Abstract
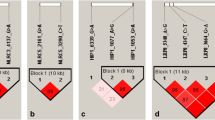
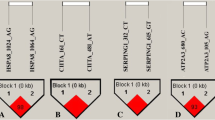
In this study, the association between PAPPA2 coding variants and gastrointestinal (GI) nematode fecal egg count (FEC) score in adult Turkish sheep was investigated. For this purpose, the FEC score was determined in adult sheep from six breeds: Karacabey Merino (n = 137), Kivircik (n = 116), Cine capari (n = 109), Karakacan (n = 102), Imroz (n = 73), and Chios (n = 50). Sheep were classified as shedders or non-shedders within breeds and flocks. The first group was the fecal egg shedders (> 50 per gram of feces), and the second group was the no fecal egg shedders (≤ 50 per gram of feces). The exon 1, exon 2, exon 5, exon 7, and a part of 5′UTR of the ovine PAPPA2 gene were genotyped by Sanger sequencing of these two groups. Fourteen synonymous and three non-synonymous single-nucleotide polymorphisms (SNPs) were found. The non-synonymous SNPs, D109N, D391H, and L409R variants, are reported for the first time. Two haplotype blocks were constructed on exon 2 and exon 7. The specific haplotype, C391G424G449T473C515A542 on the exon 2 that carries the 391H variant, was tested against four other common haplotypes. Our results indicate that C391G424G449T473C515A542 haplotype was significantly associated with fecal egg shedding status in adult Turkish sheep (p-value, 0.044).



Similar content being viewed by others
Data availability
The data that support the findings of this study are available upon reasonable request from the corresponding author.
All animal procedures in the study were reviewed and approved by the ethics committee of Sheep Breeding and Research Institute (approval number: 1282412), and the authors complied with the ARRIVE guidelines.
References
Abebe R, Gebreyohannes M, Mekuria S, Abunna F, Regassa A (2010) Gastrointestinal nematode infections in small ruminants under the traditional husbandry system during the dry season in southern Ethiopia. Tropical Animal Health and Production 42:1111–1117. https://doi.org/10.1007/s11250-010-9532-3
Atlija M, Arranz J-J, Martinez-Valladares M, Gutiérrez-Gil B (2016) Detection and replication of QTL underlying resistance to gastrointestinal nematodes in adult sheep using the ovine 50K SNP array. Genetics Selection Evolution 48:4. https://doi.org/10.1186/s12711-016-0182-4
Axford RFE, Bishop SC, Nicholas FW, Owen JB (2000) Breeding For Disease Resistance In Farm Animals, 2nd Edition. CABI Publishing
Barger IA (1989) Genetic resistance of hosts and its influence on epidemiology. Veterinary Parasitology 32:21–35. https://doi.org/10.1016/0304-4017(89)90153-2
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265. https://doi.org/10.1093/bioinformatics/bth457
Benavides MV, Sonstegard TS, Kemp S, Mugambi JM, Gibson JP, Baker RL, Hanotte O, Marshall K, Van Tassell C (2015) Identification of novel loci associated with gastrointestinal parasite resistance in a Red Maasai x Dorper backcross population. PloS one 10:e0122797. https://doi.org/10.1371/journal.pone.0122797
Borgsteede FHM, Pekelder JJ, Dercksen DP, Sol J, Vellema P, Gaasenbeek CPH, van der Linden JN (1997) A survey of anthelmintic resistance in nematodes of sheep in the Netherlands. Veterinary Quarterly. https://doi.org/10.1080/01652176.1997.9694765
Chartier C, Pors I, Hubert J, Rocheteau D, Benoit C, Bernard N (1998) Prevalence of anthelmintic resistant nematodes in sheep and goats in Western France. Small Ruminant Research. https://doi.org/10.1016/s0921-4488(97)00116-8
Coles GC (1987) World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Bulletin of the Japan Society of Precision Engineering 21:67–69. https://doi.org/10.1016/0304-4017(92)90141-U
Conover CA (2012) Key questions and answers about pregnancy-associated plasma protein-A. Trends in Endocrinology and Metabolism 23:242–249. https://doi.org/10.1016/j.tem.2012.02.008
Gallidis E, Angelopoulou K, Papadopoulos E (2012) First identification of benzimidazole resistant Haemonchus contortus in sheep in Greece. Small Ruminant Research 106:27–29. https://doi.org/10.1016/j.smallrumres.2012.04.016
González-Sánchez ME, Cuquerella M, Alunda JM (2018) Vaccination of lambs against Haemonchus contortus with the recombinant rHc23. Effect of adjuvant and antigen dose. PloS one 13:e0193118. https://doi.org/10.1371/journal.pone.0193118
Howell SB, Burke JM, Miller JE, Terrill TH, Valencia E, Williams MJ, Williamson LH, Zajac AM, Kaplan RM (2008) Prevalence of anthelmintic resistance on sheep and goat farms in the southeastern United States. Journal of the American Veterinary Medical Association 233:1913–1919. https://doi.org/10.2460/javma.233.12.1913
Jacobs HJ, Ashman K, Meeusen E (1995) Humoral and cellular responses following local immunization with a surface antigen of the gastrointestinal parasite Haemonchus contortus. Veterinary immunology and immunopathology 48:323–332
Jacobs HJ, Wiltshire C, Ashman K, Meeusen ENT (1999) Vaccination against the gastrointestinal nematode, Haemonchus contortus, using a purified larval surface antigen. Vaccine 17:362–368. https://doi.org/10.1016/S0264-410X(98)00206-0
Kaplan RM, Vidyashankar AN (2012) An inconvenient truth: Global worming and anthelmintic resistance. Veterinary Parasitology 186:70–78. https://doi.org/10.1016/j.vetpar.2011.11.048
Kemper KE, Emery DL, Bishop SC, Oddy H, Hayes BJ, Dominik S, Henshall JM, Goddard ME (2011) The distribution of SNP marker effects for faecal worm egg count in sheep, and the feasibility of using these markers to predict genetic merit for resistance to worm infections. Genetics Research 93:203–219. https://doi.org/10.1017/S0016672311000097
Matthews JB, Geldhof P, Tzelos T, Claerebout E (2016) Progress in the development of subunit vaccines for gastrointestinal nematodes of ruminants. Parasite Immunology 38:744–753. https://doi.org/10.1111/pim.12391
Munn EA, Greenwood CA, Coadwell WJ (1987) Vaccination of young lambs by means of a protein fraction extracted from adult iHaemonchus contortus/i. Parasitology 94:385–397. https://doi.org/10.1017/S0031182000054032
Nisbet AJ, McNeilly TN, Wildblood LA, Morrison AA, Bartley DJ, Bartley Y, Longhi C, McKendrick IJ, Palarea-Albaladejo J, Matthews JB (2013) Successful immunization against a parasitic nematode by vaccination with recombinant proteins. Vaccine 31:4017–4023. https://doi.org/10.1016/J.VACCINE.2013.05.026
Papadopoulos E, Gallidis E, Ptochos S (2012) Anthelmintic resistance in sheep in Europe: A selected review. Veterinary Parasitology 189:85–88. https://doi.org/10.1016/j.vetpar.2012.03.036
Pickering NK, Auvray B, Dodds KG, McEwan JC (2015) Genomic prediction and genome-wide association study for dagginess and host internal parasite resistance in New Zealand sheep. BMC Genomics 16:958. https://doi.org/10.1186/s12864-015-2148-2
Piedrafita DP, de Veer MJ, Sherrard J, Kraska T, Elhay M, Meeusen EN (2012) Field vaccination of sheep with a larval-specific antigen of the gastrointestinal nematode, Haemonchus contortus, confers significant protection against an experimental challenge infection. Vaccine 30:7199–7204. https://doi.org/10.1016/j.vaccine.2012.10.019
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MAR, Bender D, Maller J, Sklar P, de Bakker PIW, Daly MJ, Sham PC (2007) PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. The American Journal of Human Genetics 81:559–575. https://doi.org/10.1086/519795
Riggio V, Matika O, Stear MJ, Bishop SC (2013) Genome-wide association and regional heritability mapping to identify loci underlying variation in nematode resistance and body weight in Scottish Blackface lambs. Heredity 110:420–429. https://doi.org/10.1038/hdy.2012.90
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sánchez-Gracia A (2017) DnaSP 6: DNA Sequence Polymorphism Analysis of Large Data Sets. Molecular Biology and Evolution 34:3299–3302. https://doi.org/10.1093/molbev/msx248
Sallé, G., Jacquiet, P., Gruner, L., Cortet, J., Sauvé, C., Prévot, F., Grisez, C., Bergeaud, J.P., Schibler, L., Tircazes, A., François, D., Pery, C., Bouvier, .F, Thouly, J.C., Brunel, J.C., Legarra, A., Elsen, J. M., Bouix, J., Rupp, R. & Moreno, C. R. (2012) A genome scan for QTL affecting resistance to haemonchus contortus in sheep. Journal of Animal Science 90:4690–4705. https://doi.org/10.2527/jas.2012-5121
Sallé G, Moreno C, Boitard S, Ruesche J, Tircazes-Secula A, Bouvier F, Aletru M, Weisbecker JL, Prévot F, Bergeaud JP, Trumel C, Grisez C, Liénard E, Jacquiet P (2014) Functional investigation of a QTL affecting resistance to Haemonchus contortus in sheep. Veterinary Research 45:1–12. https://doi.org/10.1186/1297-9716-45-68
Smith TJ (2010) Insulin-like growth factor-I regulation of immune function: A potential therapeutic target in autoimmune diseases? Pharmacological Reviews 62:199–236. https://doi.org/10.1124/pr.109.002469
Van Houtert MFJ, Sykes AR (1996) Implications of nutrition for the ability of ruminants to withstand gastrointestinal nematode infections. International Journal for Parasitology 26:1151–1167. https://doi.org/10.1016/S0020-7519(96)00120-8
Villa-Mancera A, Reynoso-Palomar A (2019) Prevalence, economic assessment, and risk factors of gastrointestinal nematodes infecting herds in tropical, dry and temperate climate regions in Mexico. Microbial Pathogenesis 129:50–55. https://doi.org/10.1016/j.micpath.2019.01.043
Waghorn TS, Leathwick DM, Rhodes AP, Lawrence KE, Jackson R, Pomroy WE, West DM, Moffat JR (2006) Prevalence of anthelmintic resistance on sheep farms in New Zealand. New Zealand Veterinary Journal 54:271–277. https://doi.org/10.1080/00480169.2006.36710
Yaman Y, Sevim S, Önaldı T, Sezenler T, İçöz M, ŞENLİK B, Koncagul S, Yilmaz O, Ün C (2018) Effect of the Pappalysin 2 (PAPPA2) gene Exon 2 polymorphisms on genetic resistance to natural gastrointestinal parasite infestation in four native Turkish sheep breeds. In: International Agricultural Science Congress May 09–12, p 181
Zhou X, Stephens M (2012) Genome-wide efficient mixed-model analysis for association studies. Nature Genetics 44:821–824. https://doi.org/10.1038/ng.2310
Acknowledgements
We would like to thank the Ministry of Agriculture and Forestry of the Republic of Turkey, the General Directorate of Agricultural Research and Policies, and the Sheep Breeding and Research Institute in Bandirma, Balikesir/Turkey.
Funding
This research has been funded by the Republic of Turkey Ministry of Agriculture and Forestry, General Directorate of Agricultural Research and Policies (Project number: TAGEM/HAYSÜD/15/A01/P02/02–02).
Author information
Authors and Affiliations
Contributions
Y.Y. conceived and designed the study, S.S., R.A., M.K., and V.B. conducted genetics laboratory works, Y.Y., A.T.Ö., S.S., and R.A. performed field sampling, S.K. and O.Y. prepared and arranged the genotypic and phenotypic data, M.Ö. and B.Ş. conducted the parasitological experiments, and C.Ü. reviewed the manuscript and assisted in data interpretation. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yaman, Y., Bay, V., Sevim, S. et al. Ovine PAPPA2 gene coding variants are linked to decreased fecal egg shedding in native Turkish sheep naturally infected with gastrointestinal nematodes. Trop Anim Health Prod 55, 186 (2023). https://doi.org/10.1007/s11250-023-03612-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-023-03612-5