Abstract
Cystic echinococcosis (CE) is an emergent neglected disease affecting human and animals in Egypt with a wide distribution and incidence. This study aimed to evaluate the use of a polyclonal antibody-based sandwich ELISA in the detection of Echinococcus granulosus antigen in human and camel sera. Hydatid cyst protoscoleces antigen (PsAg) was isolated from hydatid cysts collected from naturally infected camel livers and lungs. PsAg was used for immunization of rabbits to raise IgG polyclonal antibodies (IgG PsAb). IgG PsAb were then precipitated, purified using Protein-A Sepharose gel and labeled with horseradish peroxidase enzyme. We assayed the purity of the IgG PsAb, and the two prepared E. granulosus antigens CPsAg from camel cysts and HPsAg from human cysts by Sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The resulted protein bands of the prepared CPsAg appeared at different molecular weights: 180, 90, 68, 54, 42 and 22 kDa while, HPsAg shared with it in 4 common bands at 68, 54, 42, and 22 kDa. The purified IgG PsAb had been resolved at two bands at 52 kDa and at 32 kDa. Sandwich ELISA were performed for the detection of circulating E. granulosus antigens in sera of human (n = 183) and camels (n = 190). The purified IgG PsAb showed strong reactivity against E. granulosus infected human and camel samples and no cross reactivity neither with free-healthy negative sera nor with others parasitic diseases (Schistosomiasis, Fascioliasis, Toxoplasmosis, Ancylostomiasis for human samples and Fascioliasis, ticks’ infestation, Eimeriosis, Cryptosporidiosis, Nasal myiasis, Toxoplasmosis for camel samples). The sensitivity of the assay was 98.25% (56/57) and 96.9% (31/32) against human and camel samples, respectively. Specificity was 100% in both human and camel samples. Sandwich ELISA detected CE in 33.3% (24/72) and 55.6% (50/90) random human and camel samples, respectively. Indirect ELISA, using CPsAg, was used for detection of antibodies in positive human and camels’ sera and detected 96.5% (55/57) and 93.8% (30/32) of human and camel samples, respectively. In our study, Genomic DNA was extracted from protoscoleces fluid of human liver hydatid cysts to identify the Echinococcus sp. isolate based on NADH dehydrogenase subunit 1 (NAD1) gene by Polymerase Chain Reaction (PCR) and the isolate (GenBank: OP785689.1) were identified as E. granulosus sensu lato genotype. In conclusion, Sandwich ELISA technique was found to be a potent and sensitive assay for detection of hydatid antigen in both human and camel samples.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cystic echinococcosis (CE) is important helminthic zoonotic disease caused by the infection with Echinococcus granulosus belonging to the family Taeniidae (Thompson 2017; Ebrahimipour et al. 2019; Zhang et al. 2023). Fecal materials of E. granulosus-infected dogs and canids (definitive hosts) contaminate the environment spreading infective eggs which are taken by fecal-oral route in humans and animals (intermediate hosts) (King and Fairley 2015) causing significant morbidity and mortality in humans as well as significant economic losses in livestock industry worldwide (Nigo et al. 2022; Cai et al. 2023). In livestock, CE could result in reduced birth rate, low yield and quality of animal meat and wool, delayed growth, destructed viscera, organ condemnation, and even death (Benner et al. 2010; Cai et al. 2023). In humans, economic losses are resulted from increased surgery costs, hospital care, and impaired labor productivity (Singh et al. 2014; Sen et al. 2019). Its prognosis depends on cyst number, stage, and location making control of this disease complex (Ali et al. 2020). The high prevalence of CE in humans and animals is usually observed in temperate regions (Mediterranean regions, North and East Africa, and Central Asia) (Grosso et al. 2012).
Cysts are fluid-filled vesicles with two layers. The outermost laminated layer which is an acellular coat secreted by the inner germinal layer. The inner germinal layer is the cellular proliferative sheet of the hydatid cyst which encloses brood capsules, protoscoleces (Pedrosa et al. 2000), undifferentiated cells, storage cells, and muscle cells (Siles-Lucas et al. 2017). Due to absence of specific clinical signs and symptoms, CE is commonly performed at necropsy in post-mortem (PM) examination in animals (Craig et al. 2015) and by laboratory techniques and radiological imaging in humans (Alli et al. 2011). Ultrasonography, computerized tomography, and serology are useful diagnostic tools for human hydatidosis, and the disease is considered a public health challenge and needs accurate differential diagnosis from any cystic mass/s in the abdomen (Elaadli et al. 2022). Immunodiagnostic laboratory techniques are widely used to confirm CE diagnosis in suspected cases or follow up after surgical and chemical treatments, especially Enzyme Linked Immunosorbent Assay (ELISA) and Indirect hemagglutination test (IHAT) (Zhang et al. 2012).
ELISA is a fast, easy and non-expensive test to perform with high sensitivity and specificity (Hassanain et al. 2021). The main obstacle of antibody tests is that they couldn’t distinguish between past and present infections. Antigen detection techniques might help to avoid this problem (Doiz et al. 2001) and might be useful in the immunodiagnosis of hydatid disease (Toaleb et al. 2023). Circulating hydatid antigens are detected in the serum only during active infection, and their levels decrease gradually after surgical removal of the cyst or successful chemotherapy (Devi and Parija 2003; Sadjjadi et al. 2009; Bauomi et al. 2015). Also, antigen detection assays could help in assessing the efficacy of treatment, especially after surgical removal of hydatid cyst (Bauomi et al. 2015).
Because of the importance of cystic echinococcosis in human and animal health, rapid and precise diagnostic methods are urgently needed (Hassanain et al. 2021). Consequently, the current research aims to evaluate a specific and simple diagnostic efficacy of purified polyclonal antibody IgG raised against E. granulosus protoscoleces antigen (PsAg) for detection circulating E. granulosus antigen in humans and animal’s sera using Sandwich ELISA and compared it with antibody detection method.
Materials and methods
Human patients and study area
This cross-sectional study was carried out during the period from December 2021 to October 2023 on 183 patients who attended in the Gastroenterology Outpatient Clinics, Internal Medicine and Surgery Departments, Al-Azhar Teaching Hospital and Al-Kasr Al-Ainy School of Medicine, Cairo University, Cairo, Egypt. An informed written consents were obtained from patients who participated in this study.
Animals and study area
One hundred ninety blood samples were collected from camels through several visits to main local abattoirs of Giza (Nahia, El-warrak and El-moneb) and Cairo (EL-Basatin abattoir), Egypt, during the period December 2021 to October 2023.
Blood samples
Human blood samples
One hundred eighty-three blood samples were collected from patients who complained about digestive disturbances, fever, and abdominal pain. These blood samples were divided into four groups:
-
I
17 blood samples collected from apparently healthy individuals free from intestinal parasites represented as negative controls.
-
II
57 blood samples represented the positive controls including 53 blood samples from individuals that were diagnosed to have CE in their livers, chest and other organs as proved by sonography, Computed Tomography (CT) and Magnetic Resonance Imaging (MRI), and 4 blood samples collected from individuals that had CE in their livers as proved by sonography, CT, and MRI to have CE with surgical confirmation and removal. Cysts removed during surgery were confirmed to be hydatid cysts with the presence of fertile protoscoleces in the aspirated cysts’ fluid. Sera from these patients were collected before surgery on admission to surgical ward (gold standard positive control).
-
III
72 blood samples were randomly obtained.
-
IV
37 blood samples were collected from individuals that were infected with other parasites [10Schistosoma mansoni (schistosomiasis), 7Fasciola gigantica (fascioliasis), 15Toxoplasma gondii (toxoplasmosis), and 5Ancylostoma duodenale (Ancylostomiasis)].
All blood samples were collected from all cases, sera were separated, aliquoted and kept at − 20 °C until used.
Camel blood samples
One hundred ninety blood samples were collected from camels through several visits to main local abattoirs during veterinary medical examinations:
-
I
32 blood samples from camels that were infected by E. granulosus collected at postmortem inspection (PM; gold standard positive control sera).
-
II
15 blood samples collected from healthy young camels, free of cysts and other parasitic infections as confirmed by PM inspection and fecal examination (gold standard negative control sera).
-
III
53 blood samples from camels that were positive for other parasites (5 infected with F. gigantica (Fascioliasis), 12 blood samples from camels infested with Hyalomma dromedarii ticks, 6 samples from camels with Coccidiosis (Eimeriosis) as proved by fecal examination, 10 samples from camels infected with Cryptosporidium parvum (Cryptosporidiosis) as proved by fecal examination, 13 samples from camels infested with Cephalopina titillator larvae as shown after examination of slaughtered camels’ skulls and 7 samples from camels that were positive to Toxoplasma as proven by ELISA).
-
IV
90 blood samples were randomly obtained.
All blood samples were collected from all cases, sera were separated, aliquoted and kept at − 20 °C until used.
Parasite
Human hydatid cysts
Four human hydatid cysts were recovered from postoperative patients admitted to the Internal Medicine and Surgery Departments, Al-Azhar Teaching Hospital and Al-Kasr Al-Ainy School of Medicine, Cairo University, Cairo, Egypt. Protoscoleces were aspirated from cysts, pooled, and washed with sterile normal saline. In our laboratory, every sample was examined by light microscope (CX41 Olympus Microscope, Olympus Corporation, Japan) and 0.1% eosin (Sigma-Aldrich, Louis, MO, USA) staining to assess the cyst fertility and viability (Daryani et al. 2007). Cyst fluids were centrifuged at 5000 rpm for 30 min and each sample was stored at − 20 °C until being used for the molecular analysis (Piccoli et al. 2013).
Camel hydatid cysts
Thirty-two hydatid cysts (12 large fertile hydatid cysts from infected livers and other 20 large fertile cysts from infected lungs) were collected from slaughtered camels during PM inspection. Hydatid cysts were placed in sterile saline solution and transported to the laboratory in ice box. Cyst contents were aseptically aspirated, centrifuged at 5000 rpm for 30 min, and examined for the presence of protoscoleces. The precipitates of the fertile cysts were stained with 0.1% eosin solution for 10 min and the viability of the protoscoleces was examined under a light microscope (Bahmani and Anisian 2020). Then, protoscoleces were collected from fertile cysts of liver, collected protoscoleces were washed several times in sterile normal saline (Dyab et al. 2017).
DNA extraction
The Genomic DNA was extracted from protoscoleces fluid of four fertile liver human hydatid cysts using the QIAamp DNA mini kit (Cat. No. 51,304, QIAGEN, Germany) according to manufacturer’s protocol. The DNA concentrations were estimated by Q9000 microvolume spectrophotometer (Quawell, USA) and stored at − 20 °C.
Polymerase chain reaction (PCR) and electrophoresis
PCR was performed on the extracted DNA using primers which target 500 bp fragment of NADH dehydrogenase subunit 1 (NAD1) gene (F: 5ʹ- AGA TTC GTA AGG GGC CTA ATA − 3ʹ and R: 5ʹ- ACC ACT AAC TAA TTC ACT TTC − 3ʹ) according to Aboelhadid et al. (2013) using T100 Thermal Cycler (BIO-RAD, Singapore). The PCR products were then visualized by Gel Doc™ XR + Molecular Imager (BIO RAD, California, USA) in agarose gel electrophoresis (1.5%), stained by RedSafe (Intron Biotechnology, Republic of Korea), and measured by a 100 bp ladder (Cat. No. 239,035, QIAGEN, USA). Negative and positive controls were involved in the PCR protocol.
Sequencing and phylogenetic analysis
After PCR, products of the most intense bands on gel electrophoresis were purified by a Gel Extraction Kit (Cat. No. NA006-0100, GeneDirex, Taiwan) according to manufacturer’s instructions. The purified products were sequenced by ABI 3130 Automated Sequencer (Applied Biosystems, USA) using Big Dye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, USA). Obtained sequences were corrected using ChromasPro 1.7 software (Technelysium Pty Ltd., Australia), compared by BLASTn (https://blast.ncbi.nlm.nih.gov/Blast.cgi) with previous sequences available in GenBank and submitted to GenBank. Then, multiple sequence alignments were performed using CLUSTAL W v1.83, in the MegAlign module of the Laser gene software package (DNASTAR, USA). Phylogenetic analysis was performed using MEGA6 software (Tamura et al. 2013).
Preparation of protoscoleces antigens
Human and camels (the used camel isolate was previously identified as GenBank: OQ443068.1; Toaleb et al. 2023) protoscoleces’ antigens (HPsAg and CPsAg) were prepared. Briefly, the hydatid cyst fluid (HCF) was obtained by aseptic puncture from fertile hydatid cysts of camel liver origin, centrifuged at 6000 rpm for 45 min at 4ºC, and the sediments containing protoscoleces were collected in sterile tubes. The protoscoleces were washed three times with Phosphate Buffered Saline (PBS) pH = 7.2, subjected to several freezing and thawing cycles and then sonicated using a 150w ultrasonic disintegrator (10 cycles/12 sec/60 Hz frequency) until no intact protoscoleces were visible microscopically. Protoscoleces mixture was centrifuged at 16,000 rpm for 30 min. The supernatant was collected, aliquoted, and stored at − 20 °C (Carmena et al. 2005). The protein content of the two prepared antigens was estimated by Lowry’s method (Lowry et al. 1951).
Rabbit polyclonal antibody IgG (anti-hydatid IgG)
We used five healthy 3 months aged New Zealand rabbits weighing 2–2.5 Kg obtained from local market. They were daily examined for 2 weeks to ensure they are parasite-free. Rabbit hyperimmune serum was raised against CPsAg as described by Guobadia and Fagbemi (1997) and Fagbemi et al. (1995). Briefly, 40 µg/Kg of CPsAg was mixed with same volume of Freund’s complete adjuvant (Sigma-Aldrich, Cat No. F5881) and subcutaneously injected into each rabbit. After 2 weeks, a booster dose of CPsAg (40 µg/Kg) with an equal volume of Freund’s incomplete adjuvant (Sigma-Aldrich, Cat No. F5506) was injected subcutaneously into each rabbit. Second and third booster doses were injected at the 21st and 28th days, respectively. Blood samples were taken by ear vein bleeding 4 days after last injection and monitored for the production of antibodies IgG to CPsAg by indirect ELISA according to Engvall and Perlmann (1971).
Purification of polyclonal antibody IgG
Anti-hydatid IgG (IgG PsAb) was precipitated using 50% Ammonium sulphate solution according to Wingfield (2016). Then, dialysis IgG PsAb against 0.15 M PBS for 3 days at 4 °C was performed. Polyethylene glycol was used to concentrate the IgG PsAb. Furthermore, purification of IgG PsAb was performed with Affinity chromatography on Protein-A Sepharose gel (Sigma, St. Louis, Missouri, USA) according to Abd El Hafez et al. (2010) and Toaleb et al. (2023). The protein content of IgG PsAb was estimated following the method of Lowry et al. (1951). The reactivity of Anti-hydatid IgG (IgG PsAb) against Echinococcus antigen (PsAg) was measured by indirect ELISA.
Labeling of antibodies (IgG PsAb) by horseradish peroxidase (HRP)
Conjugation of IgG PsAb with HRP was performed as previously described by Avrameas (1969) and Toaleb et al. (2023). In brief, HRP enzyme (10 mg) was mixed with IgG PsAb (5 mg) in 1 mL of 0.1 M phosphate buffer (pH = 6.8). Then they were dialyzed at 4 ◦C overnight against 0.1 M PBS (pH = 6.8). After that, 50 µL diluted glutaraldehyde was added to the mixture with gentle shaking for 3 h at room temperature. Glycine solution (2 M) was added, left for 2 h at room temperature and then dialyzed overnight again. The mixture was centrifuged at 10,000 ×g for 30 min at 4 ◦C and the supernatant was aspirated to sterile tubes and one volume of glycerol was added. The prepared labeled IgG PsAb was stored at − 20 ◦C until use.
Sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE)
Electrophoresis of IgG PsAb, CPsAg and HPsAg was performed in 10% polyacrylamide gels according to method described by Laemmli (1970). Each protein of IgG PsAb, CPsAg and HPsAg was mixed with sample buffer containing 2-mercaptoethanol before loading to the gel, separately. After separation, slab gel was stained with Coomassie Brilliant Blue dye. Relative molecular weights of bands were calculated using prestained protein marker (GeneDirex BLUltra, USA). Molecular weights were analyzed using Bio-Rad Imager (Gel Doc™ XR+, California, USA).
A polyclonal antibody-based sandwich ELISA
Sandwich ELISA was performed following the method of Toaleb et al. (2023). In brief, 4 µg/mL of rabbit IgG PsAb was diluted in coating buffer (dilution 1:25), added into 96-well ELISA plates (100 µL/well), and incubated at 4 ◦C overnight. The plates were washed 3 times by PBS-Tween-20 (0.1 MPBS, pH = 7.4, + 0.05% Tween 20) to remove excess antibodies. Wells were blocked with blocking buffer (1% bovine serum albumin, BSA, 200 µL/well), incubated at 37 ◦C for 2 h, and then washed 3 times with PBS-Tween-20. Triplicates of human and camel sera (diluted 1:100 with PBS; 100 µL/well) were added and the plates were incubated at 37 ◦C for 2 h. Diluted HRP-conjugated IgG PsAb (1:100; 100 µL/well) were added after another washing step, and the plates were incubated at 37 ◦C for 90 min then washed 3 times with washing buffer. The reaction was visualized by adding 100 µl/well of Ortho-phenylenediamine (OPD, Sigma-Aldrich) substrate solution for 20–30 min in the dark at room temperature. Reaction was stopped by 50 µl/well of 0.1M H2SO4. Optical densities (OD) were read at 450 nm with an ELISA microplate reader (Bio-Tek Instruments, Winooski, VT, USA). Positive and negative controls were used at the same plate and all samples and controls were assayed in triplicates. The cut-off point was obtained by mean values of OD of negative control plus three times the standard deviation (SD) according to Jin et al. (2013). Diagnostic sensitivity, specificity, diagnostic efficacy as well as positive and negative predictive values were calculated according to Parikh et al. (2008) and as described by Toaleb et al. (2023).
Indirect ELISA
Indirect ELISA, using CPsAg, was performed for detection of antibodies in human and camels’ sera as described in Hassanain et al. (2021). In Brief, an ELISA plate was coated with CPsAg (4 µg/mL) overnight, washed 3 times with washing buffer and blocked by BSA 1%. Two-fold serial dilutions of the purified IgG CPsAb were added into wells. The plate was washed and the conjugate (Anti-rabbit IgG HRP, Sigma Chemical Co., St. Louis, MO, USA) was used. The OPD substrate (Sigma-Aldrich) was used for color development in the reaction. OD were measured at 450 nm with EL×800UV BioTek microplate reader (Bio-Tek Instruments, Winooski, VT, USA).
Statistical analysis
Statistical analyses were performed by SPSS (version 19.0 for Windows, IBM Corp., USA). Data of CE infection rates, and Sandwich ELISA were analyzed using Chi square test. P < 0.05 was considered statistically significant. The diagnostic accuracy parameters of the test were evaluated by calculating the sensitivity, specificity, area under the curve (AUC), and receiver operating characteristic (ROC) curve, Chi square using SPSS software.
Results
The cysts fertility and viability
Of the total hydatid cysts of camel’s lungs and Livers examined, fertile cysts accounted for all 32 camels’ cysts were 68.75% (22/32 cysts). We found 91.7% (11/12) fertile and 8.3% (1/12) calcified cysts in camel liver hydatid cysts examined. Whereas, in camel lung hydatid cysts, 60% (12/20) were fertile, 10% (2/20) were calcified and 30% (6/20) were sterile lung cysts. Average viability of fertile cyst protoscoleces was obtained (90.9 ± 0.8) in camel liver cysts and (58.3 ± 1.7) in camel lung cysts, with a statistically significant difference (P < 0.05). In human the highest viability of fertile cyst protoscoleces was obtained in cysts removed from human during surgery (in the aspirated cyst fluid) as shown in Table 1.
PCR and phylogenetic analysis
Four DNA samples extracted from protoscoleces collected from human fertile liver cysts provided the expected amplicon size (~ 500 bp) for Echinococcus spp. in the gel electrophoresis after PCR using NAD1 gene. Blast analysis showed the presence of genotype of E. granulosus in the investigated humans (GenBank: OP785689.1). In the phylogenetic tree, the resulted genotype clustered in a well-supported branch (bootstrap value 78) with other E. granulosus references (Fig. 1).
Phylogenetic analysis using the maximum likelihood method based on NAD1 gene for Echinococcus sp. Our obtained isolate is highlighted (red dot). There was a total of 459 positions in the final dataset. Our genotype clustered in a well-supported branch (bootstrap value 78) with other E. granulosus references. The scale bar represents a 5% nucleotide sequence divergence
SDS-Page
We assayed the purity of IgG PsAb, and the two prepared E. granulosus antigens CPsAg and HPsAg by SDS-PAGE. The results showed that the protein bands of prepared CPsAg appeared at different molecular weights: 180, 90, 68, 54, 42 and 22 kDa (Fig. 2A; Lane CPsAg). Whereas HPsAg shared in four common bands with CPsAg at molecular weights: 68, 54, 42, and 22 kDa (Fig. 2A; Lane HPsAg). The purified IgG PsAb had been resolved at two bands; the heavy chain band (H- chain band) was represented at 52 kDa, and at 32 kDa was the light chain band (L-chain band) (Fig. 2B; Lane IgG PsAb).
Electrophoresis profile of protoscoleces hydatid cyst antigens, 10% SDS-PAGE slab gel stained with Coomassie brilliant blue dye. A: Camel protoscoleces hydatid cyst antigens (CPsAg): Lane CPsAg and Human protoscoleces hydatid cyst antigen (HPsAg): Lane HPsAg. B: Purified IgG PsAb after precipitation with 50% ammonium sulfate and purified by Affinity chromatography on protein-A Sepharose gel column: Lane IgG PsAb. Molecular weight standard protein: Lane M
The binding activities of purified IgG PsAb against CPsAg and HPsAg
The success of developing antibodies in rabbits (IgG PsAb) was proved by indirect ELISA in which commercial Anti-rabbit IgG HRP-conjugate was used. The immune sera (IgG PsAb) were collected at high titers against antigens (CPsAg and HPsAg) of the antibodies with maximal OD of 1.8 and 1.2 respectively, at 1/100 dilution of IgG PsAb. The protein content in the crude rabbit serum containing anti-E. granulosus PsAb was 15.5 mg/ml. After purification the protein content of IgG PsAb was 12 mg/ml. As shown in Fig. 3, the purified IgG PsAb checked by two-fold serial dilutions reached to ~ 1: 4096 and still reacted positively with CPsAg (Fig. 3A) and HPsAg (Fig. 3B).
Specificity of purified IgG PsAb in the diagnosis of CE
Comparative evaluation of specificity of the purified IgG PsAb in detection circulating E. granulosus antigen in sera of human and serum of naturally infected camels with E. granulosus or other parasites in comparison to healthy controls for diagnosis of CE was determined by Sandwich ELISA as showed in Fig. 4A&B. The purified IgG PsAb showed strong reactivity against serum samples from human and camels infected with E. granulosus and no cross reactivity neither with free-healthy negative sera nor with others parasitic diseases (Human sera with Schistosomiasis, Fascioliasis, Toxoplasmosis, Ancylostomiasis and camel sera with Fascioliasis, ticks’ infestation, Eimeriosis, Cryptosporidiosis, Nasal myiasis, Toxoplasmosis). The cut-off value was 0.347. The mean OD value of CE infected camels was 1.288 ± 0.186 and it was 1.449 ± 0.172 in infected human sera which were both higher than negative control group (0.273 ± 0.198) and the other parasites groups (0.265 ± 0.025; Fig. 4A&B).
The sensitivity of the assay was calculated to be 98.25% sensitivity against human samples, where the sandwich ELISA detected circulating hydatid antigens in 56 cases out of 57 which were E. granulosus infected with surgical confirmation, ultrasound-proven, and presumptive diagnosis of CE. While 31 camel samples were detected as E. granulosus infected from 32 samples that confirmed by PM examination and the sensitivity of the assay was 96.9%. All the negative controls and positive samples for other parasites in human and camel samples were below the cut-off recording a 100% specificity in both human and camel as shown in Table 2.
Indirect ELISA
Antibody detection by indirect ELISA, using antigen CPsAg, showed that 96.5% of human serum samples (55 cases out of 57) had CE antibodies in their sera while cross reaction was noted in a few non-CE cases (16 negative CE out of 17 cases). A sensitivity of 96.5% and specificity of 94.1% was calculated with diagnostic accuracy 95.95%. While the sensitivity of the assay was 93.8% in camel serum samples (30 out of 32) were detected as E. granulosus infected and specificity was 93.3% (14 free from CE out of 15) with diagnostic accuracy 93.62% in camel sera (Table 3).
Detection of circulating hydatid antigen in random serum of human and camels for the diagnosis of CE by sandwich ELISA
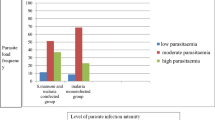
Using Sandwich ELISA, E. granulosus antigen was detected in 24 out of 72 (33.3%) of human samples (Fig. 5). Whereas 50 infected camel samples were detected out of 90 random collected sample with a percentage of 55.6% (Fig. 6).
Discussion
CE is regarded as a significant public health problem in several countries, including Egypt (Hassanain et al. 2021; Hussien et al. 2022). In animals E. granulosus causes a high rate of lungs and liver condemnation in slaughtered domestic animals (Abo-Aziza et al. 2019). CE was recently included in the World Health Organization (WHO) strategy to combat neglected tropical diseases for its eradication or control (WHO 2020, 2021). CE is clinically detectable in humans through imaging techniques such as ultrasonography or radiology (Hussien et al. 2022), and after slaughtering in animals (Toaleb et al. 2023). The primary diagnosis should be confirmed by serological tests based on the detection of antibodies against E. granulosus antigens in serum (Dyab et al. 2017; Hassanain et al. 2021; Darabi et al. 2022). One of obstacles of this method is the difficulty of obtaining enough human Echinococcus spp. cysts from patients for antigen extraction and preparation. Therefore, most of the available serological assays depend on using antigens extracted from animal hydatid cysts to diagnose human CE (Bauomi et al. 2015; Hassanain et al. 2021). Also, commercially available serological kits mainly target HCF, which consists of both the host and parasite, its components vary from one cyst to another, which might lead to different HCF-based tests from one medical center to another (Pagnozzi et al. 2014; Ahn et al. 2015).
In the present study, we investigated the use of a purified anti-hydatid polyclonal antibody IgG (IgG CPsAb) for the detection circulating E. granulosus antigen in naturally infected human and camel sera using sandwich ELISA. We chose E. granulosus protoscoleces antigen which is shown to be a suitable diagnostic antigen candidate with high sensitivity and specificity in ELISA (Shahabinejad et al. 2020). We selected protoscoleces antigen separated from camel hydatid cysts (CPsAg), because the HCF antigen extracted from human hydatid cysts was unsuitable for diagnosis as it contains host protein like IgG which may cross react with specific IgG present in sera of CE infected human (Zhang et al. 2012). However, the immunological characterization of both camel and sheep hydatid fluid and protoscoleces antigens might be helpful for diagnosis of hydatidosis in human (Hassanain et al. 2016). When human and camel samples were surveyed using ELISA, the protoscoleces antigen was more efficient as a diagnostic antigen (Pagnozzi et al. 2016. Hassanain et al. (2021) found that protoscoleces antigen extracted from hydatid cysts of camel was the most potent antigen for diagnosing hydatidosis in humans and camels.
In the present study, most of E. granulosus cysts collected from human and camels were fertile containing large numbers of protoscoleces. The mean viability of the protoscoleces was (100%) in cysts removed from human during surgery and ranged from 90.9% in camels’ liver cysts to 58.3% in camels’ lung cysts. The fertility of the cysts and viability of protoscoleces demonstrate the perfect adaptation of the strain to human (Craig et al. 2007). The high rate of human hepatic infection reported in our study might be attributed to the fact that the liver acts as the primary filter in the human body and the lung is often thought to be the second filter (Xiao et al. 2005). A similar picture of organ affection has been reported by other studies as the most frequent site of hydatid cysts was the liver (50–70%) followed by the lung (20–30%) and less frequently, kidney, heart, bones (Wen et al. 2019; Şahin et al. 2023).
Echinococcus sp. isolate recovered from hydatid cyst protoscoleces of naturally infected humans in this study (Fig. 1; Genbank: OP785689.1) was identical to those of E. granulosus sensu lato genotype G6 detected in humans from Mongolia (GenBank: MH300971.1), camels from Iran (Genbank: HM749618.1 and NC_038227.1) and Mauritania (GenBank: MH300954.1 and MH300953.1) and sheep from Sudan (Genbank: MH300941.1 and MH300947.1). These results agreed with other previous reports, which assume that G6 is common in human and animals and is widespread in camel-raising countries (Romig et al. 2017). G6 was assumed to be the dominant genotype among Echinococcus sp. human isolates with a frequency of 58.1% (Abdelbaset et al. 2021). Also, hygienic and husbandry conditions of rearing animals, might increase transmission of Echinococcus spp. to humans (Casulli et al. 2022).
In the present study, CPsAg appeared at different molecular weights that were closely similar to bands (205, 71, and 68, 52, 41, and 22 kDa; Fig. 2) found in protoscoleces antigen previously reported by Hassanain et al. (2021). The purification procedures performed in our study were satisfactory and the purified IgG PsAb was characterized by two bands indicating that, the purified IgG PsAb appeared free from other proteins. This result agreed with El Deeb et al. (2017) who reported that the purity of anti-E. granulosus IgG PAb, assayed by 12.5% SDS-PAGE under reducing conditions, was represented by H- and L-chain bands at 50 and 31 kDa, respectively. In another study, Toaleb et al. (2023) investigated that the purified IgG germinal layer antibody was represented by four bands at 77, 65, 55 and 25 kDa. This difference might be due to the different method of preparation of crude antigens, different percentage of reagents of SDS-PAGE, rabbit immunization technique, and the method of polyclonal antibody IgG (IgG PsAb) preparation. In the present study, the purified IgG PsAb gave a strong reactivity to CPsAg and HPsAg as determined by indirect ELISA. Also, the purified IgG PsAb detected the circulating E. granulosus antigens in CE infected human sera and CE infected camel sera, when used as a primary capture to coat ELISA plates. Then IgG PsAb labeled with HRP-conjugate was captured again when sandwich ELISA was adopted using a pair of IgG PsAb against CPsAg, HPsAg antigens, anti- E. granulosus IgG PsAb and peroxidase-conjugated IgG polyclonal antibodies. After detection of E. granulosus circulating antigens by sandwich ELISA in human serum samples, all values equal to or above cut off values (0.347) were considered positive and 56 cases out of 57, which were infected by E. granulosus, gave positive results with 98.25% sensitivity and 96.9% (31 as E. granulosus infected from 32 positive samples) in infected camel serum samples. Specificity was 100% in healthy human and camel control serum samples with a diagnostic accuracy 98.65% of CE detection in human sera 97.87% in camel sera. These results showed higher sensitivity and specificity than other previously study in which the sensitivity was 90.48% and specificity was 91.3% when using sandwich ELISA with purified polyclonal antibody (PAb) raised against E. granulosus Cathepsin B for detection E. granulosus circulating antigen in human serum samples (El Deeb et al. 2017). In addition, it is higher than PAb conjugated with gold nanoparticles for detection E. granulosus antigen using nano-gold dot-ELISA, which recorded a sensitivity of 94.4% and a specificity of 90% with an accuracy value 92.9% of CE detection in human, camel, and sheep sera (Rashed et al. 2019). Moreover, in the present study, the in-house sandwich ELISA used to detect circulating CE antigen in human and camels’ sera was more sensitive and specific than other previous studies which used the purified anti-hydatid cyst fluid IgG using protein-A affinity chromatography which gave a sensitivity of 25.7% and a specificity of 98% (Sadjjadi et al. 2009). Furthermore, our results were more sensitive than the latex agglutination test (LAT) in the detection of circulating hydatid antigen in human serum which recorded a sensitivity of 72% and a specificity of 98% (Devi and Parija 2003). On other hand, when we compared our in-house sandwich ELISA based on polyclonal antibody IgG PsAb and the Indirect ELISA based on CPsAg (Fig. 3), we found that the indirect ELISA gave fewer positive samples in CE infected human and camels serum samples with lower sensitivity and specificity. These results might be related to lower antibody titer, which cannot be easily detected, in cerebral, ocular, and calcified cysts in human or some CE patients especially in old persons and infants (Ravinder et al. 1997). Also, the long persistence of anti-E. granulosus antibodies after surgical removal of the cysts results in unreliable diagnosis of relapse in patients (Todorov and Stojanov 1979).
The double antibody sandwich ELISA, used in our study, is a common method for measuring the presence and/or concentration of circulating parasite antigens (Zhang et al. 2012). Our results proved that the standard double antibody IgG PsAb sandwich ELISA could be a promising method for the detection of E. granulosus circulating antigens with high sensitivity and specificity. Indirect ELISA test conducted by Darabi et al. (2022) for detection of specific anti-hydatid cyst antibodies in human CE recorded a sensitivity of 95.2% and a and specificity of 96.8%, respectively, and also, sensitivity and specificity were higher than the of the commercial ELISA test which were recorded 96.8% (Darabi et al. 2022). Circulating hydatid antigens could be detected in serum only during active infection, and their levels decrease after surgical removal of hydatid cyst or successful chemotherapy (Devi and Parija 2003). In our study, sandwich ELISA used in the detection of circulating hydatid antigens in random collected samples from humans and camels giving infection rates of 33.3 and 55.6%, respectively (Figs. 5 and 6). Previous studies reported that hydatid cyst antigen could be detected in sera of 33–85% of hydatidosis patients (Gottstein 1984; Ravinder et al. 1997). Toaleb et al. (2023) found that infection rate was 48.7% in camels. High infection rates found in camels could explain the assumption that camels play a potential role in the maintaining of transmission cycle of hydatidosis (Gareh et al. 2021).
Conclusion
The combination of clinical, radiological, and serological diagnosis is recommended in the diagnosis of hydatidosis. In our study, sandwich ELISA using IgG PsAb could offer a highly accurate, easy and low-cost assay for the diagnosis of CE in human and animals. In addition, the detection of antigen rather than antibodies could be a more reliable method for evaluating the status of infection which could be used for following up after medical or surgical treatment in humans and monitoring the efficacy of treatment in animals.
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- CE:
-
Cystic Echinococcosis
- AUC:
-
Area Under Curve
- CPsAg:
-
Camel Protoscoleces Antigen
- CT:
-
Computed Tomography
- E :
-
granulosus Echinococcus granulosus
- ELISA:
-
Enzyme Linked Immunosorbent Assay
- HCF:
-
Hydatid Cyst Fluid
- HPsAg:
-
Human Protoscoleces Antigen
- IgG:
-
PsAb IgG Polyclonal Protoscoleces Antibodies
- IHAT:
-
Indirect Hemagglutination Test
- LAT:
-
Latex Agglutination Test
- MRI:
-
Magnetic Resonance Imaging
- NAD1:
-
NADH Dehydrogenase Subunit 1 gene
- OD:
-
Optical Density
- OPD:
-
Ortho-phenylenediamine
- PAb:
-
Polyclonal Antibody
- PBS:
-
Phosphate Buffered Saline
- PCR:
-
Polymerase Chain Reaction
- PM:
-
Post-Mortem
- PsAg:
-
Protoscoleces Antigen
- ROC:
-
Receiver Operating Characteristic
- SD:
-
Standard Deviation
- SDS-PAGE:
-
Sodium Dodecyl-Sulfate Polyacrylamide Gel Electrophoresis
- WHO:
-
World Health Organization
References
Abd El Hafez SM, Anwar AM, Ibrahim AM, Mahmoud MB, Hassan HM (2010) Preparation of fluoresce isothiocyanate conjugated IgG (FITC) anti-camel and anti-buffalo. Nat Sci J 8:342–347
Abdelbaset AE, Yagi K, Nonaka N, Nakao R (2021) Cystic echinococcosis in humans and animals in Egypt: an epidemiological overview. Curr Res Parasitol Vector Borne Dis 1:100061
Abo-Aziza FAM, Oda SS, Aboelsoued D, Farag TK, Almuzaini AM (2019) Variabilities of hydatidosis in domestic animals slaughtered at Cairo and Giza abattoirs, Egypt. Vet World 12(7):998–1007
Aboelhadid SM, El-Dakhly KM, Yanai T, Fukushi H, Hassanin KM (2013) Molecular characterization of Echinococcus granulosus in Egyptian donkeys. Vet Parasitol 193:292–296
Ahn CS, Han X, Bae YA, Ma X, Kim JT, Cai H, Yang HJ, Kang I, Wang H, Kong Y (2015) Alteration of immunoproteome profile of Echinococcus granulosus hydatid fluid with progression of cystic echinococcosis. Parasite Vectors 8:10
Ali R, Khan S, Khan M, Adnan M, Ali I, Khan TA, Haleem S, Rooman M, Norin S, Khan SN (2020) A systematic review of medicinal plants used against Echinococcus Granulosus. PLoS ONE 15(10):e0240456
Alli OA, Ogbolu OD, Alaka OO (2011) Direct molecular detection of Mycobacterium tuberculosis complex from clinical samples - an adjunct to cultural method of laboratory diagnosis of tuberculosis. N Am J Med Sci 3(6):281–288
Avrameas S (1969) Coupling of enzymes to proteins with glutaraldehyde. Use of the conjugates for the detection of antigens and antibodies. Immunochem J 5:43–52
Bahmani O, Anisian A (2020) Investigation of the fertility prevalence of hydatid cysts and protoscoleces viability in livestock slaughtered in Ilam industrial slaughterhouse, Ilam Province, Iran during 2016. Egy J Vet Sci 51(3):399–403
Bauomi IR, El-Amir AM, Fahmy AM, Zalat RS, Diab TM (2015) Evaluation of purified 27.5 kDa protoscolex antigen-based ELISA for the detection of circulating antigens and antibodies in sheep and human hydatidosis. J Helminthology 89(5):577–583
Benner C, Carabin H, Serrano LPS, Budke CM, Carmena D (2010) Analysis of the economic impact of cystic echinococcosis in Spain. Bull World Health Organ 88:49–57
Cai J, Yang K, Chen Q, Zhao Q, Li J, Wang S, Yang L, Liu Y (2023) The impact of echinococcosis interventions on economic outcomes in Qinghai Province of China: evidence from county-level panel data. Front Vet Sci 10:1068259
Carmena D, Martínez J, Benito A, Guisantes JA (2005) Shared and non-shared antigens from three different extracts of the metacestode of Echinococcus Granulosus. Mem Inst Oswaldo Cruz 100(8):861–867
Casulli A, Massolo A, Saarma U, Umhang G, Santolamazza F, Santoro A (2022) Species and genotypes belonging to Echinococcus Granulosus Sensu Lato complex causing human cystic echinococcosis in Europe (2000–2021): a systematic review. Parasit Vectors 15(1):109
Craig PS, McManus DP, Lightowlers MW, Chabalgoity JA, Garcia HH, Gavidia CM, Gilman RH, Gonzalez AE, Lorca M, Naquira C, Nieto A, Schantz PM (2007) Prevention and control of cystic echinococcosis. Lancet Infect Dis 7:385–394
Craig P, Mastin A, van Kesteren F, Boufana B (2015) Echinococcus granulosus: epidemiology and state-of-the-art of diagnostics in animals. Vet Parasitol 213(3–4):132–148
Darabi E, Motevaseli E, Mohebali M, Rokni MB, Khorramizadeh MR, Gavidia CM, Gilman RH, Gonzalez AE, Lorca M, Naquira C, Nieto A, Schantz PM (2022) Evaluation of a novel Echinococcus granulosus recombinant fusion B-EpC1 antigen for the diagnosis of human cystic echinococcosis using indirect ELISA in comparison with a commercial diagnostic ELISA kit. Exp Parasitol 240:108339
Daryani A, Alaei R, Arab R, Sharif M, Dehghan MH, Ziaei H (2007) The prevalence, intensity and viability of hydatid cysts in slaughtered animals in the Ardabil province of Northwest Iran. J Helminthol 81(1):13–17
Devi CS, Parija SC (2003) A new serum hydatid antigen detection test for diagnosis of cystic echinococcosis. Am J Trop Med Hyg 69:525–528
Doiz O, Benito R, Sbihi Y, Osuna A, Clavel A, Gómez-Lus R (2001) Western blot applied to the diagnosis and post-treatment monitoring of human hydatidosis. Diag Microbiol Infect Dis 41:139–142
Dyab A, Mohamed G, Abdella O (2017) Seroprevalence of hydatidosis in camels of Assuit Province, Egypt. Madridge J Vaccines 1(1):5–8
Ebrahimipour M, Rezaeian S, Shirzadi MR, Barati M (2019) Prevalence and risk factors associated with human cystic echinococcosis in Iran. J Parasit Dis 43(3):385–392
El Deeb S, Aly I, Mahna N, Faried A, Zalat R, Younis M (2017) Purification and characterization of Echinococcus granulosus cathepsin-B protein and evaluation of its role as a diagnostic marker. Glob Vet 18(2):137–145
Elaadli H, El Adly HM, Shaapan RM, Bessat M (2022) An uncommon primary Splenic Hydatid Cyst in Human: a Case Report Study. Egypt J Hosp Med 89(1):5493–5497
Engvall E, Perlmann P (1971) Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 8:871–874
Fagbemi BO, Obarisiaghon IO, Mbuh JV (1995) Detection of circulating antigen in sera of Fasciola gigantica infected cattle with antibodies reactive with a Fasciola-specific 88 kDa antigen. Vet Parasitol 58(3):235–246
Gareh A, Saleh AA, Moustafa SM, Tahoun A, Baty RS, Khalifa RMA, Dyab AK, Yones DA, Arafa MI, Abdelaziz AR, El-Gohary FA, Elmahallawy EK (2021) Epidemiological, morphometric, and molecular investigation of cystic echinococcosis in camel and cattle from Upper Egypt: current status and zoonotic implications. Front Vet Sci 8:750640
Gottstein B (1984) An immunoassay for the detection of circulating antigens in human echinococcosis. Am J Trop Med Hyg 33:1185–1191
Grosso G, Gruttadauria S, Biondi A, Marventano S, Mistretta A (2012) Worldwide epidemiology of liver hydatidosis including the Mediterranean area. World J Gastroenterol 18(13):1425–1437
Guobadia EE, Fagbemi BO (1997) The isolation of Fasciola gigantica-specific antigens and their use in the serodiagnosis of fascioliasis in sheep by the detection of circulating antigens. Vet Parasitol 68(3):269–282
Hassanain MA, Shaapan RM, Khalil FAM (2016) Sero-epidemiological value of some hydatid cyst antigen in diagnosis of human cystic echinococcosis. J Parasit Dis 40:52–56
Hassanain MA, Toaleb NI, Shaapan RM, Hassanain NA, Maher A, Yousif AB (2021) Immunological detection of human and camel cystic echinococcosis using different antigens of hydatid cyst fluid, protoscoleces, and germinal layers. Vet World 14(1):270–275
Hussien SM, Ibrahim EA, Al Saadawy AS, Salama B (2022) Seroprevalence and epidemiological overview of human cystic echinococcosis in Egypt. Int J Early Child 14(03):2022
Jin Y, Anvarov K, Khajibaev A, Hong S, Hong S (2013) Serodiagnosis of echinococcosis by ELISA using cystic fluid from Uzbekistan sheep. Korean J Parasitol 51:313–317
King CH, Fairley JK (2015) Tapeworms (Cestodes) In: Bennett JE, Dolin R, Blaser MJ, editors. Principles and Practice of Infectious Diseases. 8th Ed. Philadelphia: 2015. pp. 3227–3236
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. Biol Chem J 193:265–275
Nigo KLS, John BT, Lobojo DL, Lita EP, Osman AY, Shuaib YA (2022) Prevalence and financial losses of cystic echinococcosis in slaughtered goats at Gumbo Slab in Juba County, South Sudan. Parasitologia 2:54–62
Pagnozzi D, Biosa G, Addis MF, Mastrandrea S, Masala G, Uzzau S (2014) An easy and efficient method for native and immunoreactive Echinococcus granulosus antigen 5 enrichment from hydatid cyst fluid. PLoS ONE 9(8):e104962
Pagnozzi D, Addis MF, Biosa G, Roggio AM, Tedde V, Mariconti M, Tamarozzi F, Meroni V, Masu G, Masala G, Brunetti E, Uzzau S (2016) Diagnostic accuracy of antigen 5-based ELISAs for human cystic echinococcosis. PLoS Neg Trop Dis 10(3):e0004585
Parikh R, Mathai A, Parikh S, Chandra Sekhar G, Thomas R (2008) Understanding and using sensitivity, specificity and predictive values. Indian J Ophthalmol 56(1):45–50
Pedrosa I, Saíz A, Arrazola J, Ferreirós J, Pedrosa CS (2000) Hydatid disease: radiologic and pathologic features and complications. Radiographics 20(3):795–817
Piccoli L, Bazzocchi C, Brunetti E, Mihailescu P, Bandi C, Mastalier B, Cordos I, Beuran M, Popa LG, Meroni V, Genco F, Cretu C (2013) Molecular characterization of Echinococcus granulosus in south-eastern Romania: evidence of G1-G3 and G6-G10 complexes in humans. Clin Microbiol Infect 19(6):578–582
Rashed S, Nasr M, Shalash I, Ali N, Kishik S, El-Ghanam AS (2019) Echinococcus Granulosus protoscolex antigen used in serodiagnosis of hydatidosis by nano-gold dot-ELISA. Parasitol United J 12:110–115
Ravinder PT, Parija SC, Rao KS (1997) Evaluation of human hydatid disease before and after surgery and chemotherapy by demonstration of hydatid antigens and antibodies in serum. J Med Microbiol 47:859–864
Romig T, Deplazes P, Jenkins D, Giraudoux P, Massolo A, Craig PS, Wassermann M, Takahashi K, de la Rue M (2017) Ecology and life cycle patterns of Echinococcus species. Adv Parasitol 95:213–314
Sadjjadi SM, Sedaghat F, Hosseini SV, Sarkari B (2009) Serum antigen and antibody detection in echinococcosis: application in serodiagnosis of human hydatidosis. Korean J Parasitol 47(2):153–157
Şahin E, Dalda Y, Dirican A (2023) Giant hydatid cyst of the liver: a case report with literature review. Egypt Liver J 13:28
Sen P, Demirdal T, Nemli SA (2019) Evaluation of clinical, diagnostic and treatment aspects in hydatid disease: analysis of an 8-year experience. Afr Health Sci 19(3):2431–2438
Shahabinejad P, Shahriarirad R, Omidian M, Ghorbani F, Barazesh A, Sarkari B (2020) Diagnostic performance of Echinococcus Granulosus protoscolices antigens in the serodiagnosis of human cystic echinococcosis. J Immunoass Immunochem 41(5):833–840
Siles-Lucas M, Casulli A, Conraths FJ, Müller N (2017) Laboratory diagnosis of Echinococcus spp. in human patients and infected animals. Adv Parasitol 96:159–257
Singh BB, Dhand NK, Ghatak S, Gill JPS (2014) Economic losses due to cystic echinococcosis in India: need for urgent action to control the disease. Prevent Vet Med 113:1–12
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Thompson RCA (2017) Biology and systematics of Echinococcus. Adv Parasitol 95:65–109
Toaleb NI, Aboelsoued D, Abdel Megeed KN, Hekal SHA (2023) A Novel designed sandwich ELISA for the detection of Echinococcus granulosus antigen in camels for diagnosis of cystic echinococcosis. Trop Med Infect Dis 8(8):400
Todorov T, Stojanov G (1979) Circulating antibodies in human echinococcosis before and after surgical treatment. Bull World Health Org 57:751–758
Wen H, Vuitton L, Tuxun T, Li J, Vuitton DA, Zhang W, McManus DP (2019) Echinococcosis: advances in the 21st Century. Clin Microbiol Rev 32(2):e00075–e00018
WHO (2020) Neglected Tropical Diseases (NTDs). https://www.Who.Int/Neglected_diseases/Diseases/En/
WHO (2021) Echinococcosis. World Health Organization, Geneva. https://www.who.int/news-room/fact-sheets/detail/echinococcosis
Wingfield PT (ed) (2016) Protein precipitation using ammonium sulfate. Current Protocols in Protein Science 84(1):A-3F
Xiao N, Qiu J, Nakao M, Li T, Yang W, Chen X, Schantz PM, Craig PS, Ito A (2005) Echinococcus shiquicus n. sp., a taeniid cestode from tibetan fox and plateau pika in China. Int J Parasitol 35:693–701
Zhang W, Li J, Lin R, Wen H, McManus DP (2012) Immunology and serological diagnosis of echinococcosis, serological diagnosis of certain human, animal and plant diseases. Dr. Moslih Al Moslih (Ed.), InTech, pp 8–150
Zhang X, Fu Y, Ma Y, Guo Z, Shen X, Li Z, Jiunai G, Wang X, Maji W, Duo H (2023) Brief report prevalence of Echinococcus species in wild foxes in parts of Qinghai Province, China. Vet Res Commun 47(2):947–952
Acknowledgements
The Sandwich ELISA method used in this study is an application of the previously prepared in-house Sandwich ELISA diagnostic kit, Immunology and Parasitology Laboratory, National Research Centre, Dokki, Giza, Egypt.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Ahmed Maher, Nagwa I. Toaleb, Dina Aboelsoued, Marwa B. Salman, and Zaky S. The first draft of the manuscript was written by Nagwa I. Toaleb and Dina Aboelsoued and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
The study was approved by the institutional guidelines of the National Research Centre’s Animal Research Committee under protocol number: 18/234.
Informed consent
Statement not applicable.
Conflict of interest
The authors have no relevant financial or non-financial interest to disclose.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maher, A., Toaleb, N.I., Shaapan, R.M. et al. Human and camel cystic echinococcosis – a polyclonal antibody-based sandwich ELISA for its serodiagnosis with molecular identification. Vet Res Commun (2024). https://doi.org/10.1007/s11259-024-10375-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11259-024-10375-3