty Acids and Effects on Multiple Sclerosis Disease Activity and Progression (S40.006). In Supplement) THURSDAY (Vol. 98, Issue 7)
Nemazannikova, N., Mikkelsen, K., Stojanovska, L., Blatch, G. L., & Apostolopoulos, V. (2018). Is there a Link between Vitamin B and Multiple Sclerosis? Medicinal Chemistry, 14(2), https://doi.org/10.2174/1573406413666170906123857
Nogueras, L., Gonzalo, H., Jové, M., Sol, J., Gil-Sanchez, A., Hervás, J., Valcheva, P., Solana, M. J., peralta, S., pamplona, R., & Brieva, L. (2019). Lipid profile of cerebrospinal fluid in multiple sclerosis patients: A potential tool for diagnosis. Scientific Reports, 9, 11313. https://doi.org/10.1038/s41598-019-47906-x
Ntranos, A., Park, H. J., Wentling, M., Tolstikov, V., Amatruda, M., Inbar, B., Kim-Schulze, S., Frazier, C., Button, J., Kiebish, M. A., Lublin, F., Edwards, K., & Casaccia, P. (2022). Bacterial neurotoxic metabolites in multiple sclerosis cerebrospinal fluid and plasma. Brain, 145(2), 569–583. https://doi.org/10.1093/brain/awab320
Oliphant, K., & Allen-Vercoe, E. (2019). Macronutrient metabolism by the human gut microbiome: major fermentation by-products and their impact on host health. Microbiome, 7(1), 91. https://doi.org/10.1186/s40168-019-0704-8
Pabich, M., & Materska, M. (2019). Biological effect of soy isoflavones in the prevention of civilization diseases. Nutrients, 11(7), https://doi.org/10.3390/nu11071660
Pang, Z., Chong, J., Zhou, G., de Lima Morais, D. A., Chang, L., Barrette, M., Gauthier, C., Jacques, P., Li, S., & Xia, J. (2021). MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Research, 49(W1), W388–W396. https://doi.org/10.1093/nar/gkab382
Pinheiro de Oliveira, F., Mendes, R. H., Dobbler, P. T., Mai, V., Pylro, V. S., Waugh, S. G., Vairo, F., Refosco, L. F., Roesch, L. F. W., & Schwartz, I. V. D. (2016). Phenylketonuria and Gut Microbiota: A Controlled Study Based on Next-Generation Sequencing. PLOS ONE, 11(6), e0157513. https://doi.org/10.1371/journal.pone.0157513
Poisson, L. M., Suhail, H., Singh, J., Datta, I., Deni, A., Labuzek, K., Hoda, N., Shankar, A., Kumar, A., Cerghet, M., Elias, S., Mohney, R. P., Rodriguez, M., Rattan, R., Mangalam, A. K., & Giri, S. (2015). Untargeted plasma metabolomics identifies endogenous metabolite with drug-like properties in chronic animal model of multiple sclerosis. Journal of Biological Chemistry, 290(52), 30697–30712. https://doi.org/10.1074/jbc.M115.679068
Poisson, L. M., Suhail, H., Singh, J., Datta, I., Denic, A., Labuzek, K., Hoda, M. N., Shankar, A., Kumar, A., Cerghet, M., Elias, S., Mohney, R. P., Rodriguez, M., Rattan, R., Mangalam, A. K., & Giri, S. (2015). Untargeted Plasma Metabolomics Identifies Endogenous Metabolite with Drug-like Properties in Chronic Animal Model of Multiple Sclerosis. The Journal of Biological Chemistry, 290(52), 30697–30712. https://doi.org/10.1074/jbc.M115.679068
Pompura, S. L., Wagner, A., Kitz, A., LaPerche, J., Yosef, N., Dominguez-Villar, M., & Hafler, D. A. (2021). Oleic acid restores suppressive defects in tissue-resident FOXP3 Tregs from patients with multiple sclerosis. Journal of Clinical Investigation, 131(2), https://doi.org/10.1172/JCI138519
R Core Team (2022). R: A Language and Environment for Statistical Computing (4.1.1)
Rangel-Huerta, O. D., Aguilera, C. M., Perez-de-la-Cruz, A., Vallejo, F., Tomas-Barberan, F., Gil, A., & Mesa, M. D. (2017). A serum metabolomics-driven approach predicts orange juice consumption and its impact on oxidative stress and inflammation in subjects from the BIONAOS study. Molecular Nutrition and Food Research, 61(2), https://doi.org/10.1002/mnfr.201600120
Ravaut, G., Légiot, A., Bergeron, K. F., & Mounier, C. (2020). Monounsaturated Fatty Acids in Obesity-Related Inflammation. International Journal of Molecular Sciences, 22(1), 330. https://doi.org/10.3390/ijms22010330
Ricciotti, E., & Fitzgerald, G. A. (2011). Prostaglandins and Inflammation. Arteriosclerosis, Thrombosis, and Vascular Biology. https://doi.org/10.1161/ATVBAHA.110.207449
Sathyapalan, T., Aye, M., Rigby, A. S., Thatcher, N. J., Dargham, S. R., Kilpatrick, E. S., & Atkin, S. L. (2018). Soy isoflavones improve cardiovascular disease risk markers in women during the early menopause. Nutrition Metabolism and Cardiovascular Diseases, 28(7), 691–697. https://doi.org/10.1016/j.numecd.2018.03.007
Spiegel, S., & Milstien, S. (2011). The outs and the ins of sphingosine-1-phosphate in immunity. Nature Reviews Immunology (Vol, 11(6), 403–415. https://doi.org/10.1038/nri2974
Storm-Larsen, C., Myhr, K. M., Farbu, E., Midgard, R., Nyquist, K., Broch, L., Berg-Hansen, P., Buness, A., Holm, K., Ueland, T., Fallang, L. E., Burum-Auensen, E., Hov, J. R., & Holmøy, T. (2019). Gut microbiota composition during a 12-week intervention with delayed-release dimethyl fumarate in multiple sclerosis-a pilot trial. https://doi.org/10.1177/2055217319888767
Sun, Y., Tao, W., Huang, H., Ye, X., & Sun, P. (2019). Flavonoids, phenolic acids, carotenoids and antioxidant activity of fresh eating citrus fruits, using the coupled in vitro digestion and human intestinal HepG2 cells model. Food Chemistry, 279, 321–327. https://doi.org/10.1016/j.foodchem.2018.12.019
Tan, J., Huang, C., Luo, Q., & Chen, Z. (2019). Soy Isoflavones Ameliorate Fatty Acid Metabolism of Visceral Adipose Tissue by Increasing the AMPK Activity in Male Rats with Diet-Induced Obesity (DIO). Molecules, 24(15), 2809. https://doi.org/10.3390/molecules24152809
Torres Santiago, G., Serrano Contreras, J. I., Meléndez Camargo, M. E., & Zepeda Vallejo, L. G. (2019). NMR-based metabonomic approach reveals changes in the urinary and fecal metabolome caused by resveratrol. Journal of Pharmaceutical and Biomedical Analysis, 162, 234–241. https://doi.org/10.1016/j.jpba.2018.09.025
Wang, S., Li, H., Song, M., Tao, Z., Wu, T., He, Z., Zhao, X., Wu, K., & Liu, X. S. (2021). Copy number signature analysis tool and its application in prostate cancer reveals distinct mutational processes and clinical outcomes. PLOS Genetics, 17(5), e1009557. https://doi.org/10.1371/journal.pgen.1009557
Warner, M., Huang, B., & Gustafsson, J. A. (2017). Estrogen Receptor β as a Pharmaceutical Target. Trends in Pharmacological Sciences (38 vol., pp. 92–99). Elsevier Ltd. 1https://doi.org/10.1016/j.tips.2016.10.006
Wickham, H. (2016). ggplot2: Elegant Graphics for Data Analysis. In ggplot2: Elegant Graphics for Data Analysis (978th-3rd-319th-24277th–4th ed.)
Wickham, H., Averick, M., Bryan, J., Chang, W., McGowan, L., François, R., Grolemund, G., Hayes, A., Henry, L., Hester, J., Kuhn, M., Pedersen, T., Miller, E., Bache, S., Müller, K., Ooms, J., Robinson, D., Seidel, D., Spinu, V., & Yutani, H. (2019). Welcome to the Tidyverse. Journal of Open Source Software, 4(43), 1686. https://doi.org/10.21105/joss.01686
Wickham, H., Francois, R., Henry, L., & Muller, K. (2022). dplyr: A Grammar of Data Manipulation. R package version 1.0.9
Wishart, D. S., Guo, A., Oler, E., Wang, F., Anjum, A., Peters, H., Dizon, R., Sayeeda, Z., Tian, S., Lee, B. L., Berjanskii, M., Mah, R., Yamamoto, M., Jovel, J., Torres-Calzada, C., Hiebert-Giesbrecht, M., Lui, V. W., Varshavi, D., Varshavi, D., & Gautam, V. (2022). HMDB 5.0: the Human Metabolome Database for 2022. Nucleic Acids Research, 50. https://doi.org/10.1093/nar/gkab1062
Xiao, C. W. (2008). Effect of soy proteins and isoflavones on lipid metabolism and involved gene expression. Frontiers in Bioscience, 13(13), 2660. https://doi.org/10.2741/2873
Yu, H., Bai, S., Hao, Y., & Guan, Y. (2022). Fatty acids role in multiple sclerosis as “metabokines. Journal of Neuroinflammation, 19(1), 157. https://doi.org/10.1186/s12974-022-02502-1
Zahoor, I., Rui, B., Khan, J., Datta, I., & Giri, S. (2021). An emerging potential of metabolomics in multiple sclerosis: a comprehensive overview. Cellular and Molecular Life Sciences (78 vol., pp. 3181–3203). Springer Science and Business. https://doi.org/10.1007/s00018-020-03733-2. Media Deutschland GmbH
Zahoor, I., Suhail, H., Datta, I., Ahmed, M. E., Poisson, L. M., Waters, J., Rashid, F., Bin, R., Singh, J., Cerghet, M., Kumar, A., Hoda, M. N., Rattan, R., Mangalam, A. K., & Giri, S. (2022). Blood-based untargeted metabolomics in relapsing-remitting multiple sclerosis revealed the testable therapeutic target. Proceedings of the National Academy of Sciences of the United States of America, 119(25), e2123265119. https://doi.org/10.1073/pnas.2123265119
Zhang, A., Sun, H., Yan, G., Wang, P., & Wang, X. (2015). Review Article Metabolomics for Biomarker Discovery: Moving to the Clinic. https://doi.org/10.1155/2015/354671
Acknowledgements
We acknowledge funding from the National Institutes of Health/NIAID 1RO1AI137075 (A.K.M), Veteran Affairs Merit Award 1I01CX002212 (A.K.M), University of Iowa Environmental Health Sciences Research Center, NIEHS/NIH P30 ES005605 (A.K.M), Gift from P. Heppelmann and M. Wacek to A.K.M and Carver Trust Pilot Grant (A.K.M). R.L.S. was supported by the Informatics Fellowship from the University of Iowa. S.N.J was supported by an institutional training grant (T32AI007485 to G. Bishop) and a diversity supplement award to A.K.M on parent 1RO1AI137075. We also thank Leeann Aguilar Meza and Stephanie Peterson (University of Iowa) for their editorial assistance on this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualizations, Nicole Cady, Samantha N. Jensen, and Ashutosh K. Mangalam; methodology, Rachel L. Shrode., Nicole Cady, Samantha N. Jensen, Nick Borcherding, and Ashutosh K. Mangalam; validation, Rachel L. Shrode, Nick Borcherding, and Ashutosh K. Mangalam; formal analysis, investigation, resources, and data curation, Rachel L. Shrode; writing – original draft preparations Rachel L. Shrode; writing – review and editing, Rachel L. Shrode, Nicole Cady, Samantha N. Jensen, Nick Borcherding, and Ashutosh K. Mangalam; supervision Nick Borcherding, and Ashutosh K. Mangalam, funding acquisitions Ashutosh K. Mangalam. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
A.K.M. is one of the inventors of a technology claiming the use of Prevotella histicola to treat autoimmune diseases. A.K.M. received royalties from Mayo Clinic (paid by Evelo Biosciences). However, no funds or products from the patent were used in the present study. All other authors declare no commercial or financial relationships that could be a potential conflict of interest.
Institutional Review Board Statement
All procedures were done according to the Institutional Animal Care and Use Committee (IACUC) guidelines at the University of Iowa.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shrode, R.L., Cady, N., Jensen, S.N. et al. Isoflavone consumption reduces inflammation through modulation of phenylalanine and lipid metabolism. Metabolomics 18, 84 (2022). https://doi.org/10.1007/s11306-022-01944-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-022-01944-1