Abstract
Introduction
The Deficiens Homologue 9-iaaM (DefH9-iaaM) gene is an ovule-specific auxin-synthesizing gene which is expressed specifically in placenta/ovules and promotes auxin-synthesis. It was introduced into the genome of two grape cultivars Thompson Seedless and Silcora and both transgenic cultivars had an increased number of berries per bunch.
Objectives
This study investigates the down-stream metabolic changes of Silcora and Thompson seedless grape cultivars when genetically modified through the insertion of the DefH9-iaaM gene into their genome.
Methods
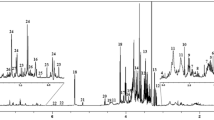
The effects of the genetic modification upon the grape metabolome were evaluated through 1H-NMR and exploratory data analysis. Chemometric tools such as Interval Partial Least Squares regression and metabolite heatmaps were employed for scrutinizing the changes in the transgenic metabolome as compared to the wild type one.
Results
The results show that the pleiotropic effect on the grape metabolome as a function of the gene modifications is relatively low, although the insertion of the transgene caused a decrement in malic acid and proline and an increment in p-coumaric acid content. In addition, the concentration of malic acid was successfully correlated with the number of inserted copies of transgene in the Silcora cultivar, proving that the increased production of berries, promoted by the inserted gene, is achieved at the expense of a decrement in malic acid concentration.
Conclusion
NMR together with chemometrics is able to identify specific metabolites that were up- or down regulated in the genetically engineered plants allowing highlighting alterations in the down-stream metabolic pathways due to the up-stream genetic modifications.





Similar content being viewed by others
References
Adamski, J., & Suhre, K. (2013). Metabolomics platforms for genome wide association studies—linking the genome to the metabolome. Current Opinion in Biotechnology, 24(1), 39–47. doi:10.1016/j.copbio.2012.10.003.
Ali, K., Maltese, F., Choi, Y. H., & Verpoorte, R. (2010). Metabolic constituents of grapevine and grape-derived products. Phytochemistry Reviews, 9(3), 357–378. doi:10.1007/s11101-009-9158-0.
Ali, K., Maltese, F., Fortes, A. M., Pais, M. S., Choi, Y. H., & Verpoorte, R. (2011). Monitoring biochemical changes during grape berry development in Portuguese cultivars by NMR spectroscopy. Food Chemistry, 124(4), 1760–1769. doi:10.1016/j.foodchem.2010.08.015.
Anastasiadi, M., Zira, A., Magiatis, P., Haroutounian, S. A., Skaltsounis, A. L., & Mikros, E. (2009). 1H NMR-Based metabonomics for the classification of greek wines according to variety, region, and vintage. Comparison with HPLC Data. Journal of Agricultural and Food Chemistry, 57(23), 11067–11074. doi:10.1021/jf902137e.
Balmer, D., Flors, V., Glauser, G., & Mauch-Mani, B. (2014). Metabolomics of cereals under biotic stress: current knowledge and techniques. Induced plant responses to microbes and insects, 146, doi:10.3389/fpls.2013.00082.
Cheynier, V., Duenas-Paton, M., Salas, E., Maury, C., Souquet, J.-M., Sarni-Manchado, P., et al. (2006). Structure and properties of wine pigments and tannins. American Journal of Enology and Viticulture, 57(3), 298–305.
Conde, C., Silva, P., Fontes, N., Dias, A. C. P., Tavares, R. M., Sousa, M. J., et al. (2007). Biochemical changes throughout grape berry development and fruit and wine quality. Food, 1, 1–22.
Costantini, E., Landi, L., Silvestroni, O., Pandolfini, T., Spena, A., & Mezzetti, B. (2007). Auxin synthesis-encoding transgene enhances grape fecundity. Plant Physiology, 143(4), 1689–1694. doi:10.1104/pp.106.095232.
Craig, A., Cloarec, O., Holmes, E., Nicholson, J. K., & Lindon, J. C. (2006). Scaling and normalization effects in NMR spectroscopic metabonomic data sets. Analytical Chemistry, 78(7), 2262–2267. doi:10.1021/ac0519312.
Dai, Z. W., Léon, C., Feil, R., Lunn, J. E., Delrot, S., & Gomès, E. (2013). Metabolic profiling reveals coordinated switches in primary carbohydrate metabolism in grape berry (Vitis vinifera L.), a non-climacteric fleshy fruit. Journal of Experimental Botany, 64(5), 1345–1355. doi:10.1093/jxb/ers396.
Davis, C., & Robinson, S. (1996). Sugar accumulation in grape berries. Plant Physiology, 111, 275–283. doi:10.1104/pp.111.1.275.
EFSA. (2006). Guidance document of the Scientific Panel on Genetically Modified Organisms for the risk assessment of genetically modified plants and derived food and feed. EFSA Journal, 99, 1–100.
Fernández de Simón, B., Hernández, T., Estrella, I., & Gómez-Cordovés, C. (1992). Variation in phenol content in grapes during ripening: low-molecular-weight phenols. European Food Research and Technology, 194(4), 351–354.
Gallo, V., Mastrorilli, P., Cafagna, I., Nitti, G. I., Latronico, M., Longobardi, F., et al. (2014). Effects of agronomical practices on chemical composition of table grapes evaluated by NMR spectroscopy. Journal of Food Composition and Analysis, 35(1), 44–52. doi:10.1016/j.jfca.2014.04.004.
Garrido, J., & Borges, F. (2013). Wine and grape polyphenols—a chemical perspective. Food Research International, 54(2), 1844–1858. doi:10.1016/j.foodres.2013.08.002.
Gerós, H., Chaves, M., & Delrot, S. (2012). The biochemistry of the grape berry. Bentham Science Publishers.
Griffin, J. L. (2006). The Cinderella story of metabolic profiling: does metabolomics get to go to the functional genomics ball? Philosophical Transactions of the Royal Society B: Biological Sciences, 361(1465), 147–161. doi:10.1098/rstb.2005.1734.
Hale, C. R. (1962). Synthesis of organic acids in the fruit of the grape. Nature, 195(4844), 917–918. doi:10.1038/195917a0.
Hall, R., Beale, M., Fiehn, O., Hardy, N., Sumner, L., & Bino, R. (2002). Plant metabolomics the missing link in functional genomics strategies. The Plant Cell, 14(7), 1437–1440. doi:10.1105/tpc.140720.
Hotelling, H. (1933). Analysis of a complex of statistical variables into principal components. Journal of Educational Psychology, 24(7), 417–441. doi:10.1037/h0071325.
Inouye, M., Kettunen, J., Soininen, P., Silander, K., Ripatti, S., Kumpula, L. S., et al. (2010). Metabonomic, transcriptomic, and genomic variation of a population cohort. Molecular System Biology, 6(1), 1–10. doi:10.1038/msb.2010.93.
James, A. K., Gordon, J. T., John, R. P., Donald, R. H., David, H., Charles, R. H., et al. (2000). Development of seed polyphenols in berries from Vitis vinifera L. cv. Shiraz. Australian Journal of Grape and Wine Research, 6(3), 244–254. doi:10.1111/j.1755-0238.2000.tb00185.x.
Kliewer, W. M. (1966). Sugars and organic acids of Vitis vinifera. Plant Physiology, 41(6), 923–931. doi:10.1104/pp.41.6.923.
Laghi, L., Versari, A., Marcolini, E., & Parpinello, G. P. (2014). Metabonomic investigation by 1H-NMR to discriminate between red wines from organic and biodynamic grapes. Food and Nutrition Sciences, 5, 52–59. doi:10.4236/fns.2014.51007.
López-Rituerto, E., Savorani, F., Avenoza, A., Busto, J. H., Peregrina, J. M., & Engelsen, S. B. (2012). Investigations of La Rioja terroir for wine production using 1H NMR metabolomics. Journal of Agicultural and Food Chemistry, 60(13), 3452–3461. doi:10.1021/jf204361d.
Mezzetti, B., Pandolfini, T., Navacchi, O., & Landi, L. (2002). Genetic transformation of Vitis vinifera via organogenesis. BMC Biotechnology, 2(18), 1–10. doi:10.1186/1472-6750-2-18.
Mohanta, T. K. (2013). Plant metabolomics: missing link in next generation functional genomics era. Journal of Applied Biology and Biotechnology, 1, 001–010. doi:10.7324/JABB.2013.1301.
Mulas, G., Galaffu, M. G., Pretti, L., Nieddu, G., Mercenaro, L., Tonelli, R., et al. (2011). NMR analysis of seven selections of vermentino grape berry: metabolites composition and development. Journal of Agricultural and Food Chemistry, 59(3), 793–802. doi:10.1021/jf103285f.
Nørgaard, L., Saudland, A., Wagner, J., Nielsen, J. P., Munck, L., & Engelsen, S. B. (2000). Interval Partial Least-Squares Regression (iPLS): a comparative chemometric study with an example from near-infrared spectroscopy. Applied Spectroscopy, 54(3), 413–419.
Oliveira, J., de Freitas, V., Silva, A. M. S., & Mateus, N. (2007). Reaction between hydroxycinnamic acids and anthocyanin—pyruvic acid adducts yielding new portisins. Journal of Agricultural and Food Chemistry, 55(15), 6349–6356. doi:10.1021/jf070968f.
Pereira, G. E., Gaudillere, J.-P., Leeuwen, C. V., Hilbert, G., Maucourt, M., Deborde, C., et al. (2006). 1H NMR metabolite fingerprints of grape berry: comparison of vintage and soil effects in Bordeaux grapevine growing areas. Analytica Chimica Acta, 563(1–2), 346–352. doi:10.1016/j.aca.2005.11.007.
Picone, G., Laghi, L., Gardini, F., Lanciotti, R., Siroli, L., & Capozzi, F. (2013). Evaluation of the effect of carvacrol on the Escherichia coli 555 metabolome by using 1 H-NMR spectroscopy. Food Chemistry, 141(4), 4367–4374. doi:10.1016/j.foodchem.2013.07.004.
Picone, G., Mezzetti, B., Babini, E., Capocasa, F., Placucci, G., & Capozzi, F. (2011). Unsupervised principal component analysis of NMR metabolic profiles for the assessment of substantial equivalence of transgenic grapes (Vitis vinifera). Journal of Agricultural and Food Chemistry, 59(17), 9271–9279. doi:10.1021/jf2020717.
Romeyer, F. M., Macheix, J. J., Goiffon, J. J., Reminiac, C. C., & Sapis, J. C. (2002). Browning capacity of grapes. 3. Changes and importance of hydroxycinnamic acid-tartaric acid esters during development and maturation of the fruit. Journal of Agricultural and Food Chemistry, 31(2), 346–349. doi:10.1021/jf00116a040.
Rotino, G. L., Perri, E., Zottini, M., Sommer, H., & Spena, A. (1997). Genetic engineering of parhenocarpic plants. Nature Biotechnology, 15, 1320–1398. doi:10.1038/nbt1297-1398.
Saito, K., & Kasai, Z. (1968). Accumulation of tartaric acid in the ripening process of grapes. Plant Cell Physiology, 9(3), 529–537.
Savage, A. K., Tucker, G., van Duynhoven, J. P. M., Wulfert, F., & Daykin, C. A. (2009). Nutrimetabolomics: development of a bio-identification toolbox to determine the bioactive compounds in grape juice. Bioanalysis, 1(9), 1537–1549. doi:10.4155/bio.09.147.
Savorani, F., Picone, G., Badiani, A., Fagioli, P., Capozzi, F., & Engelsen, S. B. (2010a). Metabolic profiling and aquaculture differentiation of gilthead sea bream by 1H NMR metabonomics. Food Chemistry, 120(3), 907–914. doi:10.1016/j.foodchem.2009.10.071.
Savorani, F., Tomasi, G., & Engelsen, S. B. (2010b). iCOSHIFT: A versatile tool for the rapid alignment of 1D NMR spectra. Journal of Magnetic Resonance, 202(2), 190–202. doi:10.1016/j.jmr.2009.11.012.
Schlosser, J., Olsson, N., Weis, M., Reid, K., Peng, F., Lund, S., et al. (2008). Cellular expansion and gene expression in the developing grape (Vitis vinifera L.). Protoplasma, 232(3), 255–265. doi:10.1007/s00709-008-0280-9.
Scorza, R., Cordts, J., Gray, D., Gonsalves, D., Emershad, R., & Ramming, D. (1996). Producing transgenicthompson seedless’ grape (Vitis vinifera L.) plants. Journal of the American Society for Horticultural Science, 121(4), 616–619.
Simó, C., Ibáez, C., Valdés, A., Cifuentes, A., & García-Cañas, V. (2014). Metabolomics of genetically modified crops. International Journal of Molecular Sciences, 15(10), 18941–18966. doi:10.3390/ijms151018941.
Son, H.-S., Hwang, G.-S., Kim, K. M., Ahn, H.-J., Park, W.-M., Van Den Berg, F., et al. (2009). Metabolomic studies on geographical grapes and their wines using 1H NMR analysis coupled with multivariate statistics. Journal of Agricultural and Food Chemistry, 57(4), 1481–1490. doi:10.1021/jf803388w.
Stines, A. P., Grubb, J., Gockowiak, H., Henschke, P. A., Høj, P. B., & Heeswijck, R. (2000). Proline and arginine accumulation in developing berries of Vitis vinifera L. in Australian vineyards: influence of vine cultivar, berry maturity and tissue type. Australian Journal of Grape and Wine Research, 6(2), 150–158. doi:10.1111/j.1755-0238.2000.tb00174.x.
Zhao, Y. (2010). auxin biosynthesis and its role in plant development. Annual Review of Plant Biology, 61, 49–64. doi:10.1146/annurev-arplant-042809-112308.
Zhao, Y. (2014). Auxin biosynthesis. The Arabidopsis Book/American Society of Plant Biologists, 12, e0173. doi:10.1199/tab.0173.
Acknowledgments
The Danish Ministry of Food, Agriculture and Fisheries is acknowledged for sponsoring the project “Urinary biomarkers for eating patterns using NMR spectroscopy and Chemometrics” (3304-FVFP-060706-01) and the SBE would like to thank the Faculty of Science for generous support to the NMR nutri-metabolomics platform. The European Union is acknowledged for the support under the FP7 programme to the CHANCE project (KBBE-4-266331).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
No informed consent was required for this study since no human participant was involved.
Conflict of interest
Picone G., Savorani F., Trimigno A., Mezzetti B., Capozzi F. and Engelsen S.B. declare that they have no conflict of interest.
Additional information
G. Picone and F. Savorani contributed equally to the research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Picone, G., Savorani, F., Trimigno, A. et al. Metabolic changes of genetically engineered grapes (Vitis vinifera L.) studied by 1H-NMR, metabolite heatmaps and iPLS. Metabolomics 12, 150 (2016). https://doi.org/10.1007/s11306-016-1095-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-016-1095-5