Abstract
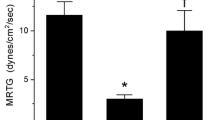
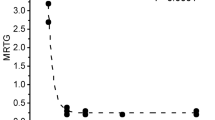
A complication of defibrinogenation therapy with snake venom enzymes such as ancrod is hypofibrinogenemia associated bleeding secondary to no human-derived inhibitor being available to inactivate or diminish the activity of such enzymes. Of interest, ancrod contains a critical histidine residue without which enzymatic activity is inhibited, and carbon monoxide has been demonstrated to inhibit biomolecular function by interacting with histidine moieties in ion channels. We tested the hypothesis that exposure of three different snake venoms containing serine proteases with thrombin-like activity (which included ancrod) to carbon monoxide derived from carbon monoxide releasing molecule-2 would diminish their effects on plasmatic coagulation as assessed by thrombelastography. In the case of the Malayan pit viper and Eastern diamondback rattlesnake venoms, carbon monoxide diminished the effects of thrombin-like activity. In contrast, timber rattlesnake venom demonstrated enhancement of “thrombin-generating” activity with simultaneous loss of thrombin-like activity in response to carbon monoxide exposure. These findings may serve as the rational basis for not just continuing to investigate the potential of snake venom enzymes as clinical defibrinogenating agents, but to also to assess the potential to stop such agents from becoming a catalytic “runaway train” by judicious application of a biochemical “brake” such as carbon monoxide.



Similar content being viewed by others
References
Huang TF, Chang MC, Peng HC, Teng CM (1992) A novel alpha-type fibrinogenase from Agkistrodon rhodostoma snake venom. Biochim Biophys Acta 1160:262–268
Nielsen VG (2016) Ancrod revisited: viscoelastic analyses of the effects of Calloselasma rhodostoma venom on plasma coagulation and fibrinolysis. J Thromb Thrombolysis 42:288–293
Collins JP, Jones JG (1974) Identification of serine and histidine as essential amino-acid residues in the coagulant enzyme ancrod. Eur J Biochem 42:81–87
Wang R, Wu L (2003) Interaction of selective amino acid residues of K(ca) channels with carbon monoxide. Exp Biol Med 228:474–480
Telezhkin V, Brazier SP, Mears R, Müller CT, Riccardi D, Kemp PJ (2011) Cysteine residue 911 in C-terminal tail of human BK(Ca)α channel subunit is crucial for its activation by carbon monoxide. Pflug Arch 461:665–675
Althaus M, Fronius M, Buchäckert Y, Vadász I, Clauss WG, Seeger W, Motterlini R, Morty RE (2009) Carbon monoxide rapidly impairs alveolar fluid clearance by inhibiting epithelial sodium channels. Am J Respir Cell Mol Biol 41:639–650
Markland FS, Damus PS (1971) Purification and properties of a thrombin-like enzyme from the venom of Crotalus adamanteus (Eastern diamondback rattlesnake). J Biol Chem 246:6460–6473
Bonilla CA (1975) Defibrinating enzyme from timber rattlesnake (Crotalus h. horridus) venom: a potential agent for therapeutic defibrination. I. Purification and properties. Thromb Res 6:151–169
Shu YY, Moran JB, Geren CR (1983) A thrombin-like enzyme from timber rattlesnake venom. Biochim Biophys Acta 748:236–244
Nielsen VG, Garza JI (2014) Comparison of the effects of CORM-2, CORM-3 and CORM-A1 on coagulation in human plasma. Blood Coagul Fibrinolysis 25:801–805
Nielsen VG (2016) Iron and carbon monoxide prevent degradation of plasmatic coagulation by thrombin-like activity in rattlesnake venom. Hum Exp Toxicol 35:1116–1122
Nielsen VG, Boyer LV, Redford DT, Ford P (2016) Thrombelastographic characterization of the thrombin-like activity of Crotalus simus and Bothrops asper venoms. Blood Coagul Fibrinolysis. doi:10.1016/j.jacc.2016.07.729
Nielsen VG, Jacobsen WK (2016) Iron modulates the alpha chain of fibrinogen. Biometals 29:235–238
Jara-Oseguera A, Ishida IG, Rangel-Yescas GE, Espinosa-Jalapa N, Pe´rez-Guzma´n JA, Elías-Vinas D, Le Lagadec R, Rosenbaum T, Islas LD (2011) Uncoupling charge movement from channel opening in voltage-gated potassium channels by ruthenium complexes. J Biol Chem 286:16414–16425
Pak O, Bakr AG, Gierhardt M, Albus J, Strielkov I, Kroschel F, Hoeres T, Hecker M, Ghofrani HA, Seeger W, Weissmann N, Sommer N (2016) Effects of carbon monoxide-releasing molecules on pulmonary vasoreactivity in isolated perfused lungs. J Appl Physiol 120:271–281
Levy DE, del Zoppo GJ, Demaerschalk BM, Demchuk AM, Diener HC, Howard G, Kaste M, Pancioli AM, Ringelstein EB, Spatareanu C, Wasiewski WW (2009) Ancrod in acute ischemic stroke: results of 500 subjects beginning treatment within 6 h of stroke onset in the ancrod stroke program. Stroke 40:3796–3803
Acknowledgment
This investigation was supported by the Department of Anesthesiology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nielsen, V.G., Bazzell, C.M. Carbon monoxide releasing molecule-2 inhibition of snake venom thrombin-like activity: novel biochemical “brake”?. J Thromb Thrombolysis 43, 203–208 (2017). https://doi.org/10.1007/s11239-016-1442-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-016-1442-4