Abstract
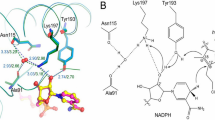
The phycobilin: Cysteine-84-phycobiliprotein lyase, CpeS1, catalyzes phycocyanobilin (PCB) and phycoerythrobilin attachment to nearly all cysteine-84 (consensus sequence) binding sites of phycoerythrin, phycoerythrocyanin, phycocyanin and allophycocyanin (Zhao et al. (2007) Proc Natl Acad Sci 104:14300–14305). We now show that CpeS1 can bind PCB, as assayed by Ni2+ chelating affinity chromatography. Binding is rapid, and the chromophore is bound in an extended conformation similar to that in phycobiliproteins but only poorly fluorescent. Upon addition of apo-biliproteins, the chromophore is transferred to the latter much slower (∼1 h), indicating that chromophorylated CpeS1 is an intermediate in the enzymatic reaction. In addition, imidazole is bound to PCB, as shown by mass spectroscopy of tryptic digests of the intermediate CpeS1–PCB complex.



Similar content being viewed by others
Abbreviations
- CPC:
-
Phycocyanin
- CpcB:
-
Apo-β-CPC
- CpeS1:
-
Phycobilin
- Cys84:
-
Phycobiliprotein lyase
- KPB:
-
Potassium phosphate buffer
- PCB:
-
Phycocyanobilin
- PEC:
-
Phycoerythrocyanin
- PecB:
-
Apo-β-PEC
References
Berkelman T, Lagarias JC (1986) Visualization of bilin-linked peptides and proteins in polyacrylamide gels. Anal Biochem 156:194–201
Bhoo S, Davis SJ, Walker J, Karniol B, Vierstra RD (2001) Bacteriophytochromes are photochromic histidine kinases using a biliverdin chromophore. Nature 414:776–779
Böhm S, Endres S, Scheer H, Zhao K-H (2007) Biliprotein chromophore attachment: chaperone-like function of PecE-subunit of α-phycoerythrocyanin lyase. J Biol Chem 282:25357–25366
Borucki B, Otto H, Rottwinkel G, Hughes J, Heyn MP, Lamparter T (2003) Mechanism of Cph1 phytochrome assembly from stopped-flow kinetics and circular dichroism. Biochemistry 42:13684–13697
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Fairchild CD, Glazer AN (1994) Oligomeric structure, enzyme kinetics, and substrate specificity of the phycocyanin alpha subunit phycocyanobilin lyase. J Biol Chem 269:8686–8694
Fairchild CD, Zhao J, Zhou J, Colson SE, Bryant DA, Glazer AN (1992) Phycocyanin α-subunit phycocyanobilin lyase. Proc Natl Acad Sci USA 89:7017–7021
Gantt B, Grabowski B, Cunningham FX (2003) Antenna systems of red algae: phycobilisomes with photosystem II and chlorophyll complexes with photosystem I. In: Green B, Parson W (eds) Light-harvesting antennas in photosynthesis. Kluwer, Dordrecht pp 307–322
Glazer AN (1994) Adaptive variations in phycobilisome structure. Adv Mol Cell Biol 10:119–149
Grossman AR, Schaefer MR, Chiang GG, Collier JL (1993) The phycobilisome, a light-harvesting complex responsive to environmental conditions. Microbiol Rev 57:725–749
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriopharge T4. Nature 227:680–685
Lamparter T, Carrascal M, Michael N, Martinez E, Rottwinkel G, Abian J (2004) The biliverdin chromophore binds covalently to a conserved cysteine residue in the N-terminus of Agrobacterium phytochrome Agp1. Biochemistry 43:3659–3669
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbour Laboratory Press, New York
Schluchter WM, Glazer AN (1999) Biosynthesis of phycobiliproteins in cyanobacteria. In: Peschek GA, Löffelhardt W, Schmetterer G (eds) The phototrophic prokaryotes, Kluwer/Plenum Press, New York, pp 83–95
Shen G, Saunee NA, Gallo E, Begovic Z, Schluchter WM, Bryant DA (2004) Identification of novel phycobiliprotein lyases in cyanobacteria. In: Niederman RA, Blankenship RE, Frank H, Robert B, van Grondelle R (eds) PS 2004 light-harvesting systems workshop, Saint Adele, Québec, Canada, pp 14–15
Shen G, Saunee NA, Williams SR, Gallo EF, Schluchter WM, Bryant DA (2006) Identification and characterization of a new class of bilin lyase: the cpcT gene encodes a bilin lyase responsible for attachment of phycocyanobilin to Cys-153 on the beta subunit of phycocyanin in Synechococcus sp. PCC 7002. J Biol Chem 281:17768–17778
Storf M, Parbel A, Meyer M, Strohmann B, Scheer H, Deng M, Zheng M, Zhou M, Zhao K-H (2001) Chromophore attachment to biliproteins: specificity of PecE/PecF, a lyase/isomerase for the photoactive 31-Cys-α84-phycoviolobilin chromophore of phycoerythrocyanin. Biochemistry 40:12444–12456
Wu S-H, Lagarias JC (2000) Defining the bilin lyase domain: lessons from the extended phytochrome superfamily. Biochemistry 39:13487–13495
Zhao K-H, Deng M-G, Zheng M, Zhou M, Parbel A, Storf M, Meyer M, Strohmann B, Scheer H (2000) Novel activity of a phycobiliprotein lyase: both the attachment of phycocyanobilin and the isomerization to phycoviolobilin are catalyzed by PecE and PecF. FEBS Lett 469:9–13
Zhao K-H, Zhu JP, Song B, Zhou M, Storf M, Böhm S, Bubenzer C, Scheer H (2004) Non-enzymatic chromophore attachment in biliproteins:conformational control by the detergent Triton X-100. Biochim Biophys Acta 1657:131–145
Zhao K-H, Wu D, Zhou M, Zhang L, Böhm S, Bubenzer C, Scheer H (2005) Amino acid residues associated with enzymatic activity of the isomerizing phycoviolobilin-lyase PecE/F. Biochemistry 44:8126–8137
Zhao K-H, Su P, Li JA, Tu JM, Zhou M, Bubenzer C, Scheer H (2006a) Chromophore attachment to phycobiliprotein ß-subunits: phycocyanobilin:cysteine-ß84 phycobiliprotein lyase activity of CpeS-like protein from Anabaena sp. PCC7120. J Biol Chem 281:8573–8581
Zhao K-H, Wu D, Zhang L, Zhou M, Böhm S, Bubenzer C, Scheer H (2006b) Chromophore attachment in phycocyanin: functional amino acids of phycocyanobilin:(-phycocyanin lyase and evidence for chromophore binding. FEBS J 273:1262–1274
Zhao K-H, Su P, Tu JM, Wang X, Liu H, Plöscher M, Eichacker L, Yang B, Zhou M, Scheer M (2007) Phycobilin: cystein-84 biliprotein lyase, a near-universal lyase for cysteine-84-binding sites in cyanobacterial phycobiliproteins. Proc Natl Acad Sci USA 104:14300–14305
Acknowledgments
HS and KHZ are grateful to Volkswagen Stiftung for the Partnership (I/77900). HS is grateful to Deutsche Forschungsgemeinschaft for the grant (SFB 533, TPA1), KHZ to Program for New Century Excellent Talents in University for the grant (NCET-04-0717, P.R. China), and JMT to Chinese Scholarship Council for a doctoral scholarship.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Tu, JM., Kupka, M., Böhm, S. et al. Intermediate binding of phycocyanobilin to the lyase, CpeS1, and transfer to apoprotein. Photosynth Res 95, 163–168 (2008). https://doi.org/10.1007/s11120-007-9251-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-007-9251-4