Abstract
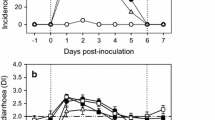
A restricted-randomized, single-blinded, placebo-controlled clinical trial was conducted to examine whether immunomodulating dose of levamisole (LMS) can stimulate certain antiviral immune markers by measuring the concentrations of interferon-γ (IFN-γ), nitric oxide (NOx), and total immunoglobulin G (IgG); prevents the gut injury; and reduces fecal consistency and dehydration scores in rotavirus type A (RVA)-positive piglet diarrhea. The trial was executed between November 2015 and May 2016 in an institute owned experimental swine production farm. The naturally RVA-exposed diarrheic piglets were used in the study. The piglets born between November 2015 and May 2016, age group of 0 to 2 weeks and confirmed for RVA-positive diarrhea, were randomized to receive supportive treatment (ST) or ST along with levamisole (LMS + ST) at immunomodulating dose. Simultaneously, six piglets were randomly selected from healthy population and kept as placebo control. The primary outcome was reduction of fecal consistency and dehydration scores (≤ 1) over the trial period. The secondary outcome was reduction of concentration of gut injury marker and stimulation of immunomodulatory function. The LMS + ST treatment progressively improved the total leukocyte, neutrophil count, IgG concentration (p < 0.05), and reduced the intestinal fatty acid-binding protein 2 (IFABP-2) concentration in RV-positive diarrheic piglets than ST only. Although NOx and IFN-γ concentrations were enhanced initially on day 3, however, the values reduced significantly on day 5 in response to LMS + ST compared to ST. Interestingly, the scores of fecal consistency and dehydration of RVA-positive diarrheic piglets were dropped much earlier (on day 3) in response to LMS + ST than ST alone. The results indicate that LMS along with supportive treatment non-specifically modulated innate immunity and restored intestinal gut health, and thus, LMS may represent an additional therapeutic agent for management of RVA-inflicted piglet diarrhea.





Similar content being viewed by others
Abbreviations
- LMS:
-
Levamisole
- IFN-γ:
-
Interferon-γ
- NOx:
-
Nitric oxide
- IgG:
-
Immunoglobulin G
- IFABP-2:
-
Intestinal fatty acid-binding protein 2
- RVA:
-
Rotavirus type A
- ST:
-
Supportive treatment
- NSP1:
-
Non-structural protein 1
- RNA-PAGE:
-
Ribonucleic acid-polyacrylamide gel electrophoresis
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- PBMCs:
-
Peripheral blood mononuclear cells
- cDNA:
-
Complimentary DNA
References
Berkes, J., Viswanathan, V.K., Savkovic, S.D. and Hecht, G., 2003. Intestinal epithelial responses to enteric pathogens:effects on the tight junction barrier, ion transport, and inflammation, Gut, 52, 439–451
Berkeveld, M., Langendijk, P., Verheijden, J.H.M., Taverne, M.A.M., Van Nes, A., Van Haard, P. and Koets, A.P., 2008. Citrulline and intestinal fatty acid-binding protein: Longitudinal markers of postweaning small intestinal function in pigs?, Journal of Animal Science, 86, 3440–3449
Besser, T.E., McGuire, T.C., Gay, C.C. and Pritchett, L.C., 1988. Transfer of functional immunoglobulin G (IgG) antibody into the gastrointestinal tract accounts for IgG clearance in calves, Journal of Virology, 62, 2234–2237
Bilandzic, N., Mitak, M. and Simic, B., 2010. Levamisole increases leukocyte count and immunoglobulin levels in young boars, Sloverian Veterinary Research, 47, 13–19
Bilbao, G.N., Chacana, P.A., Mendiburu, A., Rodriguez, E., Blackhall, J.O. and Terzolo, H.R., 2006. Prophylaxis of neonatal diarrhoea in dairy calves using egg yolk immunoglobulins (IgY), Revue de Medecine Veterinaire, 87, 135–139
Borghan, M.A., Mori, Y., El-Mahmoudy, A.B., Ito, N., Sugiyama, M., Takewaki, T. and Minamoto, N., 2007. Induction of nitric oxide synthase by rotavirus enterotoxin NSP4: implication for rotavirus pathogenicity, Journal of General Virology, 88, 2064–2072
Boshuizen, J.A., Reimerink, J.H., Korteland-van Male, A.M., van Ham, V.J., Koopmans, M.P., Buller, H.A., Dekker, J. and Einerhand, A.W., 2003 Changes in small intestinal homeostasis, morphology, and gene expression during rotavirus infection of infant mice, Journal of Virology, 77, 13005–13016
Bozic, F., Lackovic, G., Stokes, C.R. and Valpotic, I., 2002. Recruitment of intestinal CD45RA+ and CD45RC+ cells induced by a candidate oral vaccine against porcine post-weaning colibacillosis, Veterinary Immunology and Immunopathology, 86, 137–146
Bugarcic, A. and Taylor, J.A., 2006. Rotavirus nonstructural glycoprotein NSP4 is secreted from the apical surfaces of polarized epithelial cells, Journal of Virology, 80, 12343–12349
Cabrera, R.A., Lin, X., Campbell, J.M., Moeser, A.J. and Odle, J. 2012. Influence of birth order, birth weight, colostrum and serum immunoglobulin G on neonatal piglet survival, Journal of Animal Science and Biotechnology, 3, 42
Chethan, G.E., Garkhal, J., Sircar, S., Malik, Y.P.S., Mukherjee, R., Sahoo, N.R., Agarwal, R.K. and De, U.K., 2017a. Immunomodulatory potential of β-glucan as supportive treatment in porcine rotavirus enteritis, Veterinary Immunology and Immunopathology, 191, 36–43
Chethan, G.E., Garkhal, J., Sircar, S., Malik, Y.P.S., Mukherjee, R., Gupta, V.K., Sahoo, N.R., Agarwal, R.K. and De, U.K., 2017b. Changes of haemogram and serum biochemistry in neonatal piglet diarrhoea associated with porcine rotavirus type A, Tropical Animal Health and Production, 49, 1517–1522
De, U.K., Mukherjee, R., Nandi, S., Patel, B.H.M., Dimri, U., Ravishankar, C. and Verma, A.K., 2014. Alterations in oxidant/antioxidant balance, high-mobility group box 1 protein and acute phase response in cross-bred suckling piglets suffering from rotaviral enteritis, Tropical Animal Health and Production, 46, 1127–1133
Dundar, Z.D., Cander, B., Gul, M., Karabulut, K.U., Kocak, S., Girisgin, S., Mehmetoglu, I. and Toy, H., 2012. Serum intestinal fatty acid binding protein and phosphate levels in the diagnosis of acute intestinal ischemia: an experimental study in rabbits, Journal of Emergency Medicine, 42, 741–747
Festing, M. F. and Altman, D. G. 2002. Guidelines for the design and statistical analysis of experiments using laboratory animals. Institute for Laboratory Animal Research Journal, 43, 244–258
Goddard, A., Leisewitz, A.L., Christopher, M.M., Duncan, N.M. and Becker, P.J., 2008. Prognostic usefulness of blood leukocyte changes in canine parvoviral enteritis, Journal of Veterinary Internal Medicine, 22, 309–316
Greenberg, H.B. and Estes, M.K., 2009. Rotaviruses: from pathogenesis to vaccination, Gastroenterology, 136, 1939–1951
Gupta, M., 2016. Levamisole: A multi-faceted drug in dermatology, Indian journal of dermatology, venereology and leprology, 82, 230–236
Habibi, M., Ghahri, H., Zadeh, R.S. and Yeganeh, F., 2012. Effects of levamisole on the immune response of broilers against Newcastle disease vaccines, African Journal of Pharmacy and Pharmacology, 6, 1860–1864
Jain, N.C., 1986. Schalm’s Veterinary Hematology, fourth edn., (Lea and Febiger, Philadelphia)
Jin, H., Li, Y., Ma, Z., Zhang, F., Xie, Q., Gu, D. and Wang, B., 2004. Effect of chemical adjuvants on DNA vaccination, Vaccine, 22, 2925–2935
Kang, J.Y., Lee, D.K., Ha, N.J. and Shin, H.S., 2015. Antiviral effects of Lactobacillus ruminis SPM0211 and Bifidobacterium longum SPM1205 and SPM1206 on rotavirus-infected Caco-2 cells and a neonatal mouse model, Journal of Microbiology, 53, 796–803
Kim, H.J., Park, J.G., Matthijnssens, J., Lee, J.H., Bae, Y.C., Alfajaro, M.M., Park, S.I., Kang, M.I. and Cho, K.O., 2011. Intestinal and extra-intestinal pathogenicity of a bovine reassortant rotavirus in calves and piglets, Veterinary Microbiology, 152, 291–303
Kitasato, A., Tajima, Y., Kuroki, T., Tsutsumi, R., Adachi, T., Mishima, T. and Kanematsu, T., 2007. Inflammatory cytokines promote inducible nitric oxide synthase-mediated DNA damage in hamster gallbladder epithelial cells, World Journal of Gastroenterology, 13, 6379
Kittaka, H., Akimoto, H., Takeshita, H., Funaoka, H., Hazui, H., Okamoto, M., Kobata, H. and Ohishi, Y., 2014. Usefulness of intestinal fatty acid-binding protein in predicting strangulated small bowel obstruction, PLoS One, 9, e99915
Koyama, S., Ishii, K.J., Coban, C. and Akira, S., 2008. Innate immune response to viral infection, Cytokine, 43, 336–341
Kumar, S., Dewey, C.E., Friendship, R.M., Bowland, S.L. and Shewen, P.E., 1999. Improved weight gain in pigs using levamisole as an Immunomodulatory, Journal of Swine Health and Production, 7, 103–107
Kumar, S., Sharma, G., Sidiq, T., Khajuria, A., Jain, M., Bhagwat, D. and Dhar, K.L., 2014. Immunomodulatory potential of a bioactive fraction from the leaves of Phyllostachys bambusoides (Bamboo) in BALB/C mice, EXCLI Journal, 13, 137
Ladinig, A., Lunney, J.K., Souza, C.J., Ashley, C., Plastow, G. and Harding, J.C., 2014. Cytokine profiles in pregnant gilts experimentally infected with porcine reproductive and respiratory syndrome virus and relationships with viral load and fatal outcome, Veterinary Research, 45, 113
Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4, Nature, 227, 680–685
Malemud, C.J., 2017. Immunomodulators in autoimmunity and viral Infections, Journal of Clinical and Cellular Immunology, 9, 537
Malik, Y.P.S., Sharma, K., Vaid, N., Chakravarti, S., Chandrashekar, K.M., Basera, S.S., Singh, R., Prasad, G., Gulati, B.R., Bhilegaonkar, K.N. and Pandey, A.B., 2012. Frequency of group A rotavirus with mixed G and P genotypes in bovines: predominance of G3 genotype and its emergence in combination with G8/ G10 types, Journal of Veterinary Science, 13, 271–278
Malik, Y.P.S., Kumar, N., Sharma, K., Sircar, S. and Bora, D.P., 2014. Rotavirus diarrhoea in piglets: A review on epidemiology, genetic diversity and zoonotic risks, Indian Journal of Animal Sciences, 84 (10), 1035–1942
McLamb, B.L., Gibson, A.J., Overman, E.L., Stahl, C. and Moeser, A.J., 2013. Early weaning stress in pigs impairs innate mucosal immune responses to enterotoxigenic E. coli challenge and exacerbates intestinal injury and clinical disease, PLoS ONE, 8, e59838
Mehta, D.R., Ashkar, A.A. and Mossman, K.L., 2012. The nitric oxide pathway provides innate antiviral protection in conjunction with the type I interferon pathway in fibroblasts, PLoS ONE, 7, e31688
Mohamed, W.A.M. and Abdou, W., 2014. Can lactoferrin modulate the immunostimulant activity of levamisole in rats immunosuppressed by cyclophosphamide?, Journal of Clinical and Experimental Investigations, 5, 48–53
Mondal, A., Sharma, K., Malik, Y.P.S. and Joardar, S.N., 2013. Detection of group A rotavirus in faeces of diarrhoeic bovine porcine and human population from eastern India by reverse transcriptase–polymerase chain reaction, Advances in Animal and Veterinary Sciences, 1, 18–19
Osborne, M.P., Haddon, S.J., Worton, K.J., Spencer, A.J., Starkey, W.G., Thornber, D. and Stephen, J., 1991. Rotavirus-induced changes in the microcirculation of intestinal villi of neonatal mice in relation to the induction and persistence of diarrhoea, Journal of Pediatric Gastroenterology and Nutrition, 12, 111–120
Pekmezci, D. and Cakiroglu, D., 2009. Investigation of immunmodulatory effects of levamisole and vitamin E on Immunity and some blood parameters in newborn Jersey calves, Veterinary Research Communications, 33, 711–721
Pereira, S.J., Ramirez, N.E., Xiao, L. and Ward, L.A., 2002. Pathogenesis of human and bovine Cryptosporidium parvum in gnotobiotic pigs, Journal of Infectious Diseases, 186, 715–718
Perera, H.A.C.C. and Pathiratne, A., 2008. Enhancement of immune responses in Indian carp, Catla catla, following administration of Levamisole by immersion, Disease in Asian Aquaculture VI, Fish Health Section, 129–142
Reiss, C.S. and Komatsu, T., 1998. Does nitric oxide play a critical role in viral infections?, Journal of Virology, 72, 4547–4551
Rocha, T.G., Silva, F.D.F., Bortoletto, C., Silva, D.G., Buzinaro, M.G., Zafalon, L.F. and Fagliari, J.J., 2016. Serum concentrations of acute phase proteins and immunoglobulins of calves with rotavirus diarrhoea, Arquivo Brasileiro de Medicina Veterinaria e Zootecnia, 68, 865–872
Sastry, G.A., 1985. Veterinary Clinical Pathology, third eds., (CBS publishers and distributors, India)
Saurabh, S., Sircar, S., Kattoor, J.J., Ghosh, S., Kobayashi, N., Banyai, K., VinodhKumar, O.R., De, U.K., Sahoo, N.R., Dhama, K. and Malik, Y.S., 2018. Analysis of structure-function relationship in porcine rotavirus A enterotoxin gene, Journal of Veterinary Science, 19, 35–43
Shim, J.O., Son, D.W., Shim, S.Y., Ryoo, E., Kim, W. and Jung, Y.C., 2012. Clinical characteristics and genotypes of rotaviruses in a neonatal intensive care unit, Pediatrics and Neonatology, 53, 18–23
Sinha, S., Relhan, V. and Garg, V.K., 2015. Immunomodulators in warts: Unexplored or ineffective?, Indian Journal of Dermatology, 60, 118
Svenson, L., Uhnoo, I., Grandien, M. and Wadell, G., 1986. Molecular epidemiology of rotavirus infections in Upsala, Sweden in (1981): Disappearance of predominant electropherotypes, Journal of Medical Virology, 18, 101–111
Szeto, C.C., Gillespie, K.M. and Mathieson, P.W., 2000. Levamisole induces interleukin-18 and shifts type 1/type 2 cytokine balance, Immunology, 100, 217–224
Valpotic, H., Speranda, M., Kovsca-Janjatovic, A., Didara, M., Lackovic, G., Bozic, F., Habrun, B., Srecec, S., Matausic-Pisl, M. and Valpotic, I., 2013. Levamisole stimulates proliferation of circulating and intestinal immune cell subsets, gut health and performance in weaned pig, Canadian Journal of Animal Science, 94, 43–53
Valpotic, H., Baric-Rafaj, R., Mrljak, V., Bozic, F., Grabarevic, Z., Samardzija, M., Folnozic, I., Duricic, D., Gracner, D. and Valpotic, I., 2016. The influences of immune modulation with levamisole and polyoxyethylene-polyoxypropylene copolymers on the immunohematological, serum biochemical parameters and intestinal histocytomorphology of weaned pigs, Veterinarski Arhiv, 86, 667–684
Westerman, L.E., McClure, H.M., Jiang, B., Almond, J.W. and Glass, R.I., 2005. Serum IgG mediates mucosal immunity against rotavirus infection, Proceedings of the National Academy of Sciences of the United States of America, 102, 7268–7273
Wright, D.G., Kirkpatrick, C.H. and Gallin, J.I., 1977. Effects of levamisole on normal and abnormal leukocyte locomotion, Journal of Clinical Investigation, 59, 941
Yucel, A.A., Gulen, S., Dincer, S., Yucel, A.E. and Yetkin, G.I., 2012. Comparison of two different applications of the Griess method for nitric oxide measurement, Journal of Experimental and Integrative Medicine, 2, 167–171
Acknowledgements
The study was supported by All India Network Programme on Neonatal Mortality project of ICAR. CGE and JG thank ICAR-IVRI for granting research fellowship for their research program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Animal studies
Animal studies were carried out humanely and according to national and international Animal Care and Use Committee protocols and following the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India (approval number F.25/08/2016-CPCSEA).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chethan, G.E., Kumar De, U., Garkhal, J. et al. Immunomodulating dose of levamisole stimulates innate immune response and prevents intestinal damage in porcine rotavirus diarrhea: a restricted-randomized, single-blinded, and placebo-controlled clinical trial. Trop Anim Health Prod 51, 1455–1465 (2019). https://doi.org/10.1007/s11250-019-01833-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-019-01833-1