Abstract
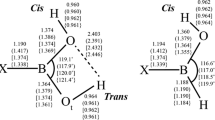
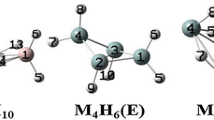
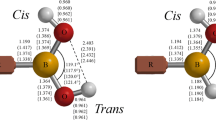
Boroxines have emerged as an important class of compounds with diverse applications in many fields. However, there are limited experimental and/or computational structural and thermal data available for these compounds or for their corresponding boronic acids. In this investigation, we report structural parameters for a variety of aliphatic monosubstituted boroxines (RH2B3O3) and their enthalpies of formation via the dehydration reactions from the boronic acids (R–B(OH)2), i.e., R–B(OH)2 + 2H–B(OH)2 → RH2B3O3 + 3H2O. Equilibrium geometries of all the boronic acids and monosubstituted boroxines involved in this article were obtained using second-order Møller–Plesset perturbation theory with the Dunning–Woon aug-cc-pVDZ and aug-cc-pVTZ basis sets; heats of formation were calculated at the G3 level of theory. The specific substituents, R, include all the second- and third-period hydrides, as well as a selection of electron-donating/electron-withdrawing groups.


Similar content being viewed by others
References
Hall DG (2005) Boronic acids: preparation and applications in organic synthesis and medicine, 1st edn. Weinheim, Wiley-VCH Verlag
Korich AL, Iovine PM (2010) Dalton Trans 39:1423–1431
Dembitsky VM, Quntar AA, Srebnik M (2004) Mini Rev Med Chem 4:1001–1018
Baker SJ, Tomsho JW, Benkovic SJ (2011) Chem Rev Soc. doi:10.1039/c0cs00131g
Bhat KL, Markham GD, Larkin JD, Bock CW (2011) J Phys Chem A 115:7785–7793
Yao L, Zeng X, Ge M, Wang D (2007) J Mol Struct 841:104–109
Porter RF, Gupta SK (1964) J Phys Chem 68:2732–2733
Chase MW (1998) NIST—JANAF thermochemical tables (Journal of Physical and Chemical Reference Data Monograph No. 9). American Institute of Physics
Ruscic B, Pinson RE, Morton ML, von Laszevski G, Bittner SJ, Nijsure SG, Amin KA, Minkoff M, Wagner AF (2004) J Phys Chem A 108:9979–9997
Ruscic B, Pinson RE, von Laszevski G, Kodeboyina D, Burcat A, Leahy D, Montoya D, Wagner AF (2005) J Phys Conf Ser 16:561–570
Gurvich LV, Veyts IV, Alcock CB (1994) Thermodynamic properties of individual substances, vol 3 (Parts 1 and 2) elements B, Al, Ga, In, Tl, Be, Mg, Ca, Sr, and Ba and their compounds. CRC Press Inc, Boca Raton
Tokunaga Y, Ueno H, Shimomura Y, Seo T (2002) Heterocycles 57:787–790
Bock CW, Larkin JD (2012) Comput Theor Chem 986:35–42
Ghiasi R (2008) J Mol Struct (THEOCHEM) 853:77–81
Guest MF, Pedley JB, Horn M (1969) J Chem Thermodyn 1:345–352
Porter RF, Gupta SK (1964) J Phys Chem 68:280–289
Tokunaga Y, Ueno H, Shimomura Y (2007) Heterocycles 74:219–223
Møller C, Plesset MS (1934) Phys Rev 46:618–622
Dunning TH Jr (1989) J Chem Phys 90:1007–1023
Woon DE, Dunning TH Jr (1993) J Chem Phys 98:1358–1371
Kendall RA, Dunning TH Jr (1992) J Chem Phys 96:6796–6806
Peterson KA, Woon DE, Dunning TH Jr (1994) J Chem Phys 100:7410–7415
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery J, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts RE, Stratmann O, Yazyev AJ, Austin R, Cammi C, Pomelli JW, Ochterski R, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2003) G03, R B.02 ed, Gaussian Inc., Wallingford, CT
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery J, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts RE, Stratmann O, Yazyev AJ, Austin R, Cammi C, Pomelli JW, Ochterski R, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) G09 Gaussian Inc., Wallingford, CT
Curtiss LA, Raghavachari K, Redfern PC, Rassolov V, Pople JA (1998) J Chem Phys 109:7764–7776
Curtiss LA, Raghavachari K (2002) Theor Chem Acc 108:61–70
Carpenter JE, Weinhold F (1988) J Mol Struct (THEOCHEM) 169:41–62
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Foster JP, Weinhold F (1980) J Am Chem Soc 102:7211–7218
Reed AE, Weinstock RB, Weinhold F (1985) J Chem Phys 83:735–746
Snyder HR, Kuck JA, Johnson JR (1938) J Am Chem Soc 60:105–111
Wagner M, van Eikema Homes NJR, Ndth H, Schleyer PVR (1995) Inorg Chem 34:607–614
Larkin JD, Bhat KL, Markham GD, Brooks BR, Schaefer HF, Bock CW III (2006) J Phys Chem A 110:10633–10642
Larkin JD, Bhat KL, Markham GD, Brooks BR, Lai JH, Bock CW (2007) J Phys Chem A 111:6489–6500
Boggs JE, Cordell FR (1981) J Mol Struct (THEOCHEM) 76:329–347
So SP (1982) J Mol Struct (THEOCHEM) 89:255–258
Duan X, Linder DP, Page M, Soto MR (1999) J Mol Struct (THEOCHEM) 465:231–242
Stefani D, Pashalidis I, Nicolaides AV (2008) J Mol Struct (THEOCHEM) 853:33–38
Bent HA (1961) Chem Rev 61:275–311
Curtiss LA, Redfern PC, Raghavachari K (2005) J Chem Phys 123:124107
Grant DJ, Dixon DA (2009) J Phys Chem A 113:777–787
Porter RF, Bidinosti DR, Watterson VF (1962) J Chem Phys 36:2104–2108
Chang CH, Porter RF, Bauer SH (1969) Inorg Chem 8:1689–1693
Beckmann J, Dakternieks D, Duthie A, Lim AEK, Tiekink ERT (2001) J Organomet Chem 633:149–156
Domenicano A, Vaciago A, Coulson C (1975) Acta Cryst Sect B 31:1630–1641
Tasi G, Iszák R, Matisz G, Császár AG, Kállay M, Ruscic B, Stanton JF (2006) ChemPhysChem 7:1664–1667
Hansch C, Leo A (1979) Substituent constants for correlation analysis in chemistry and biology. Wiley-Interscience, New York
Schlegel HB, Harris SJ (1994) J Phys Chem 98:11178–11180
Karton A, Martin JML (2007) J Phys Chem A 111:5936–5944
Acknowledgments
This research was supported in part by the National Science Foundation through XSEDE resources provided by the XSEDE Science Gateways program.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rao, N.Z., Larkin, J.D. & Bock, C.W. A computational investigation of monosubstituted boroxines(RH2B3O3): structure and formation. Struct Chem 26, 1151–1162 (2015). https://doi.org/10.1007/s11224-015-0577-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-015-0577-9