Abstract
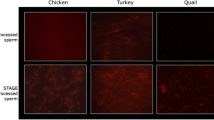
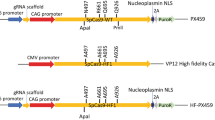
Generating transgenic and gene edited mammals involves in vitro manipulation of oocytes or single cell embryos. Due to the comparative inaccessibility of avian oocytes and single cell embryos, novel protocols have been developed to produce transgenic and gene edited birds. While these protocols are relatively efficient, they involve two generation intervals before reaching complete somatic and germline expressing transgenic or gene edited birds. Most of this work has been done with chickens, and many protocols require in vitro culturing of primordial germ cells (PGCs). However, for many other bird species no methodology for long term culture of PGCs exists. Developing methodologies to produce germline transgenic or gene edited birds in the first generation would save significant amounts of time and resource. Furthermore, developing protocols that can be readily adapted to a wide variety of avian species would open up new research opportunities. Here we report a method using sperm as a delivery mechanism for gene editing vectors which we call sperm transfection assisted gene editing (STAGE). We have successfully used this method to generate GFP knockout embryos and chickens, as well as generate embryos with mutations in the doublesex and mab-3 related transcription factor 1 (DMRT1) gene using the CRISPR/Cas9 system. The efficiency of the method varies from as low as 0% to as high as 26% with multiple factors such as CRISPR guide efficiency and mRNA stability likely impacting the outcome. This straightforward methodology could simplify gene editing in many bird species including those for which no methodology currently exists.





Similar content being viewed by others
References
Ahammad MU, Nishino C, Tatemoto H, Okura N, Okamoto S, Kawamoto Y, Nakada T (2013) Plumping fluid added to storage medium increases twofold the functional life span of fowl spermatozoa in vitro at 4 °C. Br Poult Sci 54:270–280
An L, Hu Y, Chang S, Zhu X, Ling P, Zhang F, Liu J, Liu Y, Chen Y, Yang L, Presicce GA, Du F (2016) Efficient generation of FVII gene knockout mice using CRISPR/Cas9 nuclease and truncated guided RNAs. Sci Rep 6:25199
Balic A, Garcia-Morales C, Vervelde L, Gilhooley H, Sherman A, Garceau V, Gutowska MW, Burt DW, Kaiser P, Hume DA, Sang HM (2014) Visualisation of chicken macrophages using transgenic reporter genes: insights into the development of the avian macrophage lineage. Development 141:3255–3265
Ball BA, Sabeur K, Allen WR (2008) Liposome-mediated uptake of exogenous DNA by equine spermatozoa and applications in sperm-mediated gene transfer. Equine Vet J 40:76–82
Bishop JO, Smith P (1989) Mechanism of chromosomal integration of microinjected DNA. Mol Biol Med 6:283–298
Brocal I, White RJ, Dooley CM, Carruthers SN, Clark R, Hall A, Busch-Nentwich EM, Stemple DL, Kettleborough RN (2016) Efficient identification of CRISPR/Cas9-induced insertions/deletions by direct germline screening in zebrafish. BMC Genom 17:259
Carlson DF, Tan W, Lillico SG, Stverakova D, Proudfoot C, Christian M, Voytas DF, Long CR, Whitelaw CB, Fahrenkrug SC (2012) Efficient TALEN-mediated gene knockout in livestock. Proc Natl Acad Sci USA 109:17382–17387
Celebi C, Guillaudeux T, Auvray P, Vallet-Erdtmann V, Jegou B (2003) The making of ‘transgenic spermatozoa’. Biol Reprod 68:1477–1483
Chu VT, Weber T, Wefers B, Wurst W, Sander S, Rajewsky K, Kühn R (2015) Increasing the efficiency of homology-directed repair for CRISPR-Cas9-induced precise gene editing in mammalian cells. Nat Biotechnol 33:543–548
Collares T, Campos VF, De Leon PM, Cavalcanti PV, Amaral MG, Dellagostin OA, Deschamps JC, Seixas FK (2011) Transgene transmission in chickens by sperm-mediated gene transfer after seminal plasma removal and exogenous DNA treated with dimethylsulfoxide or N, N-dimethylacetamide. J Biosci 36:613–620
Conrad K, Buckley J, Stambaugh R (1971) Studies on the nature of the block to polyspermy in rabbit ova. J Reprod Fertil 27:133–135
Dai J, Cui X, Zhu Z, Hu W (2010) Non-homologous end joining plays a key role in transgene concatemer formation in transgenic zebrafish embryos. Int J Biol Sci 6:756–768
Deriano L, Roth DB (2013) Modernizing the nonhomologous end-joining repertoire: alternative and classical NHEJ share the stage. Annu Rev Genet 47:433–455
Dhanapala P, Doran T, Tang ML, Suphioglu C (2015) Production and immunological analysis of IgE reactive recombinant egg white allergens expressed in Escherichia coli. Mol Immunol 65:104–112
Dimitrov L, Pedersen D, Ching KH, Yi H, Collarini EJ, Izquierdo S, van de Lavoir MC, Leighton PA (2016) Germline gene editing in chickens by efficient CRISPR-mediated homologous recombination in primordial germ cells. PLoS ONE 11:e0154303
Elis S, Batellier F, Couty I, Balzergue S, Martin-Magniette ML, Monget P, Blesbois E, Govoroun MS (2008) Search for the genes involved in oocyte maturation and early embryo development in the hen. BMC Genom 9:110
Gagnon JA, Valen E, Thyme SB, Huang P, Akhmetova L, Pauli A, Montague TG, Zimmerman S, Richter C, Schier AF (2014) Efficient mutagenesis by Cas9 protein-mediated oligonucleotide insertion and large-scale assessment of single-guide RNAs. PLoS ONE 9:e98186
Gandolfi F (2000) Sperm-mediated transgenesis. Theriogenol 53:127–137
García-Vázquez FA, García-Roselló E, Gutiérrez-Adán A, Gadea J (2009) Effect of sperm treatment on efficiency of EGFP-expressing porcine embryos produced by ICSI-SMGT. Theriogenology 72:506–518
Hamburger V, Hamilton HL (1951) A series of normal stages in the development of the chick embryo. J Morphol 88:49–92
Hoa NN, Akagawa R, Yamasaki T, Hirota K, Sasa K, Natsume T, Kobayashi J, Sakuma T, Yamamoto T, Komatsu K, Kanemaki MT, Pommier Y, Takeda S, Sasanuma H (2015) Relative contribution of four nucleases, CtIP, Dna2, Exo1 and Mre11, to the initial step of DNA double-strand break repair by homologous recombination in both the chicken DT40 and human TK6 cell lines. Genes Cells 20:1059–1076
Huang AY, Xu X, Ye AY, Wu Q, Yan L, Zhao B, Yang X, He Y, Wang S, Zhang Z, Gu B, Zhao HQ, Wang M, Gao H, Gao G, Zhang Z, Yang X, Wu X, Zhang Y, Wei L (2014) Postzygotic single-nucleotide mosaicisms in whole-genome sequences of clinically unremarkable individuals. Cell Res 24:1311–1327
Hwang WY, Fu Y, Reyon D, Maeder ML, Kaini P, Sander JD, Joung JK, Peterson RT, Yeh JR (2013) Heritable and precise zebrafish genome editing using a CRISPR-Cas system. PLoS ONE 8:e68708
Kleinstiver BP, Pattanayak V, Prew MS, Tsai SQ, Nguyen NT, Zheng Z, Joung JK (2016) High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 529:490–495
Lake PE (1957) Fowl semen as collected by the massage method. J Agr Sci 49:120–126
Lake PE, Ravie O (1979) Effect on fertility of storing fowl semen for 24 h at 5 °C in fluids of different pH. J Reprod Fertil 57:149–155
Lambeth LS, Raymond CS, Roeszler KN, Kuroiwa A, Nakata T, Zarkower D, Smith CA (2014) Over-expression of DMRT1 induces the male pathway in embryonic chicken gonads. Dev Biol 389:160–172
Lambeth LS, Morris KR, Wise TG, Cummins DM, O’Neil TE, Cao Y, Sinclair AH, Doran TJ, Smith CA (2016) Transgenic chickens overexpressing aromatase have high estrogen levels but maintain a predominantly male phenotype. Endocrinology 157:83–90
Lavitrano M, Camaioni A, Fazio VM, Dolci S, Farace MG, Spadafora C (1989) Sperm cells as vectors for introducing foreign DNA into eggs: genetic transformation of mice. Cell 57:717–723
Lavitrano M, French D, Zani M, Frati L, Spadafora C (1992) The interaction between exogenous DNA and sperm cells. Mol Reprod Dev 31:161–169
Lee HC, Choi HJ, Park TS, Lee SI, Kim YM, Rengaraj D, Nagai H, Sheng G, Lim JM, Han JY (2013) Cleavage events and sperm dynamics in chick intrauterine embryos. PLoS ONE 8:e80631
Liang P, Xu Y, Zhang X, Ding C, Huang R, Zhang Z, Lv J, Xie X, Chen Y, Li Y, Sun Y, Bai Y, Songyang Z, Ma W, Zhou C, Huang J (2015) CRISPR/Cas9-mediated gene editing in human tripronuclear zygotes. Protein Cell 6:363–372
Lin CY, Su YH (2016) Genome editing in sea urchin embryos by using a CRISPR/Cas9 system. Dev Biol 409:420–428
Liu T, Wu H, Cao D, Li Q, Zhang Y, Li N, Hu X (2015) Oviduct-specific expression of human neutrophil defensin 4 in lentivirally generated transgenic chickens. PLoS ONE 10:e0127922
Long C, McAnally JR, Shelton JM, Mireault AA, Bassel-Duby R, Olson EN (2014) Prevention of muscular dystrophy in mice by CRISPR/Cas9-mediated editing of germline DNA. Science 345:1184–1188
Lord T, Aitken RJ (2015) Fertilization stimulates 8-hydroxy-2′-deoxyguanosine repair and antioxidant activity to prevent mutagenesis in the embryo. Dev Biol 406:1–13
Lyall J, Irvine RM, Sherman A, McKinley TJ, Núñez A, Purdie A, Outtrim L, Brown IH, Rolleston-Smith G, Sang H, Tiley L (2011) Suppression of avian influenza transmission in genetically modified chickens. Science 331:223–226
Ma S, Chang J, Wang X, Liu Y, Zhang J, Lu W, Gao J, Shi R, Zhao P, Xia Q (2014a) CRISPR/Cas9 mediated multiplex genome editing and heritable mutagenesis of BmKu70 in Bombyx mori. Sci Rep 4:4489
Ma Y, Shen B, Zhang X, Lu Y, Chen W, Ma J, Huang X, Zhang L (2014b) Heritable multiplex genetic engineering in rats using CRISPR/Cas9. PLoS ONE 9:e89413
Mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, Norville JE, Church GM (2013) RNA-guided human genome engineering via Cas9. Science 339:823–826
Marchetti F, Bishop J, Gingerich J, Wyrobek AJ (2015) Meiotic interstrand DNA damage escapes paternal repair and causes chromosomal aberrations in the zygote by maternal misrepair. Sci Rep 5:7689
Maruyama T, Dougan SK, Truttmann MC, Bilate AM, Ingram JR, Ploegh HL (2015) Increasing the efficiency of precise genome editing with CRISPR-Cas9 by inhibition of nonhomologous end joining. Nat Biotechnol 33:538–542
McGrew MJ, Sherman A, Ellard FM, Lillico SG, Gilhooley HJ, Kingsman AJ, Mitrophanous KA, Sang H (2004) Efficient production of germline transgenic chickens using lentiviral vectors. EMBO Rep 5:728–733
Men NT, Kikuchi K, Furusawa T, Dang-Nguyen TQ, Nakai M, Fukuda A, Noguchi J, Kaneko H, Viet Linh N, Xuan Nguyen B, Tajima A (2016) Expression of DNA repair genes in porcine oocytes before and after fertilization by ICSI using freeze-dried sperm. Anim Sci J. doi:10.1111/asj.12554
Miller DJ, Gong X, Decker G, Shur BD (1993) Egg cortical granule N-acetylglucosaminidase is required for the mouse zona block to polyspermy. J Cell Biol 123:1431–1440
Niemann H, Kues WA (2003) Application of transgenesis in livestock for agriculture and biomedicine. Anim Reprod Sci 79:291–317
Oishi I, Yoshii K, Miyahara D, Kagami H, Tagami T (2016) Targeted mutagenesis in chicken using CRISPR/Cas9 system. Sci Rep 6:23980
Oliver D, Yuan S, McSwiggin H, Yan W (2015) Pervasive genotypic mosaicism in founder mice derived from genome editing through pronuclear injection. PLoS ONE 10:e0129457
Park TS, Lee HJ, Kim KH, Kim JS, Han JY (2014) Targeted gene knockout in chickens mediated by TALENs. Proc Natl Acad Sci USA 111:12716–12721
Park TS, Lee HG, Moon JK, Lee HJ, Yoon JW, Yun BN, Kang SC, Kim J, Kim H, Han JY, Han BK (2015) Deposition of bioactive human epidermal growth factor in the egg white of transgenic hens using an oviduct-specific minisynthetic promoter. FASEB J 29:2386–2396
Pereyra-Bonnet F, Gibbons A, Cueto M, Sipowicz P, Fernández-Martín R, Salamone D (2011) Efficiency of sperm-mediated gene transfer in the ovine by laparoscopic insemination, in vitro fertilization and ICSI. J Reprod Dev 57:188–196
Shemesh M, Gurevich M, Harel-Markowitz E, Benvenisti L, Shore LS, Stram Y (2000) Gene integration into bovine sperm genome and its expression in transgenic offspring. Mol Reprod Dev 56:306–308
Shen W, Li L, Pan Q, Min L, Dong H, Deng J (2006) Efficient and simple production of transgenic mice and rabbits using the new DMSO-sperm mediated exogenous DNA transfer method. Mol Reprod Dev 73:589–594
Shimada K, Ono T, Mizushima S (2014) Application of intracytoplasmic sperm injection (ICSI) for fertilization and development in birds. Gen Comp Endocrinol 196:100–105
Singh RP, Shafeeque CM, Sharma SK, Singh R, Mohan J, Sastry KV, Saxena VK, Azeez PA (2016) Chicken sperm transcriptome profiling by microarray analysis. Genome 59:185–196
Siudzinska A, Łukaszewicz E (2008) Effect of semen extenders and storage time on sperm morphology of four chicken breeds. J Appl Poult Res 17:101–108
Smith K, Spadafora C (2005) Sperm-mediated gene transfer: applications and implications. BioEssays 27:551–562
Smith CA, Roeszler KN, Ohnesorg T, Cummins DM, Farlie PG, Doran TJ, Sinclair AH (2009) The avian Z-linked gene DMRT1 is required for male sex determination in the chicken. Nature 461:267–271
Song Y, Duraisamy S, Ali J, Kizhakkayil J, Jacob VD, Mohammed MA, Eltigani MA, Amisetty S, Shukla MK, Etches RJ, de Lavoir MC (2014) Characteristics of long-term cultures of avian primordial germ cells and gonocytes. Biol Reprod 90:15
Suzuki T, Asami M, Perry AC (2014) Asymmetric parental genome engineering by Cas9 during mouse meiotic exit. Sci Rep 4:7621
Tan W, Carlson DF, Lancto CA, Garbe JR, Webster DA, Hackett PB, Fahrenkrug SC (2013) Efficient nonmeiotic allele introgression in livestock using custom endonucleases. Proc Natl Acad Sci USA 110:16526–16531
Tyack SG, Jenkins KA, O’Neil TE, Wise TG, Morris KR, Bruce MP, McLeod S, Wade AJ, McKay J, Moore RJ, Schat KA, Lowenthal JW, Doran TJ (2013) A new method for producing transgenic birds via direct in vivo transfection of primordial germ cells. Transgenic Res 22:1257–1264
van de Lavoir MC, Diamond JH, Leighton PA, Mather-Love C, Heyer BS, Bradshaw R, Kerchner A, Hooi LT, Gessaro TM, Swanberg SE, Delany ME, Etches RJ (2006) Germline transmission of genetically modified primordial germ cells. Nature 441:766–769
Wang H, Yang H, Shivalila CS, Dawlaty MM, Cheng AW, Zhang F, Jaenisch R (2013) One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153:910–918
Wang X, Niu Y, Zhou J, Yu H, Kou Q, Lei A, Zhao X, Yan H, Cai B, Shen Q, Zhou S, Zhu H, Zhou G, Niu W, Hua J, Jiang Y, Huang X, Ma B, Chen Y (2016) Multiplex gene editing via CRISPR/Cas9 exhibits desirable muscle hypertrophy without detectable off-target effects in sheep. Sci Rep 6:32271
Wei J, Wagner S, Lu D, Maclean P, Carlson DF, Fahrenkrug SC, Laible G (2015) Efficient introgression of allelic variants by embryo-mediated editing of the bovine genome. Sci Rep 5:11735
Whitworth KM, Rowland RR, Ewen CL, Trible BR, Kerrigan MA, Cino-Ozuna AG, Samuel MS, Lightner JE, McLaren DG, Mileham AJ, Wells KD, Prather RS (2016) Gene-edited pigs are protected from porcine reproductive and respiratory syndrome virus. Nat Biotechnol 34:20–22
Wishart GJ (1997) Quantitative aspects of sperm: egg interaction in chickens and turkeys. Anim Reprod Sci 48:81–92
Wu H, Wang Y, Zhang Y, Yang M, Lv J, Liu J, Zhang Y (2015) TALE nickase-mediated SP110 knockin endows cattle with increased resistance to tuberculosis. Proc Natl Acad Sci USA 112:E1530–E1539
Yen ST, Zhang M, Deng JM, Usman SJ, Smith CN, Parker-Thornburg J, Swinton PG, Martin JF, Behringer RR (2014) Somatic mosaicism and allele complexity induced by CRISPR/Cas9 RNA injections in mouse zygotes. Dev Biol 393:3–9
Yoshimi K, Kaneko T, Voigt B, Mashimo T (2014) Allele-specific genome editing and correction of disease-associated phenotypes in rats using the CRISPR-Cas platform. Nat Commun 5:4240
Zagris N, Kalantzis K, Guialis A (1998) Activation of embryonic genome in chick. Zygote 6:227–231
Zani M, Lavitrano M, French D, Lulli V, Maione B, Sperandio S, Spadafora C (1995) The mechanism of binding of exogenous DNA to sperm cells—factors controlling the DNA uptake. Expt Cell Res 217:57–64
Zhong H, Chen Y, Li Y, Chen R, Mardon G (2015) CRISPR-engineered mosaicism rapidly reveals that loss of Kcnj13 function in mice mimics human disease phenotypes. Sci Rep 5:8366
Acknowledgements
The authors would like to thank Sandy Matheson, Noel Collins, Chris Darcy, and Susan Wilson for helping care for and handle the animals used in this study. We would also like to thank Matt Bruce for technical assistance with the flow cytometer. This study was funded by the Commonwealth Scientific and Industrial Research Organisation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cooper, C.A., Challagulla, A., Jenkins, K.A. et al. Generation of gene edited birds in one generation using sperm transfection assisted gene editing (STAGE). Transgenic Res 26, 331–347 (2017). https://doi.org/10.1007/s11248-016-0003-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-016-0003-0