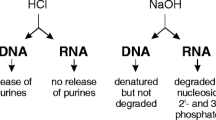
Abstract
Central to the domain of molecular biology resides the foundational process of DNA extraction and purification, a cornerstone underpinning a myriad of pivotal applications. In this research, we introduce a DNA extraction and purification technique leveraging polypropylene (PP) threads. The process commences with robust cell lysis achieved through the vigorous agitation of interwoven PP threads. The friction between the threads facilitates cell lysis especially those microbes having tough cell wall. For purification of DNA, thread-based isotachophoresis was employed which makes the whole process swift and cost-effective. Lysed cell-laden threads were submerged in a trailing electrolyte which separated DNA from other cellular contents. The process was performed with a tailored ITP device. An electric field directs DNA, cell debris, trailing electrolyte, and leading electrolyte toward the anode. Distinct ion migration resulted in DNA concentrating on the PP thread’s anode-proximal region. The SYBR green dye is used to visualize DNA as a prominent green zone under blue light. The purified DNA exhibits high purity levels of 1.82 ± 0.1 (A260/A280), making it suitable for various applications aiming at nucleic acid detection.







Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and its Supplementary Information. All images in this study were created by the authors and are original designs. There has been no use of images sourced from external locations, ensuring compliance with copyright restrictions.
References
Ahmad A, Ghasemi J (2007) New buffers to improve the quantitative real-time polymerase chain reaction. Biosci Biotechnol Biochem 71(8):1970–1978. https://doi.org/10.1271/bbb.70164
Ali N, Rampazzo R, de Costa CP, Krieger ADT (2017) Current nucleic acid extraction methods and their implications to point-of-care diagnostics. Biomed Res Int 2017:1–13. https://doi.org/10.1155/2017/9306564
Ayoib A, Hashim U, Gopinath SCB, Md Arshad MK (2017) DNA extraction on bio-chip: history and preeminence over conventional and solid-phase extraction methods. Appl Microbiol Biotechnol 101(22):8077–8088. https://doi.org/10.1007/s00253-017-8493-0
Bloom BS, Goldberg DJ (2007) Suture material in cosmetic cutaneous surgery. J Cosmet Laser Ther 9(1):41–45. https://doi.org/10.1080/14764170601140062
Brody JR, Kern SE (2004) History and principles of conductive media for standard DNA electrophoresis. Anal Biochem 333(1):1–13. https://doi.org/10.1016/j.ab.2004.05.054
Cabot JM, Breadmore MC, Paull B (2018) Thread based electrofluidic platform for direct metabolite analysis in complex samples. Anal Chim Acta 1000:283–292. https://doi.org/10.1016/j.aca.2017.10.029
Chen L, Ghiasvand A, Sanz Rodriguez E, Innis PC, Paull B (2022) Nanomaterial-assisted thread-based isotachophoresis with on-thread solute trapping. Analyst 147(9):1944–1951. https://doi.org/10.1039/d2an00287f
Chen L, Ghiasvand A, Paull B (2023) Applications of thread-based microfluidics: approaches and options for detection. TRAC Trends Anal Chem 161:117001. https://doi.org/10.1016/j.trac.2023.117001
Chernyshov SV, Tsvetkova DV, Mikoulinskaia GV (2022) A rapid and efficient technique for the isolation of Bacillus genomic DNA using a cocktail of peptidoglycan hydrolases of different type. World J Microbiol Biotechnol. https://doi.org/10.1007/s11274-022-03475-2
D’Alessandro B, Antúnez K, Piccini C, Zunino P (2007) DNA extraction and PCR detection of Paenibacillus larvae spores from naturally contaminated honey and bees using spore-decoating and freeze-thawing techniques. World J Microbiol Biotechnol 23(4):593–597. https://doi.org/10.1007/s11274-006-9261-y
Dairawan M, Shetty J P (2020) The evolution of DNA extraction methods. Am J Biomedical Sci Res 8(1):39–45. https://doi.org/10.34297/ajbsr.2020.08.001234
Demeke T, Jenkins GR (2010) Influence of DNA extraction methods, PCR inhibitors and quantification methods on real-time PCR assay of biotechnology-derived traits. Anal Bioanal Chem 396(6):1977–1990. https://doi.org/10.1007/s00216-009-3150-9
Dotti I, Bonin S (2011) Quantification of nucleic acids. Guidelines for Molecular Analysis in Archive tissues 75–79. Springer Berlin Heidelberg, Berlin, Heidelberg
Dragan AI, Pavlovic R, McGivney JB, Casas-Finet JR, Bishop ES, Strouse RJ, Schenerman MA, Geddes CD (2012) SYBR Green I: fluorescence properties and Interaction with DNA. J Fluoresc 22(4):1189–1199. https://doi.org/10.1007/s10895-012-1059-8
Eid C, Santiago JG (2018) Isotachophoresis applied to biomolecular reactions. Lab Chip 18(1):11–26. https://doi.org/10.1039/c7lc00852j
Farajikhah S, Cabot JM, Innis PC, Paull B, Wallace G (2019) Life-saving threads: advances in textile-based analytical devices. ACS Comb Sci 21(4):229–240. https://doi.org/10.1021/acscombsci.8b00126
Garg R, Prasad D (2023) Carbon dots and their interactions with recognition molecules for enhanced nucleic acid detection. Biochem Biophys Res Commun 680:93–107. https://doi.org/10.1016/j.bbrc.2023.09.033
Haque KA, Pfeiffer RM, Beerman MB, Struewing JP, Chanock SJ, Bergen AW (2003) Performance of high-throughput DNA quantification methods. BMC Biotechnol 3(1):1–10. https://doi.org/10.1186/1472-6750-3-20
Heymann F, von Trotha K-T, Preisinger C, Lynen-Jansen P, Roeth AA, Geiger M, Geisler LJ, Frank AK, Conze J, Luedde T, Trautwein C, Binnebösel M, Neumann UP, Tacke F (2019) Polypropylene mesh implantation for hernia repair causes myeloid cell–driven persistent inflammation. JCI Insight. https://doi.org/10.1172/jci.insight.123862
Hussing C, Kampmann ML, Mogensen HS, Børsting C, Morling N (2015) Comparison of techniques for quantification of next-generation sequencing libraries. For Sci Int Genet Supplement Ser 5:e276–e278. https://doi.org/10.1016/j.fsigss.2015.09.110
Ip SCY, Lin S, Lai K (2015) An evaluation of the performance of five extraction methods: Chelex® 100, QIAamp® DNA Blood Mini Kit, QIAamp® DNA Investigator Kit, QIAsymphony® DNA Investigator® Kit and DNA IQTM. Sci Justice 55:200–208
Kowalczyk A (2020) Trends and perspectives in DNA biosensors as diagnostic devices. Curr Opinion Electrochem 23:36–41. https://doi.org/10.1016/j.coelec.2020.03.003
Lucena-Aguilar G, Sánchez-López AM, Barberán-Aceituno C, Carrillo-Ávila JA, López-Guerrero JA, Aguilar-Quesada R (2016) DNA Source Selection for Downstream Applications Based on DNA Quality Indicators Analysis. Biopreservation and Biobanking 14:264–270. https://doi.org/10.1089/bio.2015.0064
Malá Z, Gebauer P (2023) Analytical isotachophoresis 1967–2022: from standard analytical technique to universal on-line concentration tool. TRAC Trends Anal Chem 158:116837. https://doi.org/10.1016/j.trac.2022.116837
Marshall LA, Han CM, Santiago JG (2011) Extraction of DNA from malaria-infected erythrocytes using isotachophoresis. Anal Chem 83(24):9715–9718. https://doi.org/10.1021/ac202567j
Mesapogu S, Jillepalli CM, Arora DK (2012) Microbial DNA extraction, purification, and quantitation. In analyzing microbes: manual of molecular biology techniques. Berlin Heidelberg: Springer Protocols Handbooks 1–16. https://doi.org/10.1007/978-3-642-34410-7_1
Nimbkar PH, Bhatt D (2022) A review on touch DNA collection, extraction, amplification, analysis and determination of phenotype. For Sci Int 336:111352. https://doi.org/10.1016/j.forsciint.2022.111352
Ning J, Liebich J, Kästner M, Zhou J, Schäffer A, Burauel P (2009) Different influences of DNA purity indices and quantity on PCR-based DGGE and functional gene microarray in soil microbial community study. Applied Microbiology and Biotechnology 82:983–993. https://doi.org/10.1007/s00253-009-1912-0
Oda Y, Yabuki M, Tonomura K, Fukunaga M (1997) A phylogenetic analysis of saccharomyces species by the sequence of 18S–28S rRNA spacer regions. Yeast 13(13):1243–1250.
Oztug M, Yalcinkaya B (2023) Development and validation of an HPLC-UV method for purity determination of DNA. Medicine Science | International Medical Journal 12:876. https://doi.org/10.5455/medscience.2023.06.088
Persad A, Marshall LA, Santiago JG (2009) Purification of nucleic acids from whole blood using isotachophoresis. Anal Chem 81(22):9507–9511. https://doi.org/10.1021/ac901965v
Prasad D, Shankaracharya, Vidyarthi AS (2011) Gold nanoparticles-based colorimetric assay for rapid detection of Salmonella species in food samples. World J Microbiol Biotechnol 27(9):2227–2230. https://doi.org/10.1007/s11274-011-0679-5
Ramachandran A, Santiago JG (2022) Isotachophoresis: theory and microfluidic applications. Chem Rev 122(15):12904–12976. https://doi.org/10.1021/acs.chemrev.1c00640
Rivas R, Velázquez E, Zurdo-Piñeiro JL, Mateos PF, Martı́nez Molina E (2004) Identification of microorganisms by PCR amplification and sequencing of a universal amplified ribosomal region present in both prokaryotes and eukaryotes. J Microbiol Methods 56(3):413–426. https://doi.org/10.1016/j.mimet.2003.11.007
Rogacs A, Marshall LA, Santiago JG (2014) Purification of nucleic acids using isotachophoresis. J Chromatogr A 1335:105–120. https://doi.org/10.1016/j.chroma.2013.12.027
Schrader C, Schielke A, Ellerbroek L, Johne R (2012) PCR inhibitors: occurrence, properties and removal. J Appl Microbiol 113(5):1014–1026. https://doi.org/10.1111/j.1365-2672.2012.05384.x
Sidstedt M, Rådström P, Hedman J (2020) PCR inhibition in qPCR, dPCR and MPS—mechanisms and solutions. Anal Bioanal Chem 412(9):2009–2023. https://doi.org/10.1007/s00216-020-02490-2
Wu L, Manchanda A, Gupta V, Paull B (2023) Graphene oxide-functionalized thread-based electrofluidic approach for DNA hybridization. ACS Omega 8(15):13569–13577. https://doi.org/10.1021/acsomega.2c06228
Acknowledgements
The authors thank the AICTE - IDEA (Idea Development, Evaluation & Application) Lab and the Central Instrumentation Facility, BIT Mesra, for the technical and instrumentation support.
Funding
D.P. has received grant from the parent Institute for this work. (Ref: BT/HOD/22–23/074). R.G. received fellowship (APO/PhD/IRS/2023–2024/61).
Author information
Authors and Affiliations
Contributions
Conceptualization: DP; Methodology: RG, AM, NKM, DP; Formal analysis and investigation: RG, DP, Writing—original draft preparation: RG, AM; Writing—review and editing: DP, NKM; Funding acquisition: DP; Resources: DP; Supervision: DP, NKM.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary material 2 (MP4 27588 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Garg, R., Maurya, A., Mani, N.K. et al. Thread-powered cell lysis and isotachophoresis: unlocking microbial DNA for diverse molecular applications. World J Microbiol Biotechnol 40, 97 (2024). https://doi.org/10.1007/s11274-024-03906-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-024-03906-2