Abstract
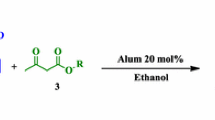
A highly atom-economic, one-pot synthesis of functionalized piperidines is reported by a multi-component condensation reaction between β-keto ester, two equivalents of aromatic aldehyde, and two equivalents of amine in the presence of a catalytic amount of ZrCl4. The advantages of this protocol are good yields, short reaction time, mild reaction conditions, no need for column chromatography, more readily available, inexpensive catalyst, and simple work-up procedure.
Graphical Abstract



Similar content being viewed by others
References
C.C.A. Cariou, G.J. Clarkson, M. Shipman, J. Org. Chem. 73, 9762 (2008)
S.M. Kupchan, Y. Komoda, W.A. Court, G.J. Thomas, R.M. Smith, A. Karim, C.J. Gilmore, R.C. Haltivanger, R.F. Bryan, J. Am. Chem. Soc. 94, 1354 (1972)
R.W. Armstrong, A.P. Combs, P.A. Tempest, S.D. Brown, T.A. Keating, Acc. Chem. Res. 29, 123 (1996)
G. Ren, J. Zhang, Z. Duan, M. Cui, Y. Wu, Aust. J. Chem. 62, 75 (2009)
C. Mukhopadhyay, A. Datta, R.J. Butcher, Tetrahedron Lett. 50, 4246 (2009)
C. Mukhopadhyay, P.K. Tapaswi, M.G.B. Drew, Tetrahedron Lett. 51, 3944 (2010)
A.D. Elbein, R. Molyneux, in Alkaloids Chemical and Biological Perspectives, ed. by S.W. Pelletier (Wiley, New York, 1987), p. 57
D. O’Hagan, Nat. Prod. Rep. 17, 435 (2000)
J.W. Daly, T.F. Spande, H.M. Garraffo, J. Nat. Prod. 68, 1556 (2005)
K. Kamei, N. Maeda, R. Katswagi-Ogino, M. Koyamaa, M. Nakajima, T. Tatsuoka, T. Ohno, T. Inone, Bioorg. Med. Chem. Lett. 15, 2990 (2005)
J.P. Yevich, F.D. Yocca, Curr. Med. Chem. 4, 295 (1997)
Y. Yamanishi, H. Ogura, T. Kosasa, Tanpakushitsu Kakusan Koso 45, 1047 (2000)
S. Targum, J. Zboroaski, M. Henry, P. Schmitz, T. Sebree, B. Wallin, Eur. Neuropsychopharmacol. 5, 4 (1995)
A. Schotte, P.F.M. Janssen, W. Gommeren, W.H.M.L. Luyten, P. van Gompel, A.S. Lasage, K. De Loore, J.E. Leysen, Psycholpharmacology 124, 57 (1996)
Y. Zhou, V.E. Gregor, B.K. Ayida, G.C. Winters, Z. Sun, D. Murphy, G. Haley, D. Bailey, J.M. Froelich, S. Fish, S.E. Webber, T. Hermann, D. Wall, Bioorg. Med. Chem. Lett. 17, 1206 (2007)
S. Petit, J.P. Nallet, M. Guillard, J. Dreux, R. Chermat, M. Poncelet, C. Bulach, P. Simon, C. Fontaine, M. Barthelmebs, J.L. Imbs, Eur. J. Med. Chem. 26, 19 (1991)
M. Misra, S.K. Pandey, V.P. Pandey, J. Pandey, R. Tripathi, R.P. Tripathi, Bioorg. Med. Chem. 17, 625 (2009)
H. Bin, A.M. Crider, J.P. Stables, Eur. J. Med. Chem. 36, 265 (2001)
P. Chand, P.L. Kotian, A. Dehghani, Y. El-Kattan, T. Lin, T.L. Hutchison, Y.S. Babu, S. Bantia, A.J. Elliott, J.A. Montgomery, J. Med. Chem. 44, 4379 (2001)
P.E. Goss, M.A. Baker, J.P. Carver, J.W. Dennis, Clin. Cancer Res. 1, 935 (1995)
J.L. Treadway, P. Mendys, D.J. Hoover, Expert Opin. Investig. Drugs 10, 439 (2001)
F. Yu, X. Zhang, Y. Jiang, Chin. J. Chem. 29, 1873 (2011)
H. Lu, Y. Chen, B. Yang, Q. You, Acta Pharm. Sin. B. 1, 240 (2011)
A.T. Khan, M. Lal, M.M. Khan, Tetrahedron Lett. 51, 4419 (2010)
A.T. Khan, T. Parvin, L.H. Choudhury, J. Org. Chem. 73, 8398 (2008)
R. Ramachandran, S. Jayanthi, Y.T. Jeong, Tetrahedron 68, 363 (2012)
A.T. Khan, M.M. Khan, K.K.R. Bannuru, Tetrahedron 66, 7762 (2010)
H.J. Wang, L.P. Mo, Z.H. Zhang, ACS. Comb. Sci. 13, 181 (2011)
G. Brahamachari, S. Das, Tetrahedron Lett. 53, 1479 (2012)
S. Pal, L.H. Choudhury, T. Parvin, Mol. Divers. (2012). doi:10.1007/s11030-011-9339-9
P.A. Clarke, A.V. Zaytzev, A.C. Whitwood, Tetrahedron Lett. 48, 5209 (2007)
P.A. Clarke, A.V. Zaytzev, A.C. Whitwood, Synthesis 3530 (2008)
S.V. Goswami, P.B. Thorat, S.R. Bhusare, Heterocycl. Commun. 18, 245 (2012)
B. Umamahesh, V. Sathesh, G. Ramachandran, M. Sathishkumar, K. Sathiyanarayanan, Catal. Lett. 142, 895 (2012)
M. R. Mohammad Shafiee, B. Hojati Najafabadi, M. Ghashang, Res. Chem. Intermed. doi 10.1007/s11164-012-0878-1
A.K. Chakraborti, A. Kondaskar, Tetrahedron Lett. 44, 8315 (2003)
S. Singh, C.D. Duffy, S.T.A. Shah, P.J. Guiry, J. Org. Chem. 73, 6429 (2008)
M. Raghu, C.S. Reddy, Indian J. Chem. B. 48, 295 (2009)
M.T. Maghsoodlou, G. Marandi, N. Hazeri, A. Aminkhani, R. Kabiri, Tetrahedron Lett. 48, 3197 (2007)
M. Ziyaadini, N. Hazeri, M.T. Maghsoodlou, S.M. Habibi-Khorassani, A.C. Willis, Tetrahedron Lett. 52, 5774 (2011)
F. Rostami-Charati, M.T. Maghsoodlou, S.M. Habibi-Khorassani, M. Makha, Tetrahedron Lett. 49, 343 (2008)
M.T. Maghsoodlou, S.M. Habibi-Khorassani, A. Moradi, N. Hazeri, A. Davodi, S.S. Sajadikhah, Tetrahedron 67, 8492 (2011)
S.S. Sajadikhah, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, S.J. Shams-Najafi, Monatsh. Chem. 143, 939 (2012)
S.S. Sajadikhah, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, A.C. Willis, Chin. Chem. Lett. 23, 569 (2012)
M. Lashkari, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, S.S. Sajadikhah, R. Doostmohamadi, Synth. Commun. (2011). doi:10.1080/00397911.601534
S.S. Sajadikhah, N. Hazeri, M.T. Maghsoodlou, S.M. Habibi-Khorassani, A. Beigbabaei, M. Lashkari, J. Chem. Res. 36, 463 (2012)
S.S. Sajadikhah, N. Hazeri, M.T. Maghsoodlou, S.M. Habibi-Khorassani, A.C. Willis, Res. Chem. Intermed. doi:10.1007/s11164-012-0997-8
N. Hazeri, M.T. Maghsoodlou, S.M. Habibi-Khorassani, J. Aboonajmi, S.S. Sajadikhah, J. Chin. Chem. Soc. 60, 355 (2013)
Acknowledgment
We gratefully acknowledge the funding support received for this project from the Research Council of the University of Sistan and Baluchestan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aboonajmi, J., Mousavi, M.R., Maghsoodlou, M.T. et al. ZrCl4 as an efficient catalyst for one-pot synthesis of highly functionalized piperidines via multi-component organic reactions. Res Chem Intermed 41, 1925–1934 (2015). https://doi.org/10.1007/s11164-013-1320-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-013-1320-z