Abstract
Introduction
Muscle invasive bladder cancer (MIBC) is an advanced stage of bladder cancer which poses a severe threat to life. Cancer development is usually accompanied by remarkable alterations in cell metabolism, and hence deep insights into MIBC at the metabolomic level can facilitate the understanding of the biochemical mechanisms involved in the cancer development and progression.
Methods
In this proof-of-concept study, the optimal cutting temperature (OCT)-embedded MIBC samples were first washed with pure water to remove the polymer compounds which could cause severe signal suppression during mass spectrometry. Further, the tissue sections were analyzed by infrared matrix-assisted laser desorption electrospray ionization mass spectrometry imaging (IR-MALDESI MSI), providing an overview on the spatially resolved metabolomic profiles.
Results
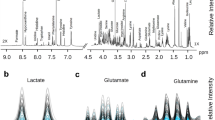
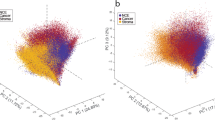
The MSI data enabled the discrimination between not only the cancerous and normal tissues, but also the subregions within a tissue section associated with different disease states. Using t-Distributed Stochastic Neighbor Embedding (t-SNE), the hyperdimensional MSI data was mapped into a two-dimensional space to visualize the spectral similarity, providing evidence that metabolomic alterations might have occurred outside the histopathological tumor border. Least absolute shrinkage and selection operator (LASSO) was further employed to classify sample pathology in a pixel-wise manner, yielding excellent prediction sensitivity and specificity up to 96% based on the statistically characteristic spectral features.
Conclusion
The results demonstrate great promise of IR-MALDESI MSI to identify molecular changes derived from cancer and unveil tumor heterogeneity, which can potentially promote the discovery of clinically relevant biomarkers and allow for applications in precision medicine.





Similar content being viewed by others
Data availability
The data sets are available at https://metaspace2020.eu/project/tu-2021.
Abbreviations
- PC:
-
Phosphatidylcholine
- PE:
-
Phosphatidylethanolamine
- PI:
-
Phosphatidylinositol
- PS:
-
Phosphatidylserine
- PG:
-
Phosphatidylglycerol
- FA:
-
Fatty acid
- MG:
-
Monoglyceride
- DG:
-
Diglyceride
- TG:
-
Triglyceride
- CE:
-
Cholesteryl ester
References
Abdelmoula, W. M., Balluff, B., Englert, S., Dijkstra, J., Reinders, M. J. T., Walch, A., et al. (2016). Data-driven identification of prognostic tumor subpopulations using spatially mapped t-SNE of Mass spectrometry imaging data. Proceedings of the National Academy of Sciences of the United States of America, 113(43), 12244–12249. https://doi.org/10.1073/pnas.1510227113
Baenke, F., Peck, B., Miess, H., & Schulze, A. (2013). Hooked on fat: The role of lipid synthesis in cancer metabolism and tumour development. DMM Disease Models and Mechanisms, 6(6), 1353–1363. https://doi.org/10.1242/dmm.011338
Bagley, M. C., Ekelöf, M., & Muddiman, D. C. (2020). Determination of optimal electrospray parameters for lipidomics in infrared-matrix-assisted laser desorption electrospray ionization mass spectrometry imaging. Journal of the American Society for Mass Spectrometry, 31(2), 319–325. https://doi.org/10.1021/jasms.9b00063
Banerjee, S., Zare, R. N., Tibshirani, R. J., Kunder, C. A., Nolley, R., Fan, R., et al. (2017). Diagnosis of prostate cancer by desorption electrospray ionization mass spectrometric imaging of small metabolites and lipids. Proceedings of the National Academy of Sciences, 114(13), 3334–3339. https://doi.org/10.1073/pnas.1700677114
Bogdanov, M., & Dowhan, W. (1998). Phospholipid-assisted protein folding: Phosphatidylethanolamine is required at a late step of the conformational maturation of the polytopic membrane protein lactose permease. EMBO Journal, 17(18), 5255–5264. https://doi.org/10.1093/emboj/17.18.5255
Bokhart, M. T., Nazari, M., Garrard, K. P., & Muddiman, D. C. (2018). MSiReader v1.0: Evolving open-source mass spectrometry imaging software for targeted and untargeted analyses. Journal of the American Society for Mass Spectrometry, 29(1), 8–16. https://doi.org/10.1007/s13361-017-1809-6
Calligaris, D., Caragacianu, D., Liu, X., Norton, I., Thompson, C. J., Richardson, A. L., et al. (2014). Application of desorption electrospray ionization mass spectrometry imaging in breast cancer margin analysis. Proceedings of the National Academy of Sciences of the United States of America, 111(42), 15184–15189. https://doi.org/10.1073/pnas.1408129111
Cordeiro, F. B., Jarmusch, A. K., León, M., Ferreira, C. R., Pirro, V., Eberlin, L. S., et al. (2020). Mammalian ovarian lipid distributions by desorption electrospray ionization–mass spectrometry (DESI-MS) imaging. Analytical and Bioanalytical Chemistry, 412, 1251–1262. https://doi.org/10.1007/s00216-019-02352-6
Cui, L., Lu, H., & Lee, Y. H. (2018). Challenges and emergent solutions for LC-MS/MS based untargeted metabolomics in diseases. Mass Spectrometry Reviews, 37(6), 772–792. https://doi.org/10.1002/mas.21562
Dixon, R. B., & Muddiman, D. C. (2010). Study of the ionization mechanism in hybrid laser based desorption techniques. The Analyst, 135(5), 880–882. https://doi.org/10.1039/b926422a
Dória, M. L., McKenzie, J. S., Mroz, A., Phelps, D. L., Speller, A., Rosini, F., et al. (2016). Epithelial ovarian carcinoma diagnosis by desorption electrospray ionization mass spectrometry imaging. Scientific Reports, 6(1), 1–11. https://doi.org/10.1038/srep39219
Eberlin, L. S., Tibshirani, R. J., Zhang, J., Longacre, T. A., Berry, G. J., Bingham, D. B., et al. (2014). Molecular assessment of surgical-resection margins of gastric cancer by mass-spectrometric imaging. Proceedings of the National Academy of Sciences of the United States of America, 111(7), 2436–2441. https://doi.org/10.1073/pnas.1400274111
Fonville, J. M., Carter, C. L., Pizarro, L., Steven, R. T., Palmer, A. D., Griffiths, R. L., et al. (2013). Hyperspectral visualization of mass spectrometry imaging data. Analytical Chemistry, 85(3), 1415–1423. https://doi.org/10.1021/ac302330a
Goto, T., Terada, N., Inoue, T., Nakayama, K., Okada, Y., Yoshikawa, T., et al. (2014). The expression profile of phosphatidylinositol in high spatial resolution imaging mass spectrometry as a potential biomarker for prostate cancer. PLoS ONE, 9(2), e90242. https://doi.org/10.1371/journal.pone.0090242
Guijas, C., Montenegro-Burke, J. R., Domingo-Almenara, X., Palermo, A., Warth, B., Hermann, G., et al. (2018). METLIN: A technology platform for identifying knowns and unknowns. Analytical Chemistry, 90(5), 3156–3164. https://doi.org/10.1021/acs.analchem.7b04424
Heisterkamp, N., Groffen, J., Warburton, D., & Sneddon, T. P. (2008). The human gamma-glutamyltransferase gene family. Human Genetics, 123(4), 321–332. https://doi.org/10.1007/s00439-008-0487-7
Humphrey, P. A., Moch, H., Cubilla, A. L., Ulbright, T. M., & Reuter, V. E. (2016). The 2016 WHO classification of tumours of the urinary system and male genital organs—Part B: Prostate and bladder tumours. European Urology, 70(1), 106–119. https://doi.org/10.1016/j.eururo.2016.02.028
Jarmusch, A. K., Pirro, V., Baird, Z., Hattab, E. M., Cohen-Gadol, A. A., & Cooks, R. G. (2016). Lipid and metabolite profiles of human brain tumors by desorption electrospray ionization-MS. Proceedings of the National Academy of Sciences, 113(6), 1486–1491. https://doi.org/10.1073/pnas.1523306113
Khodjaniyazova, S., Hanne, N. J., Cole, J. H., & Muddiman, D. C. (2019). Mass spectrometry imaging (MSI) of fresh bones using infrared matrix-assisted laser desorption electrospray ionization (IR-MALDESI). Analytical Methods, 11(46), 5929–5938. https://doi.org/10.1039/c9ay01886g
Koundouros, N., & Poulogiannis, G. (2020). Reprogramming of fatty acid metabolism in cancer. British Journal of Cancer, 122(1), 4–22. https://doi.org/10.1038/s41416-019-0650-z
Krauß, M., & Haucke, V. (2007). Phosphoinositide-metabolizing enzymes at the interface between membrane traffic and cell signalling. EMBO Reports, 8(3), 241–246. https://doi.org/10.1038/sj.embor.7400919
Lin, G., Keshari, K. R., & Park, J. M. (2017). Cancer metabolism and tumor heterogeneity: Imaging perspectives using MR imaging and spectroscopy. Contrast Media and Molecular Imaging. https://doi.org/10.1155/2017/6053879
Ma, J., & Black, P. C. (2021). Current perioperative therapy for muscle invasive bladder cancer. Hematology/oncology Clinics of North America, 35(3), 495–511. https://doi.org/10.1016/j.hoc.2021.02.002
Mao, X., He, J., Li, T., Lu, Z., Sun, J., Meng, Y., et al. (2016). Application of imaging mass spectrometry for the molecular diagnosis of human breast tumors. Scientific Reports, 6, 1–12. https://doi.org/10.1038/srep21043
Margulis, K., Chiou, A. S., Aasi, S. Z., Tibshirani, R. J., Tang, J. Y., & Zare, R. N. (2018). Distinguishing malignant from benign microscopic skin lesions using desorption electrospray ionization mass spectrometry imaging. Proceedings of the National Academy of Sciences of the United States of America, 115(25), 6347–6352. https://doi.org/10.1073/pnas.1803733115
Mirnezami, R., Spagou, K., Vorkas, P. A., Lewis, M. R., Kinross, J., Want, E., et al. (2014). Chemical mapping of the colorectal cancer microenvironment via MALDI imaging mass spectrometry (MALDI-MSI) reveals novel cancer-associated field effects. Molecular Oncology, 8(1), 39–49. https://doi.org/10.1016/j.molonc.2013.08.010
Momchilova, A., & Markovska, T. (1999). Phosphatidylethanolamine and phosphatidylcholine are sources of diacylglycerol in ras-transformed NIH 3T3 fibroblasts. International Journal of Biochemistry and Cell Biology, 31(2), 311–318. https://doi.org/10.1016/S1357-2725(98)00111-3
Morse, N., Jamaspishvili, T., Simon, D., Patel, P. G., Ren, K. Y. M., Wang, J., et al. (2019). Reliable identification of prostate cancer using mass spectrometry metabolomic imaging in needle core biopsies. Laboratory Investigation, 99(10), 1561–1571. https://doi.org/10.1038/s41374-019-0265-2
Muthukrishnan, R., & Rohini, R. (2016). LASSO: A feature selection technique in predictive modeling for machine learning. IEEE International Conference on Advances in Computer Applications (ICACA), 2016, 18–20. https://doi.org/10.1109/ICACA.2016.7887916
Nazari, M., & Muddiman, D. C. (2016). Polarity switching mass spectrometry imaging of healthy and cancerous hen ovarian tissue sections by infrared matrix-assisted laser desorption electrospray ionization (IR-MALDESI). The Analyst, 141(2), 595–605. https://doi.org/10.1039/c5an01513h
Ogawa, K., Shimizu, Y., Uketa, S., Utsunomiya, N., & Kanamaru, S. (2020). Prognosis of patients with muscle invasive bladder cancer who are intolerable to receive any anti-cancer treatment. Cancer Treatment and Research Communications, 24, 100195. https://doi.org/10.1016/j.ctarc.2020.100195
Oppenheimer, S. R., Mi, D., Sanders, M. E., & Caprioli, R. M. (2010). A molecular analysis of tumor margins by MALDI mass spectrometry in renal carcinoma. Journal of Proteome Research, 9(5), 2182–2190. https://doi.org/10.1021/pr900936z
Ouyang, Y., Liu, J., Nie, B., Dong, N., Chen, X., Chen, L., & Wei, Y. (2017). Differential diagnosis of human lung tumors using surface desorption atmospheric pressure chemical ionization imaging mass spectrometry. RSC Advances, 7(88), 56044–56053. https://doi.org/10.1039/c7ra11839b
Pace, C. L., Horman, B., Patisaul, H., & Muddiman, D. C. (2020). Analysis of neurotransmitters in rat placenta exposed to flame retardants using IR-MALDESI mass spectrometry imaging. Analytical and Bioanalytical Chemistry, 412(15), 3745–3752. https://doi.org/10.1007/s00216-020-02626-4
Palmer, A., Phapale, P., Chernyavsky, I., Lavigne, R., Fay, D., Tarasov, A., et al. (2017). FDR-controlled metabolite annotation for high-resolution imaging mass spectrometry. Nature Methods, 14(1), 57–60. https://doi.org/10.1038/nmeth.4072
Pirro, V., Alfaro, C. M., Jarmusch, A. K., Hattab, E. M., Cohen-Gadol, A. A., & Cooks, R. G. (2017). Intraoperative assessment of tumor margins during glioma resection by desorption electrospray ionization-mass spectrometry. Proceedings of the National Academy of Sciences, 114(26), 6700–6705. https://doi.org/10.1073/pnas.1706459114
Porcari, A. M., Zhang, J., Garza, K. Y., Rodrigues-Peres, R. M., Lin, J. Q., Young, J. H., et al. (2018). Multicenter study using desorption-electrospray-ionization-mass-spectrometry imaging for breast-cancer diagnosis. Analytical Chemistry, 90(19), 11324–11332. https://doi.org/10.1021/acs.analchem.8b01961
Robichaud, G., Barry, J. A., & Muddiman, D. C. (2014). IR-MALDESI mass spectrometry imaging of biological tissue sections using ice as a matrix. Journal of the American Society for Mass Spectrometry, 25(3), 319–328. https://doi.org/10.1007/s13361-013-0787-6
Robichaud, G., Garrard, K. P., Barry, J. A., & Muddiman, D. C. (2013). MSiReader: An open-source interface to view and analyze high resolving power MS imaging files on matlab platform. Journal of the American Society for Mass Spectrometry, 24(5), 718–721. https://doi.org/10.1007/s13361-013-0607-z
Röhrig, F., & Schulze, A. (2016). The multifaceted roles of fatty acid synthesis in cancer. Nature Reviews Cancer, 16(11), 732–749. https://doi.org/10.1038/nrc.2016.89
Rosen, E. P., Bokhart, M. T., Ghashghaei, H. T., & Muddiman, D. C. (2015). Influence of desorption conditions on analyte sensitivity and internal energy in discrete tissue or whole body imaging by IR-MALDESI. Journal of the American Society for Mass Spectrometry, 26(6), 899–910. https://doi.org/10.1007/s13361-015-1114-1
Sampson, J. S., Hawkridge, A. M., & Muddiman, D. C. (2008). Development and characterization of an ionization technique for analysis of biological macromolecules: Liquid matrix-assisted laser desorption electrospray ionization. Analytical Chemistry, 80(17), 6773–6778. https://doi.org/10.1021/ac8001935
Snijders, M. L. H., Zajec, M., Walter, L. A. J., de Louw, R. M. A. A., Oomen, M. H. A., Arshad, S., et al. (2019). Cryo-gel embedding compound for renal biopsy biobanking. Scientific Reports, 9(1), 1–10. https://doi.org/10.1038/s41598-019-51962-8
Struck-Lewicka, W., Kordalewska, M., Bujak, R., Yumba Mpanga, A., Markuszewski, M., Jacyna, J., et al. (2015). Urine metabolic fingerprinting using LC-MS and GC-MS reveals metabolite changes in prostate cancer: A pilot study. Journal of Pharmaceutical and Biomedical Analysis, 111, 351–361. https://doi.org/10.1016/j.jpba.2014.12.026
Szlasa, W., Zendran, I., Zalesińska, A., Tarek, M., & Kulbacka, J. (2020). Lipid composition of the cancer cell membrane. Journal of Bioenergetics and Biomembranes, 52(5), 321–342. https://doi.org/10.1007/s10863-020-09846-4
Tu, A., & Muddiman, D. C. (2019a). Internal energy deposition in infrared matrix-assisted laser desorption electrospray ionization with and without the use of ice as a matrix. Journal of the American Society for Mass Spectrometry, 30(11), 2380–2391. https://doi.org/10.1007/s13361-019-02323-2
Tu, A., & Muddiman, D. C. (2019b). Systematic evaluation of repeatability of IR-MALDESI-MS and normalization strategies for correcting the analytical variation and improving image quality. Analytical and Bioanalytical Chemistry, 411(22), 5729–5743. https://doi.org/10.1007/s00216-019-01953-5
Urayama, S., Zou, W., Brooks, K., & Tolstikov, V. (2010). Comprehensive mass spectrometry based metabolic profiling of blood plasma reveals potent discriminatory classifiers of pancreatic cancer. Rapid Communications in Mass Spectrometry, 24, 613–620. https://doi.org/10.1002/rcm.4420
Vrana, M., Goodling, A., Afkarian, M., & Prasad, B. (2016). An optimized method for protein extraction from OCT-embedded human kidney tissue for protein quantification by LC-MS/MS proteomics. Drug Metabolism and Disposition, 44(10), 1692–1696. https://doi.org/10.1124/dmd.116.071522
Xi, Y., Tu, A., & Muddiman, D. C. (2020). Lipidomic profiling of single mammalian cells by infrared matrix-assisted laser desorption electrospray ionization (IR-MALDESI). Analytical and Bioanalytical Chemistry, 412(29), 8211–8222. https://doi.org/10.1007/s00216-020-02961-6
Acknowledgements
We acknowledge the funding support from National Institutes of Health (NIH) Grants R01-CA193437R01 (to NS), the Wake Forest Baptist Comprehensive Cancer Center Tumor Tissue and Pathology Shared Resource (TTPSR), supported by the National Cancer Institute’s Cancer Center Support Grant Award Number P30CA012197, and the NIH Grant R01GM087964 (to DCM). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute. The MSI analysis was performed in the Molecular Education, Technology and Research Innovation Center (METRIC) at NC State University.
Author information
Authors and Affiliations
Contributions
AT, NS and DCM designed the experiment. AT collected and analyzed the mass spectrometry imaging data. NS provided the human samples and conducted histopathological evaluation. NS and DCM assisted in interpretation of the results. AT, NS and DCM wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tu, A., Said, N. & Muddiman, D.C. Spatially resolved metabolomic characterization of muscle invasive bladder cancer by mass spectrometry imaging. Metabolomics 17, 70 (2021). https://doi.org/10.1007/s11306-021-01819-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-021-01819-x