Abstract
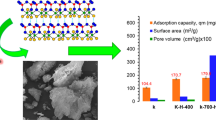
So far, the purification of dyes wastewater through adsorption and other economical and environmentally friendly methods had received great attention. Therefore, this study mainly discussed the role and rule of PA65 material in wastewater dye treatment. In the experiment, the effects of pH value of solution, initial dye concentration, amount of adsorbent and adsorption time on the removal rate of orange G were studied. It was found that under the conditions of pH < 6, initial dye concentration of about 70 mg·L−1, amount of adsorbent of about 60 mg, and adsorption time of 120 min, good adsorption performance could be achieved. Under acidic conditions, the PA65 with a pore structure exhibited certain adsorption performance for anionic dyes and had higher selectivity for binary mixed dyes. FITR analysis confirmed the presence of amide groups and amino groups interacting with dye molecules. In addition, the experimental data was fitted by adsorption isotherms and adsorption kinetics model. The results showed that the Langmuir model best describes the equilibrium isotherm data, showing a maximum adsorption capacity of 63.17 mg‧g−1, and the adsorption data follows the pseudo-second-order dynamics model. The thermodynamic properties revealed that the adsorption process was endothermic and spontaneous.















Similar content being viewed by others
Data Availability
The author declares that all data and materials are of actual availability.
References
Abbas, F. S. (2013). Dyes removal from wastewater using agricultural waste. Advances in Environmental Biology, 7(6), 1019–1026.
Ayad, M. M., & El-Nasr, A. A. (2010). Adsorption of cationic dye (methylene blue) from water using polyaniline nanotubes base. Journal of Physical Chemistry C, 114(34), 2177–2187. https://doi.org/10.1021/jp103780w
Amin, N. K. (2008). Removal of reactive dye from aqueous solutions by adsorption on to activated carbons prepared from sugarcane bagasse pith. Desalination, 223(1), 152–161. https://doi.org/10.1016/j.desal.2007.01.203
Attallah, M. F., Ahmed, I. M., & Hamed, M. M. (2013). Treatment of industrial wastewater containing Congo Red and Naphthol Green B using low-cost adsorbent. Environmental Science & Pollution Research, 20(2), 1106–1116. https://doi.org/10.1007/s11356-012-0947-4
Allen, S., Koumanova, B., (2005). Decolourisation of water/wastewater using adsorption 40(3):175-192
Bhattacharyya, K. G., & Gupta, S. S. (2007). Adsorptive accumulation of Cd(II), Co(II), Cu(II), Pb(II), and Ni(II) from water on montmorillonite: Influence of acid activation. Journal of Colloid & Interface Science, 310(2), 411–424. https://doi.org/10.1016/j.jcis.2007.01.080
Banerjee, S., Chattopadhyaya, M. C., & Sharma, Y. C. (2015). Removal of an azo dye (Orange G) from aqueous solution using modified sawdust. Journal of Water Sanitation & Hygiene for Development, 5(2), 235. https://doi.org/10.2166/washdev.2014.214
Barcelo, D., (2001). Modern fourier transform infrared spectroscopy
Bulut, Y., & Aydın, H. (2006). A kinetics and thermodynamics study of methylene blue adsorption on wheat shells. Desalination, 194(1–3), 259–267. https://doi.org/10.1016/j.desal.2005.10.032
Bai, B., Bai, F., Li, X. K., Nie, Q. K., Jia, X. X., & Wu, H. Y. (2022). The remediation efficiency of heavy metal pollutants in water by industrial red mud particle waste. Environmental Technology & Innovation, 28(10), 102944. https://doi.org/10.1016/j.eti.2022.102944-
Giraldo, S., Ramirez, A. P., Flórez, E., & Acelas, N. Y. (2019). Adsorbent materials obtained from palm waste and its potential use for contaminants removal from aqueous solutions. Journal of Physics: Conference Series, 1386(1), 012036. https://doi.org/10.1088/1742-6596/1386/1/012036
Ghorai, S., Sarkar, A., Raoufi, M., Syieluing, W., & Ngadi, N. (2014). Enhanced removal of methylene blue and methyl violet dyes from aqueous solution using a nanocomposite of hydrolyzed polyacrylamide grafted xanthan gum and incorporated nanosilica. Acs Applied Materials & Interfaces., 6(7), 4766–4777. https://doi.org/10.1021/am4055657
Ho, Y. S., & Mckay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry., 34(5), 451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Imgharn, A., Ighnih, H., Hsini, A., Naciri, Y., Laabd, M., Kabli, H., Elamine, M., Lakhmiri, R., Souhail, B., & Albourine, A. (2021). Synthesis and characterization of polyaniline-based biocomposites and their application for effective removal of Orange G dye using adsorption in dynamic regime. Chemical Physics Letters, 778(9), 138811. https://doi.org/10.1016/j.cplett.2021.138811
Imgharn, A., Anchoum, L., Hsini, A., Naciri, Y., Laabd, M., Mobarak, M., Aarab, N., Bouziani, A., Szunerits, S., Boukherroub, R., Lakhmiri, R., & Albourine, A. (2022). Effectiveness of a novel polyaniline@Fe-ZSM-5 hybrid composite for Orange G dye removal from aqueous media: Experimental study and advanced statistical physics insights. Chemosphere, 295, 133786. https://doi.org/10.1016/j.chemosphere.2022.133786
Jeppu, G. P., & Clement, T. P. (2012). A modified Langmuir-Freundlich isotherm model for simulating pH-dependent adsorption effects. Journal of Contaminant Hydrology., 129–130, 46–53. https://doi.org/10.1016/j.jconhyd.2011.12.001
Kishor, R., Purchase, D., Saratale, G. D., Saratale, R. G., Ferreira, L. F. R., Bilal, M., Chandra, R., & Bharagava, R. N. (2021). Ecotoxicological and health concerns of persistent coloring pollutants of textile industry wastewater and treatment approaches for environmental safety. Journal of Environmental Chemical Engineering, 9(2), 105012. https://doi.org/10.1016/j.jece.2020.105012
Kyzas, G. Z., Kostoglou, M., Vassiliou, A. A., & Lazaridis, N. K. (2011). Treatment of real effluents from dyeing reactor: Experimental and modeling approach by adsorption onto chitosan. Chemical Engineering Journal, 168(2), 577–585. https://doi.org/10.1016/j.cej.2011.01.026
Lee, L. Y., Gan, S., Tan, M. S. Y., Lim, S. S., Lee, S. J., & Lam, Y. F. (2016). Effective removal of Acid Blue 113 dye using overripe Cucumis sativus peel as an eco-friendly biosorbent from agricultural residue. Journal of Cleaner Production, 113(1), 194–203. https://doi.org/10.1016/j.jclepro.2015.11.016
Li, S. B., Jia, Z. G., Li, Z. Y., Li, Y. H., & Zhu, R. G. (2016). Synthesis and characterization of mesoporous carbon nanofibers and its adsorption for dye in wastewater. Advanced Powder Technology the Internation Journal of the Society of Powder Technology Japan, 27(2), 591–598. https://doi.org/10.1016/j.apt.2016.01.024
Mukurala, N., Mokurala, K., Suman, S., & Kushwaha, A. K. (2021). Synthesis of porous Cu2FeSnS 4 particles via solvothermal process for removal of organic acid fuchsin dye pollutant from wastewater. Nano-Structures & Nano-Objects, 26, 100697. https://doi.org/10.1016/j.nanoso.2021.100697
Morales-Gámez, L., Ricart, A., Franco, L., & Puiggalí, J. (2010). Study on the brill transition and melt crystallization of nylon 65: A polymer able to adopt a structure with two hydrogen-bonding directions. European Polymer Journal, 46(10), 2063–2077. https://doi.org/10.1016/j.eurpolymj.2010.07.010
Navarro, E., Franco, L., Subirana, J. A., & Puiggalí, J. (1995). Nylon 65 has a unique structure with two directions of hydrogen bonds. Macromolecules, 28(26), 8742–8750. https://doi.org/10.1021/ma00130a006
Nordin, A. H., Ahmad, K., Xin, L. K., Syieluing, W., & Ngadi, N. (2021). Efficient adsorptive removal of methylene blue from synthetic dye wastewater by green alginate modified with pandan. Materials Today: Proceedings, 9(2), 979–982. https://doi.org/10.1016/j.matpr.2020.04.564
Ogata, N., Sanui, K., Tanaka, H., & Takahashi, Y. (1981). Cellular affinity of polyamides having various functional groups. I. Platelet Adhesio. Journal of Applied Polymer Science, 26(7), 2293–2303. https://doi.org/10.1002/app.1981.070260716
Ouachtak, H., Guerdaoui, A. E. I., Haounati, R., Akhouairi, S., Haouti, R. E. I., Hafid, N., Addi, A. A., Šljukic´, B., Santos, D. M. F., Taha, M. L. (2021). Highly efficient and fast batch adsorption of Orange G dye from polluted water using superb organo-montmorillonite: Experimental study and molecular dynamics investigation. Journal of Molecular Liquids, 335, 116560. https://doi.org/10.1016/j.molliq.2021.116560
Qureshi, U. A., Khatri, Z., Ahmed, F., Ibupoto, A. S., Khatri, M., Mahar, F. A., Brohi, R. Z., & Kim, I. S. (2017). Highly efficient and robust electrospun nanofibers for selective removal of acid dye. Journal of Molecular Liquids, 244(10), 478–488. https://doi.org/10.1016/j.molliq.2017.08.129
Rohles, C. M., Gläser, L., Kohlstedt, M., Gießelmann, G., Pearson, S., Campo, A. D., Becker, J., & Wittmann, C. (2018). A bio-based route to the carbon-5 chemical glutaric acid and to bionylon-6,5 using metabolically engineered Corynebacterium glutamicum. Green Chemistry, 20, 4662. https://doi.org/10.1039/C8GC01901K
Rizvi, A., Chu, R. K. M., Lee, J. H., & Park, C. B. (2014). Superhydrophobic and oleophilic open-cell foams from fibrillar blends of polypropylene and polytetrafluoroethylene. Acs Applied Materials & Interfaces, 6(23), 21131–21140. https://doi.org/10.1021/am506006v
Selvaraj, V., Karthika, T. S., Mansiya, C., & Alagar, M. (2020). An over review on recently developed techniques, mechanisms and intermediate involved in the advanced azo dye degradation for industrial applications. Journal of Molecular Structure, 1224(2), 129195. https://doi.org/10.1016/j.molstruc.2020.129195
Srinivasan, A., & Viraraghavan, T. (2010). Decolorization of dye wastewaters by biosorbents: A review. Journal of Environmental Management, 91(10), 1915–1929. https://doi.org/10.1016/j.jenvman.2010.05.003
Sahnoun, S., & Boutahala, M. (2018). Adsorption removal of tartrazine by chitosan/polyaniline composite: Kinetics and equilibrium studies. International Journal of Biological Macromolecules, 114, 1345. https://doi.org/10.1016/j.ijbiomac.2018.02.146
Saini, J., Garg, V. K., Gupta, R. K., & Kataria, N. (2017). Removal of Orange G and Rhodamine B dyes from aqueous system using hydrothermally synthesized zinc oxide loaded activated carbon (ZnO-AC). Journal of Environmental Chemical Engineering, 5(1), 884–892. https://doi.org/10.1016/j.jece.2017.01.012
Sutirman, Z. A., Sanagi, M. M., Karim, K. J. A., Naim, A. A., & Ibrahim, W. A. W. (2019). Enhanced removal of Orange G from aqueous solutions by modified chitosan beads: Performance and mechanism. International Journal of Biological Macromolecules, 133, 1260–1267. https://doi.org/10.1016/j.ijbiomac.2019.04.188
Salam, M. A., Kosa, S. A., & Al-Beladi, A. A. (2017). Application of nanoclay for the adsorptive removal of Orange G dye from aqueous solution. Journal of Molecular Liquids, 241, 467–477. https://doi.org/10.1016/j.molliq.2017.06.055
Salleh, M. A. M., Mahmoud, D. K., Karim, W. A. W. A., & Idris, A. (2011). Cationic and anionic dye adsorption by agricultural solid wastes: A comprehensive review. Desalination, 280(1–3), 1–13. https://doi.org/10.1016/j.desal.2011.07.019
Taher, T., Munandar, A., Mawaddah, N., Wisnubroto, M. S., Siregar, P. M. S. B. N., Palapa, N. R., Lesbani, A., & Wibowo, Y. G. (2022). Synthesis and characterization of montmorillonite – Mixed metal oxide composite and its adsorption performance for anionic and cationic dyes removal. Inorganic Chemistry Communications, 147(11), 110231. https://doi.org/10.1016/j.inoche.2022.110231
Wojnárovits, L., & Takács, E. (2008). Irradiation treatment of azo dye containing wastewater: An overview. Radiation Physics and Chemistry, 77(3), 225–244. https://doi.org/10.1016/j.radphyschem.2007.05.003
Wang, Y., Jaromír, M., Jakub, W., Zhu, G.C., (2015). Study on the acid dye removal by polyamide 6 nanofibrous membrane. NANOCON, 10
Wang, X. Q., Zhang, L., Shi, X. H., Gao, Z. C., Zuo, Y. (2021). A kind of nylon 65 salt melt polymerization to prepare spinnable nylon 65. CN111253568A
Wang, L., Ai, F. F., Liu, Y. S., Liu, Y. (2017). Porous polyamide monolith by continuous solution foaming as reusable oils and organic solvents absorbent. Materials Letters, 213(FEB.15), 44-47. https://doi.org/10.1016/j.matlet.2017.11.017
Wan, Z. X., Zhang, T. K., Liu, Y. Z., Liu, P., Zhang, J. W., Fang, L., & Sun, D. H. (2022). Enhancement of desulfurization by hydroxyl ammonium ionic liquid supported on active carbon. Environmental Research, 213(7), 113637. https://doi.org/10.1016/j.envres.2022.113637-
Yagub, M. T., Sen, T. K., Afroze, S., & Ang, H. M. (2014). Dye and its removal from aqueous solution by adsorption: A review. Advances in Colloid and Interface Science, 209(2), 172–184. https://doi.org/10.1016/j.cis.2014.04.002
Zarrini, K., Allah, R. A., Alihosseini, F., & Fashandi, H. (2017). Highly efficient dye adsorbent based on polyaniline-coated nylon-6 nanofibers. Journal of Cleaner Production, 142(3), 3645–3654. https://doi.org/10.1016/j.jclepro.2016.10.103
Zhou, Y. B., Lu, J., Zhou, Y., & Liu, Y. D. (2019). Recent advances for dyes removal using novel adsorbents: A review. Environmental Pollution, 252(6), 19–25. https://doi.org/10.1016/j.envpol.2019.05.072
Zheng, X. Y., Zheng, H. L., Zhou, Y. H., Sun, Y. J., Zhao, R., Liu, Y. Z., & Zhang, S. X. (2019). Enhanced adsorption of Orange G from aqueous solutions by quaternary ammonium group-rich magnetic nanoparticles. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 580, 123746. https://doi.org/10.1016/j.colsurfa.2019.123746
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
The authors declare that this experiment did not involve human participants and animal studies.
Consent to Participate
All authors agree to participate.
Consent to Publish
All authors agree to publish.
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, Z., Zhang, L., Shi, X. et al. Study on the Adsorption of Orange G dye by the PA65 with Pore Structure. Water Air Soil Pollut 234, 230 (2023). https://doi.org/10.1007/s11270-023-06248-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-023-06248-1