Abstract
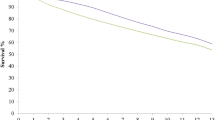
Minimizing bird mortality in the commercial quail breeding industry is important from an economic and welfare perspective. Genetic and non-genetic factors can influence on the cumulative survival of the birds (CS). Accordingly, this study aimed to investigate non-genetic factors on CSs (cumulative survival of the birds from hatch to 5 (CS1), 10 (CS2), 15 (CS3), 20 (CS4), 25 (CS5), 30 (CS6), 35 (CS7), 40 (CS8), and 45 (CS9) days of age), and estimation of the genetic parameters for CSs in crossbred population of quail. Data set included 1794 records from crossbred chicks hatched from 70 sires and 72 dams. The fixed effects were analyzed using an animal model by ASReml software, and all traits were analyzed using Bayesian method via Gibbs sampling by fitting of 6 threshold animal models including the direct genetic effect, the maternal permanent environmental effect, and the maternal genetic effect. The best fitted model for each trait was selected based on the deviance information criteria. Hatch number, the month of hatch, and combination of chickens showed a significant effect on CSs, but the sex of chickens does not have a significant effect on CSs. However, females have higher survival than males (except for CS1). With the best model, the highest and lowest direct heritability was estimated for CS5 (0.386) and CS3 (0.250), respectively. The maternal genetic effect was significant for CS1, CS2, CS3, and CS4 traits, but the maternal permanent environmental effect was significant only for CS1. The range of maternal heritability for CS1 to CS4 traits was estimated from 0.064 to 0.111, and ratio of the permanent environmental variance to phenotypic variance for CS1 was 0.021. The result showed that increasing of the birds’ survival could be performed by correcting non-genetic factors and genetic selection for CSs considering the maternal genetic effects in younger ages.
Highlights
• In the commercial quail breeding industry, the bird mortality is important from an economic and welfare perspective.
• Improving quail survival can be achieved by controlling the genetic and non-genetic factors affecting on survival, so knowledge of these factors is necessary.
• The combination of crossbred chickens had a significant effect on cumulative survival traits.
• The Cumulative survival traits in the crossbred population had relatively high genetic diversity, so genetic selection for these traits could be effective.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Aggrey, S.E., Marks, H.L., 2002. Analysis of censored survival data in Japanese quail divergently selected for growth and their control. Poult. Sci. 81(11), 1618-1620. https://doi.org/10.1093/ps/81.11.1618.
Alemu, S.W., Calus, M.P., Muir, W.M., Peeters, K., Vereijken, A., Bijma, P., 2016. Genomic prediction of survival time in a population of brown laying hens showing cannibalistic behavior. Genet. Sel. Evol. 48(1), 1-10. https://doi.org/10.1186/s12711-016-0247-4.
Behie Eldin, M.M., ElTahawy, W.S., Attia, Y.A., Meky, M.A., 2009. Inheritance of age at sexual maturity and its relationship with some productive traits of Japanese quails. Egypt. Poultry Sci. J. 28, 1217-1232.
Berg, A., Meyer, R., Yu, J., 2004. Deviance information criterion for comparing stochastic volatility models. J. Bus & Econ. Stat. 22, 107-120. https://doi.org/10.1198/073500103288619430.
Bowen, S.J., Washburn, K.W., 1984. Genetics of heat tolerance in Japanese quail. Poult. Sci. 63(3), 430-435. https://doi.org/10.3382/ps.0630430.
Brinker, T., Ellen, E.D., Veerkamp, R.F., Bijma, P., 2015. Predicting direct and indirect breeding values for survival time in laying hens using repeated measures. Genet. Sel. Evol. 47(1), 1-10. https://doi.org/10.1186/s12711-015-0152-2.
Carter, P.S., Rollins, D., Scott, C.B., 2002. Initial effects of prescribed burning on survival and nesting success of northern bobwhite in west-central Texas. Proceedings of the National Quail Symposium. 5,129–134.
Craig, J.V., Muir, W.M., 1996. Group selection for adaptation to multiple-hen cages: beak-related mortality, feathering, and body weight responses. Poult. Sci. 75, 294–302. https://doi.org/10.3382/ps.0750294.
Damgaard, L.H., Korsgaard, I.R., 2006. A bivariate quantitative genetic model for a threshold trait and a survival trait. Genet. Sel. Evol. 38(6), 1-17. https://doi.org/10.1186/1297-9686-38-6-565.
Ducrocq, V., 1997. Survival analysis, a statistical tool for longevity data. In: Proceedings of the 48th Annual Meeting European Association Animal Production, Vienna, Austria, 1997, 25-28.
Ebrahimi, K.h., Dashab, G.H., Faraji-Arough, H., Rokouei, M., 2019. Estimation of additive and non-additive genetic variances of body weight in crossbreed populations of the Japanese quail. Poult. Sci. 98(1), 46-55. https://doi.org/10.3382/ps/pey357.
Ellen, E.D., Bijma, P., 2019. Can breeders solve mortality due to feather pecking in laying hens?. Poult. Sci. 98(9), 3431-3442. https://doi.org/10.3382/ps/pez250.
Ellen, E.D., Visscher, J., van Arendonk, J.A.M., Bijma, P., 2008. Survival of laying hens: Genetic parameters for direct and associative effects in three purebred layer lines. Poult. Sci. 87, 233–239. https://doi.org/10.3382/ps.2007-00374.
Ellen, E.D., Ducrocq, V., Ducro, B.J., Veerkamp, R.F., Bijma. P., 2010. Genetic parameters for social effects on survival in cannibalistic layers: Combining survival analysis and a linear animal model. Genet. Sel. Evol. 42, 27. https://doi.org/10.1186/1297-9686-42-27.
Famula, T., 1981. Exponential stayability model with censoring and covariates. J. Dairy Sci. 64, 538–545. https://doi.org/10.3168/jds.S0022-0302(81)82606-9.
Faraji-Arough, H., Rokouei, M., Maghsoudi, A., Ghazaghi, M., 2018. Comparative study of growth patterns in seven strains of Japanese quail using nonlinear regression modeling. Turk. J. Vet & Anim. Sci. 42(5), 441-451. https://doi.org/10.3906/vet-1801-13.
Gilmour, A.R., Gogel, B.J., Cullis, B.R., Welham, S.J., Thompson, R., 2002. ASREML Reference manual. VSN International Ltd, Hemel Hempstead, UK. http://www.vsn-intl.com. Accessed 26 Jan 2011.
Hagger, C., Hofer, A., 1990. Genetic analyses of calving traits in the Swiss Black and White،Braunvieh and Simmental Breeds by REML and MAPP procedures. Livest. Prod. Sci. 24, 93–107. https://doi.org/10.1016/0301-6226(90)90070-M.
Iranian Council of Animal Care. 1995. Guide to the care and use of expremental animals. Vol. 1. Isfahan University of Technology, Isfahan, Iran.
Jahan, M., Maghsoudi, A., Rokouei, M., Faraji-Arough, H., 2020. Prediction and optimization of slaughter weight in meat-type quails using artificial neural network modeling. Poult. Sci. 99, 1363–1368. https://doi.org/10.1016/j.psj.2019.10.072.
Jamdar, J., Eskandarinasab, M.P., 2014. Comparison of linear and threshold models for estimation genetic and phenotypic parameters of success of conception at first service and inseminations to conception in Holstein Cattles in East Azarbayjan province. Int. J. Advan. Biol & Biomed. Res. 2(5), 1593–1598.
Kaps, M., Lamberson, W.R., 2004. Biostatistics for animal science, CABI publishing, UK.
Kulkarni, P., Mourits, M., Nielen, M., van den Broek, J., Steeneveld, W., 2021. Survival analysis of dairy cows in the Netherlands under altering agricultural policy. Preven. Vet. Med. 105398. https://doi.org/10.1016/j.prevetmed.2021.105398.
Lynch, M., Walsh, B., 1998. Genetics and Analysis of Quantitative Traits. Sinauer Associates Inc., Sunderland, MA.
Meijering, A., Gianola, D., 1985. Linear versus nonlinear metods of sire evolutions forcategorical traits: a simulation study. Genet. Sel. Evol. 17, 115-132. https://doi.org/10.1186/1297-9686-17-1-115
Merilä, J., Sheldon, B.C., 1999. Genetic architecture of fitness and nonfitness traits: Empirical patterns and development of ideas. Hered. 83,103–109. https://doi.org/10.1046/j.1365-2540.1999.00585.x
Mielenz, N., Schmutz, M., Schuler, L., 2005. Mortality of laying hens housed in single and group cages. Arch. Anim. Breed. 48(4), 404-411.
Minvielle, F., Hirigoyen, E., Boulay, M., 1999. Associated effects of the roux plumage color mutation on growth, carcass traits, egg production, and reproduction of Japanese quail. Poult. Sci. 78, 1479-1484. https://doi.org/10.1093/ps/78.11.1479.
Misztal, I., Tsuruta, S., Lourenco, D., Aguilar, I., Legarra, A., Vitezica, Z., 2014. Manual for BLUPF90 family of programs. University of Georgia, Athens. USA http://nce.ads.uga.edu/wiki/lib/exe/fetch.php?media=blupf90_all2.pdf..
Mohammadi-Tighsiah, A., Maghsoudi, A., Bagherzadeh-Kasmani, F., Rokouei, M., Faraji-Arough, H., 2018. Bayesian analysis of genetic parameters for early growth traits and humoral immune responses in Japanese quail. Livest. Sci. 2018, 197-202. https://doi.org/10.1016/j.livsci.2018.07.012.
Nelson, J.N., 2007. Survival and nest site characteristics of translocated mountain quail on Steens Mountain, Oregon. Thesis, Oregon State University, Corvallis, USA.
Okamoto, S., kobayashi, S., Matsuo, T., 1989. Feed conversion to body weight gain and egg production in large and small Japanese quail lines selected for 6-Week body weight. J. Poult. Sci. 26 (4), 227-234. https://doi.org/10.2141/jpsa.26.227.
Peeters, K., Eppink, T.T., Ellen, E.D., Visscher, J., Bijma, P., 2012. Indirect genetic effects for survival in domestic chickens (Gallus gallus) are magnified in crossbred genotypes and show a parent-of-origin effect. Genet. 192(2), 705-713. https://doi.org/10.1534/genetics.112.142554.
Pleasant, G.D., Dabbert, C.B., Mitchell, R.B., 2006. Nesting ecology and survival of scaled quail in the southern High Plains of Texas. J. Wildl. Manag. 70, 632–639. https://doi.org/10.2193/0022-541X(2006)70[632:NEASOS]2.0.CO;2.
Pope, M.D., Crawford, J. A., 2004. Survival rates of translocated and native Mountain Quail in Oregon. West. N. Am. Nat. 64, 331–337.
Quinton, C.D., Wood, B.J., Miller, S.P., 2011. Genetic analysis of survival and fitness in turkeys with multiple-trait animal models. Poult. Sci. 90(11), 2479-2486. https://doi.org/10.3382/ps.2011-01604.
Ramirez-Valverde, R., Misztal, I., Bertrand, J K., 2001. Comparison of threshold vs linear and animal vs sire models for predicting direct and maternal genetic effects on calving difficulty in beef cattle. J. Anim. Sci. 79(2), 333-338. https://doi.org/10.2527/2001.792333x.
Rollins, D., Taylor, B.D., Sparks, T.D., Buntyn, R.J., Lerich, S.E., Harveson, L.A., Wadell, T.E., Scott, C.B., 2009. Survival of female scaled quail during the breeding season at three sites in the Chihuahuan Desert. National Quail Symposium Proceedings, 6, 456-466.
Saghi, R., Rokouei, M., Dashab, G.R., Saghi, D.A., Faraji-Arough, H., 2022. Using a linear-threshold model to investigate the genetic relationship between survival and productive traits in Japanese quail. Ital. J. Anim. Sci. 21(1), 605-611. https://doi.org/10.1080/1828051X.2021.2023332
Silva, L.P., Ribeiro, J.C., Crispim, A.C., Silva, F.G., Bonafé, C.M., Silva, F.F., Torres, R.A., 2013. Genetic parameters of body weight and egg traits in meat-type quail. Livest. Sci. 153(1-3), 27-32. https://doi.org/10.1016/j.livsci.2013.01.014.
Silvestre, A.M., Ginja, M.M.D., Ferreira, A.G.A., Colaco, J., 2007. Comparison of estimates of hip dysplasia genetic parameters in Estrela Mountain Dog using linear and threshold models. J. Anim. Sci. 85, 1880–1884. https://doi.org/10.2527/jas.2007-0166.
Stephenson, J., Reese, K.P., Zager, P., Heekin, P.E., Nelle, P.J., Martens, A., 2011. Factors influencing survival of native and translocated mountain quail in Idaho and Washington. J. Wildl. Manag. 75, 1315–1323. https://doi.org/10.1002/jwmg.189.
Troy, R.J., Coates, P.S., Connelly, J.W., Gillette, G., Delehanty, D.J., 2013. Survival of mountain quail translocated from two distinct source populations. J. Wildl. Manag. 77(5), 1031-1037. https://doi.org/10.1002/jwmg.549.
Acknowledgements
The authors would like to thank the Research Center of Domestic Animals (RCDA) in the University of Zabol, Zabol, Iran, for the cooperation in performing this research.
Author information
Authors and Affiliations
Contributions
H. Faraji-Arough: data curation, conceptualization, visualization, formal analysis, methodology, writing—review and editing; A. Maghsoudi: conceptualization, methodology, writing—review and editing; M. Rokouei: formal analysis, methodology, investigation, writing—original draft.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants performed by any of the authors. Animal handling and experimental procedures of the study were performed based on following the general ethical guideline of the Research Animal Committee of the RCSDA and Iranian Council of Animal Care (1995).
Consent to participate
All authors agree on their participation in the work herein reported.
Consent for publication
All authors confirm to publish the manuscript in Tropical Animal Health and Production.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Faraji-Arough, H., Maghsoudi, A. & Rokouei, M. Study of genetic and non-genetic effects on cumulative survival in a crossbred population of quail. Trop Anim Health Prod 55, 5 (2023). https://doi.org/10.1007/s11250-022-03418-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-022-03418-x