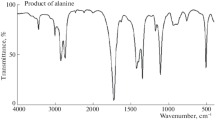
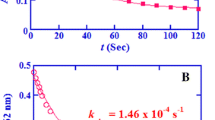
Abstract
The kinetics of oxidation of β-alanine by the Ag(III) complex has been studied by spectrophotometry. The reaction is first order with respect to the Ag(III) complex. The values of k obs decrease with an increase of [OH−], and then increase with the increase of [OH−]. The concentration of OH− was 0.03 mol · L−1 at the turning point at which the rate was the slowest. A plausible mechanism of reaction is proposed, and the rate law has been obtained, which could be applied to explain all experimental phenomena. The activation parameters of the rate-determining step have been also calculated.
Similar content being viewed by others
References
Gowda B.T., Jagan P. and Rao M. (1989). Bull. Chem. Soc. Jpn. 62: 3303
Katre Y.R., Singh A.K., Patil S. and Joshi G.K. (2006). Oxidation Commun. 29: 129
Katre Y.R., Singh A.K., Joshi G.K. and Patil S. (2006). Oxidation Commun. 29: 137
Nalwaya N., Jain A. and Hiran B.L. (2004). Kinet. Catal. 45: 345
Shan J.H., Li S.M., Huo S.Y., Shen S.G. and Sun H.W. (2006). Chinese J.␣Chem. 24: 478
Shan J.H., Huo S.Y., Shen S.G., Sun H.W. and Wang A.Z. (2005). Chem. J. Chin. Univ 26: 706
Balikungeri A., Pelletier M. and Monnier D. (1977). Inorg. Chem. Acta, 22: 7
Feigl F. (1956). Spot Tests in Organic Analysis. Elsevier, New York, 208
J. Aveston, (1969). J. Chem Soc(A), 273 .
Shan J.H. and Liu T.Y. (1994). Acta Chim. Sinica 52: 1140
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huo, SY., Song, CY. & Zhen, YJ. Oxidation of β-alanine by Ag(III) complex in alkaline medium: a kinetic and mechanism study. Transition Met Chem 32, 936–939 (2007). https://doi.org/10.1007/s11243-007-0257-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-007-0257-8