Abstract
Layered plaque, a signature of previous plaque destabilization and healing, is a known predictor for rapid plaque progression; however, the mechanism of which is unknown. The aim of the current study was to compare the level of vascular inflammation and plaque vulnerability in layered plaques to investigate possible mechanisms of rapid plaque progression. This is a retrospective, observational, single-center cohort study. Patients who underwent both coronary computed tomography angiography (CTA) and optical coherence tomography (OCT) for stable angina pectoris (SAP) were selected. Plaques were defined as any tissue (noncalcified, calcified, or mixed) within or adjacent to the lumen. Perivascular inflammation was measured by pericoronary adipose tissue (PCAT) attenuation at the plaque levels on CTA. Features of plaque vulnerability were assessed by OCT. Layered plaques were defined as plaques presenting one or more layers of different optical densities and a clear demarcation from underlying components on OCT. A total of 475 plaques from 195 patients who presented with SAP were included. Layered plaques (n = 241), compared with non-layered plaques (n = 234), had a higher level of vascular inflammation (-71.47 ± 10.74 HU vs. -73.69 ± 10.91 HU, P = 0.026) as well as a higher prevalence of the OCT features of plaque vulnerability, including lipid-rich plaque (83.8% vs. 66.7%, P < 0.001), thin-cap fibroatheroma (26.1% vs. 17.5%, P = 0.026), microvessels (61.8% vs. 34.6%, P < 0.001), and cholesterol crystals (38.6% vs. 25.6%, P = 0.003). Layered plaque was associated with a higher level of vascular inflammation and a higher prevalence of plaque vulnerability, which might play an important role in rapid plaque progression.
Clinical trial registration: https://classic.clinicaltrials.gov/ct2/show/NCT04523194.
Graphical abstract
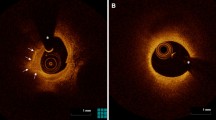
Level of vascular inflammation and plaque vulnerability in patients with versus without layered plaque phenotype. In patients with stable angina pectoris, layered plaques had a higher level of pericoronary adipose tissue attenuation indicating a higher level of perivascular inflammation and a higher prevalence of optical coherence tomography features of plaque vulnerability




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- CAD:
-
Coronary artery disease
- CT:
-
Computed tomography
- CTA:
-
Coronary computed tomography angiography
- DS:
-
Diameter stenosis
- ECG:
-
Electrocardiography
- FCT:
-
Fibrous cap thickness
- HU:
-
Hounsfield unit
- LL:
-
Lesion length
- MLD:
-
Minimal lumen diameter
- OCT:
-
Optical coherence tomography
- PCAT:
-
Pericoronary adipose tissue
- RD:
-
Reference vessel diameter
- SAP:
-
Stable angina pectoris
- TCFA:
-
Thin-cap fibroatheroma
References
Mann J, Davies MJ (1999) Mechanisms of progression in native coronary artery disease: Role of healed plaque disruption. Heart 82:265–268. https://doi.org/10.1136/hrt.82.3.265
Burke AP, Kolodgie FD, Farb A et al (2001) Healed plaque ruptures and sudden coronary death evidence that subclinical rupture has a role in plaque progression. Circulation 103:934–940. https://doi.org/10.1161/01.cir.103.7.934
Virmani R, Kolodgie FD, Burke AP et al (2000) Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler Thromb Vasc Biol 20:1262–1275. https://doi.org/10.1161/01.atv.20.5.1262
Otsuka F, Joner M, Prati F et al (2014) Clinical classification of plaque morphology in coronary disease. Nat Rev Cardiol 11:379–389. https://doi.org/10.1038/nrcardio.2014.62
Ross R. Atherosclerosis—an inflammatory disease. N Engl J Med 340:115–26. https://doi.org/10.1056/NEJM199901143400207
Antoniades C, Antonopoulos AS, Deanfield J (2020) Imaging residual inflammatory cardiovascular risk. Eur Heart J 41:748–758. https://doi.org/10.1093/eurheartj/ehz474
Antonopoulos AS, Sanna F, Sabharwal N et al (2017) Detecting human coronary inflammation by imaging perivascular fat. Sci Transl Med 9:eaal2658. https://doi.org/10.1126/scitranslmed.aal2658
Oikonomou EK, Marwan M, Desai MY et al (2018) Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): a post-hoc analysis of prospective outcome data. The Lancet 392:929–939. https://doi.org/10.1016/S0140-6736(18)31114-0
Stone GW, Maehara A, Lansky AJ et al (2011) A prospective natural-history study of coronary atherosclerosis. N Engl J Med 364:226–235. https://doi.org/10.1056/nejmoa1002358
Xing L, Higuma T, Wang Z et al (2017) Clinical significance of lipid-rich plaque detected by optical coherence tomography: a 4-year follow-up study. J Am Coll Cardiol 69:2502–2513. https://doi.org/10.1016/j.jacc.2017.03.556
Aguirre AD, Arbab-Zadeh A, Soeda T et al (2021) Optical coherence tomography of plaque vulnerability and rupture: JACC focus seminar part 1/3. J Am Coll Cardiol 78:1257–1265. https://doi.org/10.1016/j.jacc.2021.06.050
Araki M, Park SJ, Dauerman HL et al (2022) Optical coherence tomography in coronary atherosclerosis assessment and intervention. Nat Rev Cardiol 19:684–703. https://doi.org/10.1038/s41569-022-00687-9
Lin A, Nerlekar N, Munnur RK et al (2020) Cholesterol crystal-induced coronary inflammation: insights from optical coherence tomography and pericoronary adipose tissue computed tomography attenuation. J Cardiovasc Comput Tomogr 14(3):277–278. https://doi.org/10.1016/j.jcct.2019.11.011
Yuki H, Sugiyama T, Suzuki K et al (2023) Coronary inflammation and plaque vulnerability: a coronary computed tomography and optical coherence tomography study. Circ Cardiovasc Imaging 16:E014959. https://doi.org/10.1161/CIRCIMAGING.122.014959
Sugiyama T, Kanaji Y, Hoshino M et al (2023) Relationship of OCT-defined plaque characteristics with CCTA-derived coronary inflammation and CMR-derived global coronary flow reserve in patients with acute coronary syndrome. PLoS One 18:e0286196. https://doi.org/10.1371/journal.pone.0286196
Abbara S, Blanke P, Maroules CD et al (2016) SCCT guidelines for the performance and acquisition of coronary computed tomographic angiography: a report of the society of cardiovascular computed tomography guidelines committee: endorsed by the north American society for cardiovascular imaging (NASCI). J Cardiovasc Comput Tomogr 10:435–449. https://doi.org/10.1016/j.jcct.2016.10.002
Lee SE, Sung JM, Andreini D et al (2022) Association between changes in perivascular adipose tissue density and plaque progression. JACC Cardiovasc Imaging 15:1760–1767. https://doi.org/10.1016/j.jcmg.2022.04.016
Ferencik M, Mayrhofer T, Bittner DO et al (2018) Use of high-risk coronary atherosclerotic plaque detection for risk stratification of patients with stable chest pain: a secondary analysis of the promise randomized clinical trial. JAMA Cardiol 3:144–152. https://doi.org/10.1001/jamacardio.2017.4973
Goeller M, Achenbach S, Cadet S et al (2018) Pericoronary adipose tissue computed tomography attenuation and high-risk plaque characteristics in acute coronary syndrome compared with stable coronary artery disease. JAMA Cardiol 3:858–863. https://doi.org/10.1001/jamacardio.2018.1997
Araki M, Sugiyama T, Nakajima A et al (2022) Level of vascular inflammation is higher in acute coronary syndromes compared with chronic coronary disease. Circ Cardiovasc Imaging 15:E014191. https://doi.org/10.1161/CIRCIMAGING.122.014191
Nakajima A, Sugiyama T, Araki M et al (2022) Plaque rupture, compared with plaque erosion, is associated with a higher level of pancoronary inflammation. JACC Cardiovasc Imaging 15:828–839. https://doi.org/10.1016/j.jcmg.2021.10.014
Fracassi F, Crea F, Sugiyama T et al (2019) Healed culprit plaques in patients with acute coronary syndromes. J Am Coll Cardiol 73:2253–2263. https://doi.org/10.1016/j.jacc.2018.10.093
Shimokado A, Matsuo Y, Kubo T et al (2018) In vivo optical coherence tomography imaging and histopathology of healed coronary plaques. Atherosclerosis 275:35–42. https://doi.org/10.1016/j.atherosclerosis.2018.05.025
Oikonomou EK, Antoniades C (2019) The role of adipose tissue in cardiovascular health and disease. Nat Rev Cardiol 16:83–99. https://doi.org/10.1038/s41569-018-0097-6
Guglielmo M, Lin A, Dey D et al (2021) Epicardial fat and coronary artery disease: role of cardiac imaging. Atherosclerosis 321:30–38. https://doi.org/10.1016/j.atherosclerosis.2021.02.008
Antonopoulos AS, Tousoulis D (2017) The molecular mechanisms of obesity paradox. Cardiovasc Res 113:1074–1086. https://doi.org/10.1093/cvr/cvx106
Tzolos E, Williams MC, McElhinney P et al (2022) Pericoronary adipose tissue attenuation, low-attenuation plaque burden, and 5-year risk of myocardial infarction. JACC Cardiovasc Imaging 15:1078–1088. https://doi.org/10.1016/j.jcmg.2022.02.004
Russo M, Fracassi F, Kurihara O, et al (2020) Healed plaques in patients with stable angina pectoris. Arterioscler Thromb Vasc Biol 1587–1597. https://doi.org/10.1161/ATVBAHA.120.314298
Kimura S, Isshiki A, Shimizu M et al (2023) Clinical significance of coronary healed plaques in stable angina pectoris patients undergoing percutaneous coronary intervention. Circ J. https://doi.org/10.1253/circj.cj-23-0031
Araki M, Yonetsu T, Kurihara O et al (2021) Predictors of rapid plaque progression: an optical coherence tomography study. JACC Cardiovasc Imaging 14:1628–1638. https://doi.org/10.1016/j.jcmg.2020.08.014
Uemura S, Ishigami K, Soeda T et al (2012) Thin-cap fibroatheroma and microchannel findings in optical coherence tomography correlate with subsequent progression of coronary atheromatous plaques. Eur Heart J 33:78–85. https://doi.org/10.1093/eurheartj/ehr284
Nakamura S, Inami S, Murai K et al (2014) Relationship between cholesterol crystals and culprit lesion characteristics in patients with stable coronary artery disease: an optical coherence tomography study. Clin Res Cardiol 103:1015–1021. https://doi.org/10.1007/s00392-014-0748-5
Kolodgie FD, Gold HK, Burke AP et al (2003) Intraplaque hemorrhage and progression of coronary atheroma. N Engl J Med 349:2346–2425. https://doi.org/10.1056/NEJMoa035655
Sluimer JC, Kolodgie FD, Bijnens APJJ et al (2009) Thin-walled microvessels in human coronary atherosclerotic plaques show incomplete endothelial junctions. relevance of compromised structural integrity for intraplaque microvascular leakage. J Am Coll Cardiol 53:1517–1527. https://doi.org/10.1016/j.jacc.2008.12.056
Virmani R, Kolodgie FD, Burke AP et al (2005) Atherosclerotic plaque progression and vulnerability to rupture: angiogenesis as a source of intraplaque hemorrhage. Arterioscler Thromb Vasc Biol 25:2054–2061. https://doi.org/10.1161/01.ATV.0000178991.71605.18
Abela GS, Aziz K (2006) Cholesterol crystals rupture biological membranes and human plaques during acute cardiovascular events - A novel insight into plaque rupture by scanning electron microscopy. Scanning 28:1–10. https://doi.org/10.1002/sca.4950280101
Crea F, Liuzzo G (2013) Pathogenesis of acute coronary syndromes. J Am Coll Cardiol 61:1–11. https://doi.org/10.1016/j.jacc.2012.07.064
Funding
Dr. Ik-Kyung Jang’s research has been supported by Mrs. Gillian Gray through the Allan Gray Fellowship Fund in Cardiology and Mukesh and Priti Chatter through the Chatter Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
Dr. Niida received a grant from the Japan Heart Foundation/Bayer Yakuhin Research Grant Abroad. Outside the present study, Dr. Dey has received software royalties from Cedars-Sinai Medical Center and has a patent. Dr. Ferencik has received consulting fees from Siemens Healthineers, HeartFlow, and Elucid and stock options from Elucid. The remaining authors have nothing to disclose. Dr. Jang has received educational grants from Abbott Vascular and a consulting fee from Svelte Medical Systems.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Layered plaque is a predictor for rapid plaque progression, the mechanism of which is unknown.
• The level of vascular inflammation and plaque vulnerability can be assessed by coronary computed tomography and optical coherence tomography, respectively.
• A level of vascular inflammation and a prevalence of the OCT features of plaque vulnerability were higher in layered plaques, compared with non-layered plaques.
• High levels of vascular inflammation and plaque vulnerability in layered plaque may explain the mechanism of rapid plaque progression.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Niida, T., Kinoshita, D., Suzuki, K. et al. Layered plaque is associated with high levels of vascular inflammation and vulnerability in patients with stable angina pectoris. J Thromb Thrombolysis (2024). https://doi.org/10.1007/s11239-024-02982-3
Accepted:
Published:
DOI: https://doi.org/10.1007/s11239-024-02982-3