Abstract
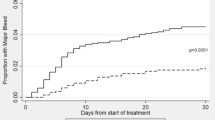
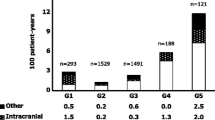
Drugs exhibiting high protein binding have potential increased action in patients with hypoalbuminemia. Rivaroxaban is 92–95% protein bound, but the clinical effects of rivaroxaban in patients with low albumin are largely unknown. The purpose of this study was to evaluate the relationship between albumin and bleeding in rivaroxaban treated patients. This was a retrospective cohort study of hospitalized adults who received rivaroxaban and had an albumin level measured during admission between January and October 2017. Patients who experienced bleeding events while receiving rivaroxaban therapy where compared to those who did not. A multivariable logistic regression model was used to evaluate the association between albumin levels and bleeding events. A total of 368 patients were included; 30 experienced a bleeding event and 338 did not. The mean ± standard deviation albumin level nearest to the time of rivaroxaban initiation was significantly lower in patients who experienced a bleeding event (3.0 ± 0.75 g/dL vs 3.66 ± 0.54 g/dL, p < 0.0001). The multivariable logistic regression model yielded an almost 4.5 fold higher risk of bleeding (adjusted odds ratio 4.405; 95% confidence interval 2.21–9) with any 1 g/dL reduction in albumin. Admission hemoglobin was also associated with bleed risk in the model. Albumin levels were significantly associated with bleed risk in patients receiving rivaroxaban. Albumin levels should be considered when evaluating candidates for rivaroxaban therapy.

Similar content being viewed by others
References
Cabrerizo S, Cuadras D, Gomez-Busto F, Artaza-Artabe I, Marin-Ciancas F, Malafarina V (2015) Serum albumin and health in older people: review and meta-analysis. Maturitas 81:17–27. https://doi.org/10.1016/j.maturitas.2015.02.009
Nicholson JP, Wolmarans MR, Park GR (2000) The role of albumin in critical illness. Br J Anaesth 85:599–610. https://doi.org/10.1093/bja/85.4.599
Tincani E, Mazzali F, Morini L (2002) Hypoalbuminemia as a risk factor for over-anticoagulation. Am J Med 112:247–248. https://doi.org/10.1016/s0002-9343(01)00957-3
Abdelhafiz AH, Myint MP, Taek JA, Wheeldon NM (2009) Anemia, hypoalbuminemia, and renal impairment as predictors of bleeding complications in patients receiving anticoagulation therapy for nonvalvular atrial fibrillation: a secondary analysis. Clin Ther 31:1534–1539. https://doi.org/10.1016/j.clinthera.2009.07.015
Kubitza D, Roth A, Becka M, Alatrach A, Halabi A, Hinrichsen H, Mueck W (2013) Effect of hepatic impairment on the pharmacokinetics and pharmacodynamics of a single dose of rivaroxaban, an oral, direct Factor Xa inhibitor. Br J Clin Pharmacol 76:89–98. https://doi.org/10.1111/bcp.12054
Mueck W, Stampfuss J, Kubitza D, Becka M (2014) Clinical pharmacokinetic and pharmacodynamics profile of rivaroxaban. Clin Pharmacokinet 53:1–16. https://doi.org/10.1007/s40262-013-0100-7
Xarelto (rivaroxaban) tablets [package insert] (2016). Janssen Pharmaceutical Company, Titusville
Kearon C, Akl EA, Ornelas J et al (2016) Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest 149:315–352. https://doi.org/10.1016/j.chest.2015.11.026
January CT, Wann LS, Alpert JS et al (2014) 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol 64:e1–e76. https://doi.org/10.1016/j.jacc.2014.03.022
Writing Group Members, January CT, Wann LS, Calkins H et al (2019) 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm 16:e66–e93. https://doi.org/10.1016/j.hrthm.2019.01.024
Lip GYH, Banerjee A, Boriani G et al (2018) Antithrombotic therapy for atrial fibrillation: CHEST guideline and expert panel report. Chest 154:1121–2120. https://doi.org/10.1016/j.chest.2018.07.040
Kaatz S, Ahmad D, Spyropoulos AC et al (2015) Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost 13:2119–2126. https://doi.org/10.1111/jth.13140
Patel MR, Mahaffey KW, Garg J et al (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365:883–891. https://doi.org/10.1056/NEJMoa1009638
Investigators EINSTEIN, Bauersachs R, Berkowitz SD et al (2010) Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med 363:2499–2510. https://doi.org/10.1056/NEJMoa1007903
Einstein–PE Investigators, Buller HR, Prins MH et al (2012) Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med 366:1287–1297. https://doi.org/10.1056/NEJMoa1113572
Stollberger C, Bastovansky A, Finsterer J (2015) Fatal intracerebral bleeding under rivaroxaban. Int J Cardiol 201:110–112. https://doi.org/10.1016/j.ijcard.2015.06.081
Hankey GJ, Stevens SR, Piccini JP et al (2014) Intracranial hemorrhage among patients with atrial fibrillation anticoagulated with warfarin or rivaroxaban: the rivaroxaban once daily, oral, direct factor Xa inhibition compared with vitamin K antagonism for prevention of stroke and embolism trial in atrial fibrillation. Stroke 45:1304–1312. https://doi.org/10.1161/STROKEAHA.113.004506
Gage B, Yan Y, Milligan P, Waterman AD et al (2006) Clinical classification schemes for predicting hemorrhage: Results from the National Registry of Atrial Fibrillation (NRAF). Am Heart J 151:713–719. https://doi.org/10.1016/j.ahj.2005.04.017
Barletta JF, Bruno JJ, Buckley MS, Cook DJ (2016) Stress ulcer prophylaxis. Crit Care Med 44:1395–1405. https://doi.org/10.1097/CCM.0000000000001872
Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crigns HJ, Lip GY (2010) A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest 138:1093–1100. https://doi.org/10.1378/chest.10-0134
Woodruff AE, Wovkulich MM, Mogle BT, Hassan AK (2018) Association between prothrombin time and bleeding in hospitalized patients receiving rivaroxaban. Am J Health Syst Pharm 75:1783–1789. https://doi.org/10.2146/ajhp180031
Gatta A, Verardo A, Bolognesi M (2012) Hypoalbuminemia. Intern Emerg Med Suppl 3:S193–S199. https://doi.org/10.1007/s11739-012-0802-0
Liang CC, Wang SM, Kuo HL et al (2014) Upper gastrointestinal bleeding in patients with CKD. Clin J Am Soc Nephrol 9:1354–1359. https://doi.org/10.2215/CJN.09260913
Tatami Y, Ishil H, Aoki T et al (2017) Decreased serum albumin predicts bleeding events in patients on antiplatelet therapy after percutaneous coronary intervention. Circ J 81:999–1005. https://doi.org/10.1253/circj.CJ-17-0015
Tung CF, Chow WK, Chang CS, Peng YC, Hu WH (2007) The prevalence and significance of hypoalbuminemia in non-variceal upper gastrointestinal bleeding. Hepatogastroenterology 54:1153–1156
Stokes ME, Ye X, Shah M et al (2011) Impact of bleeding-related complications and/or blood product transfusions on hospital costs in inpatient surgical patients. BMC Health Serv Res 11:135. https://doi.org/10.1186/1472-6963-11-135
Coumadin (warfarin) tablets [package insert] (2011) Bristol-Myers Squibb Company, Princeton
Eliquis (apixaban) tablets [package insert] (2019) Bristol-Myers Squibb Company, Princeton
Pradaxa (dabigatran) tablets [package insert] (2019) Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by EW, CC, AH, LC, MR and AW. The first draft of the manuscript was written by EW, AW, MH and NW and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
This article does not contain nay studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wojakowski, E., Cheruvil, C., Hassan, A. et al. Albumin and bleed risk in rivaroxaban treated patients. J Thromb Thrombolysis 50, 1004–1011 (2020). https://doi.org/10.1007/s11239-020-02092-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-020-02092-w