Abstract
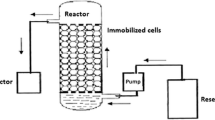
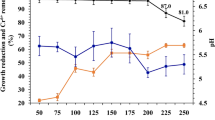
Immobilized microbial cells for the biological treatment have the potential to degrade toxic chemicals faster than conventional wastewater treatment systems. In the present study, suitability of immobilized Pseudomonas fluorescens SM1 strain in calcium alginate beads for remediation of the major toxicants in Indian water bodies was tested by means of GC/HPLC and AAS techniques. Roughly 80% reduction in the concentration of phenols was observed by immobilized SM1 cells compared with 60% by the free cells. Also, in the case of the bioremediation of heavy metals, immobilized SM1 cells were found to be more efficient compared with the free cells. Suspension of P. fluorescens SM1 cells in the test model water for 24 h brought down the concentrations of Cu++, Cd++, Ni++, and Pb++ by more than 75% under free cell state and 7–9% better efficiency under the immobilized conditions. However, Cr(VI) could show only 44% removal by the cell immobilized system, whereas a mere 35% reduction in the Cr(VI) levels was shown in the test model water by the free SM1 cells under the same conditions. Moreover, a model water containing 2,000 ppb of BHC, 1,248 ppb mancozeb, and 312 ppb 2,4-D passed through the cell immobilized column resulted in the decline in their concentrations up to 362 ppb, 750 ppb, and 126 ppb, respectively. Generally, AAS, HPLC, and GC analyses of treated test model waters with the free and immobilized SM1 cells exhibited high potential of immobilized SM1 in detoxification of test water. From the results, we conclude that immobilized cells of P. fluorescens SM1 strain were quite effective in bioremediation of major toxicants present in Indian water bodies, and we also recommend the use of immobilized bacterial cells rather than the free cells for the bioremediation/detoxification process.


Similar content being viewed by others
References
Aggelis, G., Ehiiotis, C., Nerud, F., Stoychev, I., Lyberatos, G., & Zervakis, G. I. (2002). Evaluation of white-rot fungi for detoxification and decolorization of effluents from the green olive debittering process. Applied Microbiology and Biotechnology, 59, 353–360.
American Public Health Association (APHA). (1998). Standard Methods for the Examination of Water and Wastewater (20th ed.). Washington, DC: APHA.
Audet, P., Paquin, C., & Lacroix, C. (1989). Sugar utilization and acid production by free and entrapped cells of Streptococcus salivarius subsp. thermophilus, Lactobacillus delbrueckii subsp. bulgaricus, and Lactococcus lactis subsp. lactis in a whey permeate medium. Applied and Environmental Microbiology, 55, 185–189.
Bachmann, A., Walet, P., Wijnen, P., De Bruin, W., Hunljens, J. L. M., Roelofsen, W., et al. (1988). Biodegradation of alpha- and beta-hexachlorocyclohexane in a soil slurry under different redox conditions. Applied and Environmental Microbiology, 54, 143–149.
Barlett, R., & James, B. R. (1979). Behaviour of chromium in soils. III. Oxidation. Journal of Environmental Quality, 8, 31–34.
Bettman, H., & Rehm, H. J. (1984). Degradation of phenol by polymer entrapped microorganisms. Applied Microbiology Biotechnology, 20, 285–290.
Bhushan, R., Thapar, S., & Mathtir, R. P. (1997). Accumulation pattern of pesticides in tropical fresh waters. Biomedical Chromatography, 11, 143–150.
BIS 13832 (1993). Bureau of Indian Standards, New Delhi.
Cassidy, M. B., Lee, H., & Trevors, J. T. (1996). Environmental applications of immobilized microbial cells, a review. Journal of Industrial Microbiology, 16, 79–101.
Cassidy, M. B., Shaw, K. W., Lee, H., & Trevors, J. T. (1997). Enhanced mineralization of pentachlorophenol by κ-carrageenan encapsulated Pseudomonas sp. UG30. Applied Microbiology and Biotechnology, 47, 108–113.
Cheetham, P. S. J., Blunt, K. W., & Bocke, C. (1979). Physical studies on cell immobilization using calcium alginate gels. Biotechnology and Bioengineering, 21(12), 2155–2168.
De, A. K., (1994). In: Environmental chemistry, Wiley Eastern Limited, New Age International Publishers Ltd., New Delhi
de-Bashan, L. E., & Bashan, Y. (2008). Joint immobilization of plant growth-promoting bacteria and green microalgae in alginate beads as an experimental model for studying plant–bacterium interactions. Applied and Environmental Microbiology, 74(21), 6797–6802.
Fang, S. C. (1969). In P. C. Kearney & D. D. Kaufman (Eds.), Degradation of herbicides. New York: Marcel Dekker.
Frioni, L., Le Roux, C., Dommergues, Y. R., & Diem, H. G. (1994). Inoculant made of encapsulated Frankia, assessment of Frankia growth within alginate beads. World Journal of Microbiology & Biotechnology, 10, 118–121.
Fulthorpe, R. R., Rhodes, A. N., & Tiedje, J. M. (1996). Pristine soils mineralize 3-chlorobenzoate and 2, 4-dichlorophenoxyacetate via different microbial populations. Applied and Environmental Microbiology, 62, 1159–1166.
Ganguli, A., & Tripathi, A. K. (2002). Bioremediation of toxic chromium from electroplating effluent by chromate reducing Pseudomonas aeruginosa A2chr in two bioreactors. Applied Microbiology and Biotechnology, 58, 416–420.
Gardin, H., & Pauss, A. (2001). κ-carrageenan/gelatin gel beads for the co-immobilization of aerobic and anaerobic microbial communities degrading 2, 4, 6-trichlorophenol under air-limited conditions. Applied Microbiology and Biotechnology, 56, 517–523.
Haider, K. (1979). Degradation and metabolization of lindane and other hexachlorocyclohexane isomers by anaerobic and aerobic soil microorganisms. Zeitschrift für Naturforschung. Teil C, 34, 1066–1069.
Heitkamp, M. A., Camel, V., Reuter, T. J., & Adams, W. J. (1990). Biodegradation of p-nitrophenol in an aqueous waste stream by immobilized bacteria. Applied and Environmental Microbiology, 56(10), 2967–2973.
Heritage, A. D., & MacRae, I. C. (1977). Degradation of lindane by cell-free preparations of Clostridium sphenoides. Applied and Environmental Microbiology, 34(2), 222–224.
Ignatov, O. V., Gulii, O. I., Singirtsev, I. N., Scherbakov, A. A., Makarov, O. E., & Ignatov, V. V. (2002). Effects of p-nitrophenol and organophosphorous nitroaromatic insecticides on the respiratory activity of free and immobilized cells of strains S-11 and BA-11 of Pseudomonas putida. Prikladnaia Biokhimiia i Mikrobiologiia, 38, 278–285.
Imai, R., Nagata, Y., Fukuda, M., Takagi, M., & Yano, K. (1991). Molecular cloning of a Pseudomonas paucimobilis gene encoding a 17-kilodalton polypeptide that eliminates HCl molecules from gamma-hexachlorocyclohexane. Journal of Bacteriology, 173, 6811–6819.
Jagnow, G., Haider, K., & Ellwardt, P. C. (1977). Anaerobic dechlorination and degradation of hexachlorocyclohexane isomers by anaerobic and facultative anaerobic bacteria. Archives of Microbiology, 115, 285–292.
Johri, A. K., Dua, M., Tuteja, D., Saxena, R., Saxena, D. M., & Lal, R. (1998). Degradation of alpha, beta, gamma and delta-hexachlorocylohexanes by Sphingomonas paucimobilis. Biotechnological Letters, 20, 885–889.
Kaufman, D. D. (1967). Degradation of carbamate herbicides in soil. Journal of Agricultural and Food Chemistry, 15, 582–591.
Kearney, P. C., & Kaufman, D. D. (1965). Enzyme from soil bacterium hydrolyses phenylcarbamate herbicides. Science, 147, 740–741.
Keweloh, H., Heipieper, H. J., & Rehm, H. J. (1989). Protection of bacteria against toxicity of phenol by immobilization in calcium alginate. Applied Microbiology and Biotechnology, 31, 383–389.
Kissi, M., Mountadar, M., Assobhei, O., Gargiulo, E., Palmieri, G., Giardina, P., et al. (2001). Roles of two white-rot basidiomycete fungi in decolorisation and detoxification of olive mill waste water. Applied Microbiology and Biotechnology, 57, 221–226.
Kumari, R., Subudhi, S., Suar, M., Dhingra, G., Raina, V., Dogra, C., et al. (2002). Cloning and characterization of lin genes responsible for the degradation of hexachlorocyclohexane isomers by Sphingomonas paucimobilis strain B90. Applied and Environmental Microbiology, 68, 6021–6028.
Kurosawa, H., Nomura, N., & Tanaka, H. (1989). Ethanol production from starch by a co-immobilized mixed culture system of Aspergillus awamori and Saccharomyces cerevisiae. Biotechnology and Bioengineering, 33(5), 716–723.
Lebeau, T., Bagot, D., Jezequel, K., & Fabre, B. (2002). Cadmium biosorption by free and immobilised microorganisms cultivated in a liquid soil extract medium, effects of Cd, pH and techniques of culture. The Science of the Total Environment, 291, 73–83.
Li, W. Y., Xu, Y., & Feng, J. (2009). Treatment of coking wastewater by using an immobilized-microbial-cell anaerobic–aerobic system. Energy Sources, 31, 1397–1405.
Linko, P., & Linko, Y. Y. (1983). Application of immobilized microbial cells. In I. Chibata & L. B. Wingard Jr. (Eds.), Immobilized microbial cells. Applied Biochemistry and Bioengineering (pp. 54–151). New York: Academic.
Lo, W., Chua, H., Wong, M. F., & Yu, P. (2003). Bacterial biosorbent for removing and recovering copper from electroplating effluents. Water Science and Technology, 47, 251–256.
MacRae, I. C., Raghu, K., & Castro, T. F. (1967). Persistence and biodegradation of four common isomers of benzene hexachloride in submerged soils. Journal of Agricultural and Food Chemistry, 15, 911–914.
MacRae, I. C., Yamaya, & Yoshida, T. (1984). Persistence of hexachlorocyclohexane isomers in soil suspensions. Soil Biology and Biochemistry, 16, 285–286.
Mater, D. D. G., Barbotin, J. N., Saucedo, J. E. N., & Thomes, T. N. (1995). Effect of gelation temperature on gel-dissolving solution on cell viability and recovery of two Pseudomonas putida strains co-immobilized within calcium alginate or κ-carrageenan gel beads. Biotechnology Techniques, 9, 747–752.
Megharaj, M., Avudainayagam, S., & Naidu, R. (2003). Toxicity of hexavalent chromium and its reduction by bacteria isolated from soil contaminated with tannery waste. Current Microbiology, 47(1), 51–54.
Nagata, Y., Nariya, T., Ohtomo, R., Fukuda, M., Yano, K., & Takagi, M. (1993). Cloning and sequencing of a dehalogenase gene encoding an enzyme with hydrolase activity involved in the degradation of gamma-hexachlorocyclohexane in Pseudomonas paucimobilis. Journal of Bacteriology, 75(20), 6403–6410.
Nawab, A., Aleem, A., & Malik, A. (2003). Determination of organochlorine pesticides in agricultural soil with special reference to γ-HCH degradation by Pseudomonas strains. Bioresource Technology, 88(1), 41–46.
Ohisa, N., & Yamaguchi, M. (1978). Gamma-BHC degradation accompanied by the growth of Clostridium rectum isolated from paddy field soil. Agricultural and Biological Chemistry, 42, 1819–1823.
Ohisa, N., Yamaguchi, M., & Kurihara, N. (1980). Lindane degradation by cell-free extracts of Clostridium rectum. Archives of Microbiology, 125, 221–225.
Pandey, A. K., Pandey, S. D., Misra, V., & Srimal, A. K. (2003). Removal of chromium and reduction of toxicity to Microtox system from tannery effluent by the use of calcium alginate beads containing humic acid. Chemosphere, 51, 329–333.
Paul, E., Fages, J., Blanc, P., Goma, G., & Pareilleux, A. (1993). Survival of alginate-entrapped cells of Azospirillum lipoferum during dehydration and storage in relation to water properties. Applied Microbiology and Biotechnology, 40, 34–39.
Podowski, A. A., Feroz, M., Mertens, P., & Khan, M. A. Q. (1984). HPLC analysis of organochlorines using UV and radioactivity flow detectors. Bulletin of Environmental Contamination and Toxicology, 32, 301–309.
Rani, M. J., Hemambika, J., Hemapriya, J., & Kannan, V. R. (2010). Comparative assessment of heavy metal removal by immobilized and dead bacterial cells: a biosorption approach. African Journal of Environmental Science Technology, 4(2), 77–83.
Rehana, Z., Malik, A., & Ahmad, M. (1995). Mutagenic activity of the Ganges water with special reference to the pesticide pollution in the river between Kachla to Kannauj (U.P.), India. Mutation Research, 343, 137–144.
Sag, Y., Ozer, D., & Kutsal, T. (1995). A comparative study of the biosorption of Pb(l1) ions to Z. ramigera and R. arrhitus. Process Biochemistry, 30, 169–174.
Sahu, S. K., Patnaik, K. K., Sharmila, M., & Sethunathan, N. (1990). Degradation of alpha-, beta-, and gamma-hexachlorocyclohexane by a soil bacterium under aerobic conditions. Applied and Environmental Microbiology, 56, 3620–3622.
Saxena, D., Joshi, N., & Srivaslava, S. (2002). Mechanism of copper resistance in a copper mine isolate Pseudomonas putida strain S4. Current Microbiology, 45, 410–414.
Senoo, K., & Wada, H. (1989). Isolation and identification of an aerobic y-HCH-decomposing bacterium from soil. Soil Science and Plant Nutrition, 35, 79–87.
Singh, K. P., Takroo, R., Ray, P. K. (1987). Analysis of pesticide residues in water. ITRC Manual No. 1, Industrial Toxicology Research Centre, Lucknow (UP) India
Thomas, J. C., Berger, F., Jacquicr, M., Bernikkon, D., Baud-Grasset, F., Truffaul, N., et al. (1996). Isolation and characterization of a novel y-hexachlorohexane degrading bacterium. Journal of Bacteriology, 178, 6049–6055.
Trevors, J. T., Elasas, J. D., Lee, H., & Welters, A. C. (1993). Survival of alginate-encapsulated Pseudomonas fluorescens cells in soil. Applied Microbiology and Biotechnology, 39, 637–643.
Tsekova, K., & Ilieva, S. (2001). Copper removal from aqueous solution using Aspergillus niger mycelia in free and polyurethane bound form. Applied Microbiology and Biotechnology, 42, 636–637.
van Elsas, J. D., Trevors, J. T., Jain, D., Wolters, A. C., Heijnen, C. E., & van Overbeek, L. S. (1992). Survival of, and colonization by, alginate-encapsulated Pseudomonas fluorescens cells following introduction into soil. Biology and Fertility of Soils, 14, 14–22.
Vílchez, C., & Vega, J. M. (1994). Nitrite uptake by Chlamydomonas reinhardtii cells immobilized in calcium alginate. Applied Microbiology Biotechnology, 41, 137–141.
Wasi, S., Jeelani, G., & Ahmad, M. (2008). Biochemical characterization of a multiple heavy metal, pesticides and phenol resistant Pseudomonas fluorescens strain. Chemosphere, 71, 1348–1355.
Wasi, S., Tabrez, S., & Ahmad, M. (2010). Isolation and characterization of a Pseudomonas fluorescens strain tolerant to major Indian water pollutants. Journal of Bioremediation and Biodegradation, 1, 101. doi:10.4172/2155-6199.1000101.
Windholz, M., Budavari, S., Stroumtosos, L. Y., & Fertig, M. N. (1976). The Merck index (9th ed., pp. 580–581). Rathway: Merck.
Xu, X., Philip, S., Stewart, S. P., & Chen, X. (1996). Transport limitation of chlorine disinfection of Pseudomonas aeruginosa entrapped in alginate beads. Biotechnology and Bioengineering, 49, 93–100.
Zezza, N., Pasini, G., Lonbardi, A., Mercenier, A., Spettoli, P., Zamorani, A., et al. (1993). Production of a bacteriocin active on lactate-fermenting clostridia by Lactobacillus lactis sp. lactis immobilized in coated alginate beads. The Journal of Dairy Research, 60, 581–591.
Acknowledgment
The authors gratefully acknowledge the financial assistance of the department by the UGC, New Delhi under its DRS program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wasi, S., Tabrez, S. & Ahmad, M. Suitability of Immobilized Pseudomonas fluorescens SM1 Strain for Remediation of Phenols, Heavy Metals, and Pesticides from Water. Water Air Soil Pollut 220, 89–99 (2011). https://doi.org/10.1007/s11270-010-0737-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-010-0737-x