Abstract
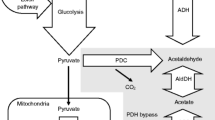
During the mezcal fermentation process, yeasts are affected by several stresses that can affect their fermentation capability. These stresses, such as thermal shock, ethanol, osmotic and growth inhibitors are common during fermentation. Cells have improved metabolic systems and they express stress response genes in order to decrease the damage caused during the stress, but to the best of our knowledge, there are no published works exploring the effect of oxidants and prooxidants, such as H2O2 and menadione, during growth. In this article, we describe the behavior of Kluyveromyces marxianus isolated from spontaneous mezcal fermentation during oxidative stress, and compared it with that of Saccharomyces cerevisiae strains that were also obtained from mezcal, using the W303-1A strain as a reference. S. cerevisiae strains showed greater viability after oxidative stress compared with K. marxianus strains. However, when the yeast strains were grown in the presence of oxidants in the media, K. marxianus exhibited a greater ability to grow in menadione than it did in H2O2. Moreover, when K. marxianus SLP1 was grown in a minibioreactor, its behavior when exposed to menadione was different from its behavior with H2O2. The yeast maintained the ability to consume dissolved oxygen during the 4 h subsequent to the addition of menadione, and then stopped respiration. When exposed to H2O2, the yeast stopped consuming oxygen for the following 8 h, but began to consume oxygen when stressors were no longer applied. In conclusion, yeast isolated from spontaneous mezcal fermentation was able to resist oxidative stress for a long period of time.





Similar content being viewed by others
References
Arellano M, Gschaedler A, Alcazar M (2012) Major volatile compounds analysis produced from mezcal fermentation using gas chromatography equipped headspace (GC–HS), gas chromatography in plant science, wine technology, toxicology and some specific applications. In: Salih B, Çelikbıçak Ö (eds) InTech. pp 73–88. ISBN: 978-953-51-0127-7
Blomberg A (2000) Metabolic surprises in Saccharomyces cerevisiae during adaptation to saline conditions: questions, some answers and a model. FEMS Microbiol Lett 182:1–8
Cardona F, Carrasco P, Pérez-Ortín J, del Olmo M, Aranda A (2007) A novel approach for the improvement of stress resistance in wine yeasts. Int J Food Microbiol 114:83–91
Carrasco P, Querol A, del Olmo M (2001) Analysis of the stress resistance of commercial wine yeast strains. Arch Microbiol 175:450–457
Cohen T, Lee K, Rutkowski H, Strich R (2003) Ask10p mediates the oxidative stress-induced destruction of the Saccharomyces cerevisiae C-type cyclin Ume3p/Srb11p. Eukaryot Cell 2:962–970
Cyrne L, Antunes F, Sousa-Lopes A, Diaz-Bérrio J, Marinho S (2010) Glyceraldehyde-3-phosphate dehydrogenase is largely unresponsive to low regulatory levels of hydrogen peroxide in Saccharomyces cerevisiae. BMC Biochem 11:49
De León-Rodríguez A, González-Hernández L, Barba de la Rosa P, Escalante-Minakata P, López M (2006) Characterization of volatile compounds of mezcal, an ethnic alcoholic beverage obtained from Agave salmiana. J Agric Food Chem 54:1337–1341
Dellomonaco C, Amaretti A, Zanoni S, Pompei A, Matteuzzi D, Rossi M (2007) Fermentative production of superoxide dismutase with Kluyveromyces marxianus. J Ind Microbiol Biotechnol 34:27–34
Demasi A, Pereira G, Netto L (2006) Yeast oxidative stress response. Influences of cytosolic thioredoxin peroxidase I and of the mitochondrial functional state. FEBS J 273:805–816
Eskander J, Lavaud C, Harakat D (2010) Steroidal saponins from leaves of Agave macrocantha. Fitoterapia 81:371–374
Estruch F (2000) Stress-controlled transcription factors, stress-induced genes and stress tolerance in budding yeast. FEMS Microbiol Rev 24:469–486
Fitzgerald DJ, Stratford M, Gasson MJ, Ueckert J, Bos A, Narbad A (2004) Mode of antimicrobial action of vanillin against Escherichia coli, Lactobacillus plantarum and Listeria innocua. J Appl Microbiol 97:104–113
Flattery-O’Brien J, Collinson LP, Dawes IW (1993) Saccharomyces cerevisiae has an inducible response to menadione which differs from that to hydrogen peroxide. J Gen Microbiol 139:501–507
Gershon H, Gershon D (2000) The budding yeast, Saccharomyces cerevisiae, as a model for aging research: a critical review. Mech Ageing Dev 120:1–22
Gille G, Sigler K (1995) Oxidative stress and living cells. Folia Microbiol 40:131–152
Gschaedler A, Ramírez J, Díaz D, Herrera E, Arrizón J, Pinal L, Arellano M (2004) Fermentación. En: Ciencia y Tecnología del Tequila Avances y Perspectivas. CIATEJ, Guadalajara, Jalisco, México, pp 61–120
Halliwell B, Gutteridge C (2007) Free radicals in biology and medicine, 4th edn. Oxford University Press, Oxford, pp 488–613
Herdeiro RS, Pereira MD, Panek AD, Eleutherio EC (2006) Trehalose protects Saccharomyces cerevisiae from lipid peroxidation during oxidative stress. Biochim Biophys Acta 1760:340–346
Jamieson DJ (1992) Saccharomyces cerevisiae has distinct adaptive responses to both hydrogen peroxide and menadione. J Bacteriol 174:6678–6681
Joon L, Ian WD, Jung-Hye R (1995) Adaptive response of Schizosaccharomyces pombe to hydrogen peroxide and menadione. Microbiology 141:3127–3132
Kim IS, Moon HY, Yun HS, Jin I (2006) Heat shock causes stress and induces a variety of cell rescue proteins in Saccharomyces cerevisiae KNU5377. J Microbiol 44:492–501
Lappe-Oliveras P, Moreno-Terrazas R, Arrizón-Gaviño J, Herrera-Suárez T, García-Mendoza A, Gschaedler-Mathis A (2008) Yeasts associated with the production of Mexican alcoholic nondistilled and distilled Agave beverages. FEMS Yeast Res 8:1037–1052
Martínez-Pastor MT, Marchler G, Schüller C, Marchler-Bauer A, Ruis H (1996) The Saccharomyces cerevisiae zinc finger proteins Msn2p and Msn4p are required for transcriptional induction through the stress response element (STRE). EMBO J 15:2227–2235
Moradas-Ferreira P, Costa V, Piper P, Mager W (1996) The molecular defenses against reactive oxygen species in yeast. Mol Microbiol 19:651–658
NOM-070-SCFI-1997 (1997) Bebidas alcohólicas, Mezcal-Especificaciones. SCFI, México, DF, pp 2–31
Ojovan SM, Knorre DA, Markova OV, Smirnova EA, Bakeeva LE, Severin FF (2011) Accumulation of dodecyltriphenylphosphonium in mitochondria induces their swelling and ROS-dependent growth inhibition in yeast. J Bioenerg Biomembr 43:175–180
Osorio H, Carvalho E, del Valle M, Günther-Sillero A, Moradas-Ferreira P, Sillero A (2003) H2O2, but not menadione, provokes a decreased in the ATP and increase in the inosine levels in Saccharomyces cerevisiae. An experimental and theoretical approach. Eur J Biochem 270:1578–1589
Palmqvist J, Almeida S, Hahn-Hägerdal B (1999) Influence of furfural on anaerobic glycolytic kinetics of S. cerevisiae in bath culture. Biotechnol Bioeng 62:447–454
Pereira da Silva B, Valente AP, Paz Parente P (2006) A new steroidal saponin from Agave shrevei. Nat Prod Res 20:385–390
Pérez-Torrado R, Gómez-Pastor R, Larsson C, Matallana E (2009) Fermentative capacity of dry active wine yeast requires a specific oxidative stress response during industrial biomass growth. Appl Microbiol Biotechnol 81:951–960
Pinheiro R, Belo I, Mota M (2002) Oxidative stress response of Kluyveromyces marxianus to hydrogen peroxide, paraquat and pressure. Appl Microbiol Biotechnol 58:842–847
Rodríguez-Manzaneque MT, Ros J, Cabiscol E, Sorribas A, Herrero E (1999) Grx5 glutaredoxin plays a central role in protection against protein oxidative damage in Saccharomyces cerevisiae. Mol Cell Biol 19:8180–8190
Romano P, Capece A, Jespersen L (2006) Taxonomic and ecological diversity of food and beverage yeasts. In: Querol A, Fleet G (eds) Yeasts in food and beverages. Springer, Berlin, pp 13–53
Sánchez Y, Taulien J, Borkovich KA, Lindquist S (1992) Hsp104 is required for tolerance to many forms of stress. EMBO J 11:2357–2364
Stephen S, Jamieson DJ (1997) Amino acid-dependent regulation of the Saccharomyces cerevisiae GSH1 gene by hydrogen peroxide. Mol Microbiol 23:203–210
Thorpe GW, Fong CS, Alic N, Higgins VJ, Dawes IW (2004) Cells have distinct mechanisms to maintain protection against different reactive oxygen species: oxidative-stress-response genes. Proc Natl Acad Sci USA 101:6564–6569
Westwater C, Balish E, Schofield DA (2005) Candida albicans conditioned medium protects yeast cells from oxidative stress: a possible link between quorum sensing. Eukaryot Cell 10:1654–1661
Zakrajsek T, Raspor P, Jamnik P (2011) Saccharomyces cerevisiae in the stationary phase as a model organism—characterization at cellular and preotome level. J Proteomics 74:2837–2845
Acknowledgments
The authors appreciate the partial economic support from the Coordinación de la Investigación Científica-UMSNH to AS-M (2.16), to SM-A (2.37), and from the Consejo Nacional de Ciencia y Tecnología-México to AS-M (169093). The authors would also like to thank the Consejo Nacional de Ciencia y Tecnología-México (CONACyT) for the grant that made this study possible.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arellano-Plaza, M., Gschaedler-Mathis, A., Noriega-Cisneros, R. et al. Respiratory capacity of the Kluyveromyces marxianus yeast isolated from the mezcal process during oxidative stress. World J Microbiol Biotechnol 29, 1279–1287 (2013). https://doi.org/10.1007/s11274-013-1291-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1291-7