Abstract
Purpose
The objectives of this study were to test whether elevated free fatty acids (FFA) from visceral fat accumulation is related to increased urinary albumin excretion and whether fenofibrate has renal protective effects by regulating vascular endothelial growth factor-nitric oxide (VEGF-NO) axis in rats with diet-induced obesity.
Methods
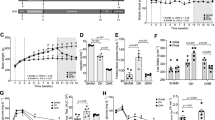
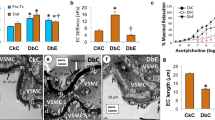
Wistar rats were randomly divided into groups fed a normal diet, a high-fat diet, and a high-fat diet plus fenofibrate. Blood and urine samples were collected. Endothelial function was determined by measuring endothelium-dependent vasodilatation (EDV) of the aorta. Renal tissues were collected for CD31 immunohistochemistry. Glomerular NO and VEGF expression were measured by Griess reaction and Western blot, respectively.
Results
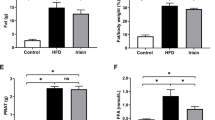
At the end of 24 weeks, plasma FFA and triglyceride levels significantly increased in the obese rats. Fenofibrate intervention decreased serum FFA and triglyceride levels by 43.4 and 48 %, respectively, accompanied by a reduced visceral fat index. Urinary albumin/creatinine ratio increased in obese rats, which decreased 62.6 % after fenofibrate intervention. Severe EDV impairment was observed in obese rats; this was partially improved by fenofibrate. CD31 expression in glomeruli increased in obese rats, indicating increased endothelial cell proliferation. Obese rats showed increased glomerular VEGF expression and reduced NO levels. This uncoupling of VEGF-NO axis was partially improved by fenofibrate.
Conclusion
Elevated circulating FFA level may cause increased microalbuminuria in obese rats due to impairment of EDV; increased microalbuminuria can be improved by fenofibrate intervention. The mechanism may be related to FFA-induced uncoupling of VEGF-NO axis and endothelial dysfunction.





Similar content being viewed by others
References
Dey A, Williams RS, Pollock DM, Stepp DW, Newman JW, Hammock BD et al (2004) Altered kidney CYP2C and cyclooxygenase-2 levels are associated with obesity-related albuminuria. Obes Res 12(8):1278–1289
Kawar B, Bello AK, El NAM (2009) High prevalence of microalbuminuria in the overweight and obese population: data from a UK population screening programme. Nephron Clin Pract 112(3):c205–c212
Klausen KP, Parving HH, Scharling H, Jensen JS (2009) Microalbuminuria and obesity: impact on cardiovascular disease and mortality. Clin Endocrinol (Oxf) 71(1):40–45
Tebbe U, Bramlage P, Thoenes M, Paar WD, Danchin N, Volpe M et al (2009) Prevalence of microalbuminuria and its associated cardiovascular risk: German and Swiss results of the recent global i-SEARCH survey. Swiss Med Wkly 139(33–34):473–480
Kalaitzidis RG, Siamopoulos KC (2011) The role of obesity in kidney disease: recent findings and potential mechanisms. Int Urol Nephrol 43(3):771–784
de Jong PE, Verhave JC, Pinto-Sietsma SJ, Hillege HL (2002) Obesity and target organ damage: the kidney. Int J Obes Relat Metab Disord 26(Suppl 4):S21–S24
Tamba S, Nakatsuji H, Kishida K, Noguchi M, Ogawa T, Okauchi Y et al (2010) Relationship between visceral fat accumulation and urinary albumin-creatinine ratio in middle-aged Japanese men. Atherosclerosis 211(2):601–605
Foster MC, Hwang SJ, Massaro JM, Hoffmann U, Deboer IH, Robins SJ et al (2011) Association of subcutaneous and visceral adiposity with albuminuria: the framingham heart study. Obesity (Silver Spring) 19(6):1284–1289
Kume S, Uzu T, Araki S, Sugimoto T, Isshiki K, Chin-Kanasaki M et al (2007) Role of altered renal lipid metabolism in the development of renal injury induced by a high-fat diet. J Am Soc Nephrol 18(10):2715–2723
Deji N, Kume S, Araki S, Soumura M, Sugimoto T, Isshiki K et al (2009) Structural and functional changes in the kidneys of high-fat diet-induced obese mice. Am J Physiol Renal Physiol 296(1):F118–F126
Stehouwer CD, Smulders YM (2006) Microalbuminuria and risk for cardiovascular disease: analysis of potential mechanisms. J Am Soc Nephrol 17(8):2106–2111
Nakagawa T (2007) Uncoupling of the VEGF-endothelial nitric oxide axis in diabetic nephropathy: an explanation for the paradoxical effects of VEGF in renal disease. Am J Physiol Renal Physiol 292(6):F1665–F1672
Nakagawa T, Sato W, Sautin YY, Glushakova O, Croker B, Atkinson MA et al (2006) Uncoupling of vascular endothelial growth factor with nitric oxide as a mechanism for diabetic vasculopathy. J Am Soc Nephrol 17(3):736–745
Li H, Li H, Bao Y, Zhang X, Yu Y (2010) Free fatty acids induce endothelial dysfunction and activate protein kinase C and nuclear factor-kappaB pathway in rat aorta. Int J Cardiol 152(2):218–224
Srivastava RA, Jahagirdar R, Azhar S, Sharma S, Bisgaier CL (2006) Peroxisome proliferator-activated receptor-alpha selective ligand reduces adiposity, improves insulin sensitivity and inhibits atherosclerosis in LDL receptor-deficient mice. Mol Cell Biochem 285(1–2):35–50
Deng G, Long Y, Yu YR, Li MR (2010) Adiponectin directly improves endothelial dysfunction in obese rats through the AMPK-eNOS Pathway. Int J Obes (Lond) 34(1):165–171
Lane PH, Steffes MW, Mauer SM (1992) Estimation of glomerular volume: a comparison of four methods. Kidney Int 41(4):1085–1089
Kono T, Saito M, Kinoshita Y, Satoh I, Shinbori C, Satoh K (2006) Real-time monitoring of nitric oxide and blood flow during ischemia-reperfusion in the rat testis. Mol Cell Biochem 286(1–2):139–145
Asaba K, Tojo A, Onozato ML, Goto A, Quinn MT, Fujita T et al (2005) Effects of NADPH oxidase inhibitor in diabetic nephropathy. Kidney Int 67(5):1890–1898
Kambham N, Markowitz GS, Valeri AM, Lin J, D’Agati VD (2001) Obesity-related glomerulopathy: an emerging epidemic. Kidney Int 59(4):1498–1509
Henegar JR, Bigler SA, Henegar LK, Tyagi SC, Hall JE (2001) Functional and structural changes in the kidney in the early stages of obesity. J Am Soc Nephrol 12(6):1211–1217
Pinto-Sietsma SJ, Navis G, Janssen WM, de Zeeuw D, Gans RO, de Jong PE (2003) A central body fat distribution is related to renal function impairment, even in lean subjects. Am J Kidney Dis 41(4):733–741
Stehouwer CD, Gall MA, Twisk JW, Knudsen E, Emeis JJ, Parving HH (2002) Increased urinary albumin excretion, endothelial dysfunction, and chronic low-grade inflammation in type 2 diabetes: progressive, interrelated, and independently associated with risk of death. Diabetes 51(4):1157–1165
Deen WM (2004) What determines glomerular capillary permeability. J Clin Invest 114(10):1412–1414
Feliers D, Chen X, Akis N, Choudhury GG, Madaio M, Kasinath BS (2005) VEGF regulation of endothelial nitric oxide synthase in glomerular endothelial cells. Kidney Int 68(4):1648–1659
Satoh M, Fujimoto S, Arakawa S, Yada T, Namikoshi T, Haruna Y et al (2008) Angiotensin II type 1 receptor blocker ameliorates uncoupled endothelial nitric oxide synthase in rats with experimental diabetic nephropathy. Nephrol Dial Transplant 23(12):3806–3813
Davis TM, Ting R, Best JD, Donoghoe MW, Drury PL, Sullivan DR et al (2011) Effects of fenofibrate on renal function in patients with type 2 diabetes mellitus: the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) Study. Diabetologia 54(2):280–290
Tanaka Y, Kume S, Araki S, Isshiki K, Chin-Kanasaki M, Sakaguchi M et al (2011) Fenofibrate, a PPARalpha agonist, has renoprotective effects in mice by enhancing renal lipolysis. Kidney Int 79(8):871–882
Goetze S, Eilers F, Bungenstock A, Kintscher U, Stawowy P, Blaschke F et al (2002) PPAR activators inhibit endothelial cell migration by targeting Akt. Biochem Biophys Res Commun 293(5):1431–1437
Olukman M, Sezer ED, Ulker S, Sozmen EY, Cinar GM (2010) Fenofibrate treatment enhances antioxidant status and attenuates endothelial dysfunction in streptozotocin-induced diabetic rats. Exp Diabetes Res 2010:828531
Acknowledgments
This work was supported by National Natural Science Foundation of China (Grants no. 81070675).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, X., Yu, Y. & Han, L. High FFA levels related to microalbuminuria and uncoupling of VEGF-NO axis in obese rats. Int Urol Nephrol 45, 1197–1207 (2013). https://doi.org/10.1007/s11255-013-0428-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-013-0428-9