Abstract
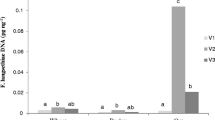
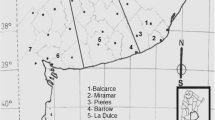
The cereal-pathogenic Fusarium culmorum (W.G. Smith), causal agent of various blights and rot diseases, is considered as a chronic fungus of economic concern worldwide including North African countries such as Algeria. This pathogen produces a wide range of mycotoxins, amongst which the type B-trichothecene deoxynivalenol (DON). In addition to its acute and chronic side effects in livestock and humans, DON is believed to play a determinant role in the pathogenesis toward Triticeae. However, regardless its significant occurrence and impact, little is known about trichothecenes-producing ability of F. culmorum infecting cereals in Algeria. The PCR assay based on Tri genes of 12 F. culmorum strains (designated Fc1–Fc12), which were recovered from several cropping areas of North Algeria, revealed their trichothecenes-producing ability with 3-AcDON genotype. The molecular prediction was confirmed by HPLC analysis. All strains were able to produce the toxin at detectable levels. Strains Fc1 and Fc12 were the highest producers of this mycotoxin with 220 and 230 µg g−1, respectively. The evaluation of pathogenic ability of strains through a barley infesting experiment exhibited the significant disease impact of most strains. Significant correlation between the DON-producing ability of strains and the increase in both disease severity (r = 0.88, P = 0.05) and disease occurrence (r = 0.70, P = 0.05) was observed. Chemotyping of F. culmorum isolates and evaluation of their pathogenic ability are reported for the first time for isolates from Algeria, and highlights the important potential of F. culmorum to contaminate cultivated cereal with DON trichothecenes.




Similar content being viewed by others
References
Bensassi F, Gallerne C, Sharaf El Dein O, Lemaire C, Hajlaoui MR, Bacha H (2012) Involvement of mitochondria-mediated apoptosis in deoxynivalenol cytotoxicity. Food Chem Toxicol 50:1680–1689
Boutigny AL, Barreau C, Atanasova-Penichon V, Verdal-Bonnin MN, Pinson-Gadais L, Richard-Forget F (2009) Ferulic acid, an efficient inhibotor of type B trichothecene biosynthesis and Tri gene expression in Fusarium liquid cultures. Mycol Res 113:746–753
Champeil A, Dore T, Fourbet JF (2004) Fusarium head blight: epidemiological origin of the effects of cultural practices on head blight attacks and the production of mycotoxins by Fusarium in wheat grains. Plant Sci 166:1389–1415
Chandler EA, Simpson DR, Thomsett MA, Nicholson P (2003) Development of PCR assays to Tri7 and Tri13 trichothecene biosynthetic genes, and characterisation of chemotypes of Fusarium graminearum, Fusarium culmorum and Fusarium cerealis. Physiol Mol Plant P 62:355–367
Covarelli L, Beccari G, Steed A, Nicholson P (2012) Colonization of soft wheat following infection of the stem base by Fusarium culmorum and translocation of deoxynivalenol to the head. Plant Pathol 61:1121–1129
Czerwiecki L, Wilczyńska G (2003) Determination of deoxynivalenol in cereals by HPLC-UV. Mycotoxin Res 19:31–34
Desjardins AE (2006) Fusarium mycotoxins: chemistry, genetics, and biology. American Phytopathological Society, St Paul
Desmond OJ, Manners JM, Stephens AE, Maclean DJ, Schenk PM, Gardiner DM, Munn AL, Kazan K (2008) The Fusarium mycotoxin deoxynivalenol elicits hydrogen peroxide production, programmed cell death and defence responses in wheat. Mol Plant Pathol 9:435–445
Fernandez MR, Jefferson PG (2004) Fungal populations in roots and crowns of common and durum wheat in Saskatchewan. Can J Plant Pathol 26:325–334
Guo XW, Fernando WGD, Seow-Brock HY (2008) Population structure, chemotype diversity, and potential chemotype shifting of Fusarium graminearum in wheat fields of Manitoba. Plant Dis 92:756–762
Hestbjerg H, Felding G, Elmholt S (2002) Fusarium culmorum infection of barley seedlings: correlation between aggressiveness and deoxynivalenol content. J Phytopathol 150:308–312
Jennings P, Coates ME, Turner JA, Chandler EA, Nicholson P (2004) Determination of deoxynivalenol and nivalenol chemotypes of Fusarium culmorum isolates from England and Wales by PCR assay. Plant Pathol 53:182–190
Kammoun GL, Gargouri S, Hajlaoui MR, Marrakchi M (2009) Occurrence and distribution of Microdochium and Fusarium species isolated from durum wheat in northern Tunisia and detection of mycotoxins in naturally infested grain. J Phytopathol 57:546–551
Kammoun LG, Gargouri S, Barreau C, Richard-Forget F, Hajlaoui MR (2010) Trichothecene chemotypes of Fusarium culmorum infecting wheat in Tunisia. Int J Food Microbiol 140:84–89
Khan MR, Fischer S, Egan D, Doohan FM (2006) Biological control of Fusarium seedling blight disease of wheat and barley. Phytopathology 96:386–394
Langevin F, Eudes F, Comeau A (2004) The effect of trichothecenes produced by Fusarium graminearum during Fusarium head blight development in six cereal species. Eur J Plant Patholol 110:735–746
Leslie JF, Summerell BA (2006) The Fusarium laboratory manual. Blackwell, Iowa. doi:10.1002/9780470278376
Liu D, Coloe S, Baird R, Pedersen J (2000) Rapid mini-preparation of fungal DNA for PCR. J Clin Microbiol 38:471
Llorens A, Hinojo MJM, Mateo R, Gonzalez-Jaen MT, Valle-Algarra FM, Logrieco A, Jimenez M (2006) Characterization of Fusarium spp. isolates by PCR–RFLP analysis of the intergenic spacer region of the rRNA gene (rDNA). Int J Food Microbiol 106:287–306
Logrieco A, Bottalico A, Mulé G, Moretti A, Perrone G (2003) Epidemiology of toxigenic fungi and their associated mycotoxins for some Mediterranean crops. Eur J Plant Pathol 109:645–667
McCormick SP, Stanley AM, Stover NA, Alexander NJ (2011) Trichothecenes: from simple to complex mycotoxins. Toxins 3:802–814
Merhej J, Richard-Forget F, Barreau C (2011) Regulation of trichothecene biosynthesis in Fusarium: recent advances and new insights. Appl Microbiol Biotechnol 91:519–528
Mirocha CJ, Xie W, Xu Y, Wilcoxson RD, Woodward RP, Etebarian RH, Behele G (1994) Production of trichothecene mycotoxins by Fusarium graminearum and Fusarium culmorum or barley and wheat. Mycopathologia 128:19–23
Nganje WE, Kaitibie S, Wilson WW, Leistritz FL, Bangsund DA (2004) Economic impacts of Fusarium head blight in wheat and barley: 1993–2001. Agribusiness and applied economics report No. 538. North Dakota State University
Niessen ML, Vogel RF (1998) Group specific PCR-detection of potential trichothecene-producing Fusarium species in pure cultures and cereal samples. Syst Appl Microbiol 21:618–631
Pasquali M, Migheli Q (2014) Genetic approaches to chemotype determination in type B-trichothecene producing Fusaria. Int J food Microbiol 189:164–182
Quarta A, Mita G, Haidukowski M, Santino A, Mulè G, Visconti A (2005) Assessment of trichothecene chemotypes of Fusarium culmorum occurring in Europe. Food Addit Contam 22:309–315
Rocha O, Ansari K, Doohan FM (2005) Effects of trichothecene mycotoxins on eukaryotic cells: a review. Food Addit Contam 22:369–378
Schilling AG, Moller EM, Geiger HH (1996) Polymerase chain reaction-based assays for species-specific detection of Fusarium culmorum, F. graminearum and F. avenaceum. Phytopathology 86:515–522
Suzuki T, Iwahashi Y (2014) Phytotoxicity evaluation of type B trichothecenes using a Chlamydomonas reinhardtii model system. Toxins 6:453–463
Tunali B, Obanor F, Erginbaş G, Westecott RA, Nicol J, Chakraborty S (2012) Fitness of three Fusarium pathogens of wheat. FEMS Microbiol Ecol 81:596–609
Vogel HJ (1956) A convenient growth medium for Neurospora (medium N). Microb Genet Bull 13:42–43
Wagacha JM, Muthomi JW (2007) Fusarium culmorum: infection process, mechanisms of mycotoxin production and their role in pathogenesis in wheat. Crop Prot 26:877–885
Winter M, Koopmann B, Döll K, Karlovsky P, Kropf U, Schlüter K, von Tiedemann A (2013) Mechanisms regulating grain contamination with trichothecenes translocated from the stem base of wheat (Triticum aestivum) infected with Fusarium culmorum. Phytopathology 103:682–689
Yekkour A, Sabaou N, Zitouni A, Errakhi R, Mathieu F, Lebrihi A (2012) Characterization and antagonistic properties of Streptomyces strains isolated from Saharan soils, and evaluation of their ability to control seedling blight of barley caused by Fusarium culmorum. Lett Appl Microbiol 55:427–435
Acknowledgments
We thank Dr. Boureghda H, Department of Plant Pathology-High School of Agriculture, Algiers, Algeria, for help with the isolation and the morphologic identification of fungi.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yekkour, A., Toumatia, O., Meklat, A. et al. Deoxynivalenol-producing ability of Fusarium culmorum strains and their impact on infecting barley in Algeria. World J Microbiol Biotechnol 31, 875–881 (2015). https://doi.org/10.1007/s11274-015-1841-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-015-1841-2