Abstract
The aims of this work were to identify in vivo manifestations of antioxidant activity of Lactobacillus strains isolated from healthy human biotopes and to show the possibility of protective action of the selected strain on the model of oxidative stress induced by paraquat in the model of early Parkinson’s disease (PD) in mice. We studied the protective effects of 14 Lactobacillus strains belonging to five species on the lifespan of the soil nematode Caenorhabditis elegans experiencing oxidative stress induced by paraquat. The Lactobacillus strains used in this study were selected previously based on their ability to reduce oxidative stress in vitro. One of the strains that showed promising results on C. elegans was tested in a mouse model of PD in which C57/BL6 mice were injected regularly with paraquat. We assessed the state of their internal organs, the preservation of dopaminergic neurons in the substantia nigra as well as their motor coordination. The positive impact of Lactobacillus fermentum U-21 strain supplementation on paraquat treated animals was observed. L. fermentum U-21 strain reduced the toxicity of paraquat in C. elegans model: the lifespan of the soil nematode C. elegans was extended by 25%. L. fermentum U-21 protected the mice against anatomical and behavioral changes typical of PD: there were no changes in the coordination of movement and the preservation of dopaminergic neurons in the brain.
Graphic abstract
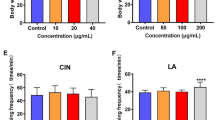
Life span of the nematode C. elegans pre-grown on a lawn of E. coli OP50 + Lactobacillus under oxidative stress conditions; the concentration of the oxidizing agent paraquat in the S medium was 50 mmol l−1.



Similar content being viewed by others

References
Achuthan AA, Duary RK, Madathil A, Panwar H, Kumar H, Batish VK, Grover S (2012) Antioxidative potential of lactobacilli isolated from the gut of Indian people. Mol Biol Rep 39:7887–7897. https://doi.org/10.1007/s11033-012-1633-9
Attia HN, Maklad YA (2018) Neuroprotective effects of coenzyme Q10 on paraquat-induced Parkinson's disease in experimental animals. Behav Pharmacol 29:79–86. https://doi.org/10.1097/FBP.0000000000000342
Baltazar MT, Dinis-Oliveira RJ, Bastos ML, Tsatsakis AM, Duarte JA, Carvalho F (2014) Pesticides exposure as etiological factors of Parkinson’s disease and other neurodegenerative diseases—a mechanistic approach. Toxicol Lett 230:85–103. https://doi.org/10.1016/j.toxlet.2014.01.039
Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, Lesin VM et al (2012) SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19:455–477. https://doi.org/10.1089/cmb.2012.0021
Bastías-Candia S, Zolezzi JM, Inestrosa NC (2019) Revisiting the paraquat-induced sporadic Parkinson's disease-like model. Mol Neurobiol 56:1044–1055. https://doi.org/10.1007/s12035-018-1148-z
Blanco-Ayala T, Andérica-Romero AC, Pedraza-Chaverri J (2014) New insights into antioxidant strategies against paraquat toxicity. Free Radic Res 48:623–640. https://doi.org/10.3109/10715762.2014.899694
Brooks AI, Chadwick CA, Gelbard HA, Cory-Slechta DA, Federoff HJ (1999) Paraquat elicited neurobehavioral syndrome caused by dopaminergic neuron loss. Brain Res 823:1–10. https://doi.org/10.1016/s0006-8993(98)01192-5
Byerly L, Cassada RC, Russell RL (1976) The life cycle of the nematode Caenorhabditis elegans. I. Wild-type growth and reproduction. Dev Biol 51:23–33. https://doi.org/10.1016/0012-1606(76)90119-6
DeMan JC, Rogosa M, Sharpe M (1960) A medium for the cultivation of lactobacilli. J Appl Microbiol 23:130–135
Dinis-Oliveira RJ, Duarte JA, Sánchez-Navarro A, Remião F, Bastos ML, Carvalho F (2008) Paraquat poisonings: mechanisms of lung toxicity, clinical features, and treatment. Crit Rev Toxicol 38:13–71. https://doi.org/10.1080/10408440701669959
Dou T, Yan M, Wang X, Lu W, Zhao L, Lou D, Wu C et al (2016) Nrf2/ARE pathway involved in oxidative stress induced by paraquat in human neural progenitor cells. Oxid Med Cell Longev. https://doi.org/10.1155/2016/8923860
Goyal N, Rishi P, Shukla G (2013) Lactobacillus rhamnosus GG antagonizes Giardia intestinalis induced oxidative stress and intestinal disaccharidases: an experimental study. World J Microbiol Biotechnol 29:1049–1057. https://doi.org/10.1007/s11274-013-1268-6
Grompone G, Martorell P, Llopis S, González N, Genovés S (2012) Anti-inflammatory Lactobacillus rhamnosus CNCM I-3690 strain protects against oxidative stress and increases lifespan in Caenorhabditis elegans. PLoS ONE 7:e52493. https://doi.org/10.1371/journal.pone.0052493
Hunt PR (2017) The C. elegans model in toxicity testing. J Appl Toxicol 37:50–59. https://doi.org/10.1002/jat.3357
Jones RM, Desai C, Darby TM, Luo L, Wolfarth AA, Scharer CD, Ardita CS et al (2015) Lactobacilli modulate epithelial cytoprotection through the Nrf2 pathway. Cell Rep 12:1217–1225. https://doi.org/10.1016/j.celrep.2015.07.042
Khan F, Jain S, Oloketuyi SF (2018) Bacteria and bacterial products: foe and friends to Caenorhabditis elegans. Microbiol Res 215:102–113. https://doi.org/10.1016/j.micres.2018.06.012
Kim B, Wang YC, Hespen CW, Espinosa J, Salje J, Rangan KJ, Oren DA et al (2019) Enterococcus faecium secreted antigen A generates muropeptides to enhance host immunity and limit bacterial pathogenesis. ELife 8:e45343. https://doi.org/10.7554/eLife.45343
Kleniewska P, Hoffmann A, Pniewska E, Pawliczak R (2016) The influence of probiotic Lactobacillus casei in combination with prebiotic inulin on the antioxidant capacity of human plasma. Oxid Med Cell Longev 2016:1340903. https://doi.org/10.1155/2016/1340903
Kobatake E, Nakagawa H, Seki T, Miyazaki T (2017) Protective effects and functional mechanisms of Lactobacillus gasseri SBT2055 against oxidative stress. PLoS ONE 12:0177106. https://doi.org/10.1371/journal.pone.0177106
Kovalzon VM, Moiseenko LS, Ambaryan AV, Kurtenbach S, Shestopalov VI, Panchin YV (2017) Sleep-wakefulness cycle and behavior in pannexin1 knockout mice. Behav Brain Res 318:24–27. https://doi.org/10.1016/j.bbr.2016.10.015
Kruskal WH, Wallis WA (1952) Use of ranks in one-criterion variance analysis. JASA 47:583–621
Kullisaar T, Songisepp E, Zilmer M (2012) Probiotics and oxidative stress. In Oxidative stress – environmental induction and dietary antioxidants. IntechOpen, Rijeka. https://www.intechopen.com/books/oxidative-stress-environmental-induction-and-dietary-antioxidants/probiotics-and-oxidative-stress
Lebeer S, Bron PA, Marco ML, Van Pijkeren JP, O’Connell Motherway M, Hill C, Pot B et al (2018) Identification of probiotic effector molecules: present state and future perspectives. Curr Opin Biotechnol 49:217–223. https://doi.org/10.1016/j.copbio.2017.10.007
Li S, Zhao Y, Zhang L, Zhang X, Huang L, Li D, Niu C et al (2012) Antioxidant activity of Lactobacillus plantarum strains isolated from traditional Chinese fermented foods. Food Chem 135:1914–1919. https://doi.org/10.1016/j.foodchem.2012.06.048
Ma L, Zhao Y, Chen Y, Cheng B, Peng A, Huang K (2018) Caenorhabditis elegans as a model system for target identification and drug screening against neurodegenerative diseases. Eur J Pharmacol 819:169–180. https://doi.org/10.1016/j.ejphar.2017.11.051
Makarenko MS, Chistyakov VA, Usatov AV, Mazanko MS, Prazdnova EV, Bren AB, Gorlov IF et al (2019) The impact of Bacillus subtilis KATMIRA1933 supplementation on telomere length and mitochondrial DNA damage of laying hens. Probiotics Antimicrob Proteins 11:588–593. https://doi.org/10.1007/s12602-018-9440-9
Marsova MV, Abilev SK, Poluektova EU, Danilenko VN (2018) A bioluminescent test system reveals valuable antioxidant properties of Lactobacillus strains from human microbiota. World J Microbiol Biotechnol 34:27. https://doi.org/10.1007/s11274-018-2410-2
Matsuura K, Kabuto H, Makino H, Ogawa N (1997) Pole test is a useful method for evaluating the mouse movement disorder caused by striatal dopamine depletion. J Neurosci Methods 73:45–48. https://doi.org/10.1016/s0165-0270(96)02211-x
Mishra V, Shah C, Mokashe N, Chavan R, Yadav H, Prajapati J (2015) Probiotics as potential antioxidants: a systematic review. J Agric Food Chem 63:3615–3626. https://doi.org/10.1021/jf506326t
Nakagawa H, Shiozaki T, Kobatake E, Hosoya T, Moriya T, Sakai F, Taru H et al (2016) Effects and mechanisms of prolongevity induced by Lactobacillus gasseri SBT2055 in Caenorhabditis elegans. Aging Cell 15:227–236. https://doi.org/10.1111/acel.12431
Niedzielska E, Smaga I, Gawlik M, Moniczewski A, Stankowicz P, Pera J, Filip M (2016) Oxidative stress in neurodegenerative diseases. Mol Neurobiol 53:4094–4125. https://doi.org/10.1007/s12035-015-9337-5
Park MR, Ryu S, Maburutse BE, Oh NS, Kim SH, Oh S, Jeong S-Y et al (2018) Probiotic Lactobacillus fermentum strain JDFM216 stimulates the longevity and immune response of Caenorhabditis elegans through a nuclear hormone receptor. Sci Rep 8:7441. https://doi.org/10.1038/s41598-018-25333-8
Peran L, Camuesco D, Comalada M, Nieto A, Concha A, Adrio JL, Olivares M et al (2006) Lactobacillus fermentum, a probiotic capable to release glutathione, prevents colonic inflammation in the TNBS model of rat colitis. Int J Colorectal Dis 21:737–746. https://doi.org/10.1007/s00384-005-0773-y
Rangan KJ, Pedicord VA, Wang YC, Kim B, Lu Y, Shaham S, Mucida D et al (2016) A secreted bacterial peptidoglycan hydrolase enhances tolerance to enteric pathogens. Science 353:1434–1437. https://doi.org/10.1126/science.aaf3552
Rani V, Deep G, Singh RK, Palle K, Yadav UC (2016) Oxidative stress and metabolic disorders: pathogenesis and therapeutic strategies. Life Sci 148:183–193. https://doi.org/10.1016/j.lfs.2016.02.002
Reczek CR, Birsoy K, Kong H, Martínez-Reyes I, Wang T, Gao P, Sabatini DM et al (2017) A CRISPR screen identifies a pathway required for paraquat-induced cell death. Nat Chem Biol 13:1274–1279. https://doi.org/10.1038/nchembio.2499
Schifano E, Zinno P, Guantario B, Roselli M, Marcoccia S, Devirgiliis C, Uccelletti D (2019) The foodborne strain Lactobacillus fermentum MBC2 triggers pept-1-dependent pro-longevity effects in Caenorhabditis elegans. Microorganisms 7:45. https://doi.org/10.3390/microorganisms7020045
Serata M, Iino T, Yasuda E, Sako T (2012) Roles of thioredoxin and thioredoxin reductase in the resistance to oxidative stress in Lactobacillus casei. Microbiology 158:953–962. https://doi.org/10.1099/mic.0.053942-0
Serrano LM, Molenaar D, Wels M, Teusink B, Bron PA, de Vos WM, Smid EJ (2007) Thioredoxin reductase is a key factor in the oxidative stress response of Lactobacillus plantarum WCFS1. Microb Cell Fact 6:29. https://doi.org/10.1186/1475-2859-6-29
Sies H, Berndt C, Jones DP (2017) Oxidative stress. Annu Rev Biochem 86:715–748. https://doi.org/10.1146/annurev-biochem-061516-045037
Smeyne RJ, Breckenridge CB, Beck M, Jiao Y, Butt MT, Wolf JC, Zadory D et al (2016) Assessment of the effects of MPTP and paraquat on dopaminergic neurons and microglia in the substantia nigra pars compacta of C57BL/6 Mice. PLoS ONE 11:e0164094. https://doi.org/10.1371/journal.pone.0164094
Solis GM, Petrascheck M (2011) Measuring Caenorhabditis elegans life span in 96 well microtiter plates. J Vis Exp 49:2496. https://doi.org/10.3791/2496
Songisepp E, Kals J, Kullisaar T, Mändar R, Hütt P, Zilmer M, Mikelsaar M (2005) Evaluation of the functional efficacy of an antioxidative probiotic in healthy volunteers. Nutr J 4:22. https://doi.org/10.1186/1475-2891-4-22
Wang AN, Yi XW, Yu HF, Dong B, Qiao SY (2009) Free radical scavenging activity of Lactobacillus fermentum in vitro and its antioxidative effect on growing-finishing pigs. J Appl Microbiol 107:1140–1148. https://doi.org/10.1111/j.1365-2672.2009.04294.x
Wang Y, Wu Y, Wang Y, Xu H, Mei X, Dongyou Yu, Wang Y et al (2017) Antioxidant properties of probiotic bacteria. Nutrients 9:521. https://doi.org/10.3390/nu9050521
Westfall S, Lomis N, Singh SP, Prakash S (2016) Ferulic acid produced by Lactobacillus fermentum NCIMB 5221 reduces symptoms of metabolic syndrome in Drosophila melanogaster. J Microb Biochem Technol 8:272–284. https://doi.org/10.1007/s12033-018-0119-y
Yan F, Polk DB (2012) Characterization of a probiotic-derived soluble protein which reveals a mechanism of preventive and treatment effects of probiotics on intestinal inflammatory diseases. Gut Microbes 3:25–28. https://doi.org/10.4161/gmic.19245
Yan F, Cao H, Cover TL, Whitehead R, Washington MK, Polk DB (2007) Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology 132:562–575. https://doi.org/10.1053/j.gastro.2006.11.022
Zhao Y, Kan Hong K, Zhao J, Hao Zhang H, Zhai O, Chen W (2019) Lactobacillus fermentum and its potential immunomodulatory properties. J Funct Foods 56:21–32
Acknowledgements
This work was funded by the State budget of Russian Federation for Genetic technologies in biology, medicine, agricultural and environmental activities (N 0112-2019-0002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors declare a conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Marsova, M., Poluektova, E., Odorskaya, M. et al. Protective effects of Lactobacillus fermentum U-21 against paraquat-induced oxidative stress in Caenorhabditis elegans and mouse models. World J Microbiol Biotechnol 36, 104 (2020). https://doi.org/10.1007/s11274-020-02879-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-020-02879-2