Abstract
Purpose
To summary the clinical features of premenopausal women with functioning gonadotroph adenomas (FGAs) and preliminarily explore their molecular characterization.
Methods
12 premenopausal females with FGAs in our center were retrospectively analyzed. Previously reported cases were also summarized. The patients were clinically divided into FSH- or LH-predominant types according to their preoperative serum FSH/LH ratio. The expressions of related genes in the tumor tissues of female FGAs, non-functioning gonadotroph adenomas (NFGAs), and silent corticotropin adenomas were evaluated by RT-qPCR.
Results
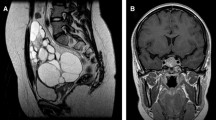
Of all the 12 patients with FGAs from our center, 11 (91.7%) were diagnosed as FSH-predominant type, and they all had menstrual disorders, including 9 with spontaneous ovarian hyperstimulation syndrome (sOHSS). Their hormonal profiles showed non-suppressed FSH (12.45 ± 7.34 IU/L) with hyperestrogenemia [median estradiol level 1353.0 pg/mL (636.0, 3535.0)]. The other patient (8.3%) with LH-predominant type mainly manifested with infertility and sustained elevated serum LH without FSH or estradiol increasing. 65 premenopausal FGAs patients were systematic reviewed. 60 patients (92.3%) were FSH-predominant type, including 86.7% presented with menstrual disorders, 16.7% reported infertility, and 98.2% (55/56) showed sOHSS. No sOHSS or hyperestrogenemia were found in the 5 patients (7.7%) with LH-predominant type. Pituitary imaging data revealed macroadenomas and microadenomas accounted for 89.2% and 10.8%, respectively. Of 63 patients (96.9%) who underwent pituitary adenoma resection, 77.8% had complete tumor resection and no recurrence at the last follow-up. The relative expressions of KISS1 mRNA were significantly higher in FGA group than in NFGA group (p = 0.018), and significantly positively correlated with the preoperative serum estradiol levels (p = 0.004).
Conclusions
Different clinical features were observed in premenopausal women with FGAs of FSH- or LH-predominant types. The elevated KISS1 expression in tumor tissues might involve in the secretion function of FGAs.




Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this article and its supplementary material file. Further enquiries can be directed to the corresponding authors.
References
Melmed S (2020) Pituitary-tumor endocrinopathies. N Engl J Med 382:937–950. https://doi.org/10.1056/NEJMra1810772
Mete O, Lopes MB (2017) Overview of the 2017 WHO classification of pituitary tumors. Endocr Pathol 28:228–243. https://doi.org/10.1007/s12022-017-9498-z
Ntali G, Capatina C, Grossman A et al (2014) Clinical review: functioning gonadotroph adenomas. J Clin Endocrinol Metab 99:4423–4433. https://doi.org/10.1210/jc.2014-2362
Kottler ML, Seret-Bégué D, Lahlou N et al (1998) The GnRH receptor gene is preferentially expressed in functioning gonadotroph adenomas and displays a Mae III polymorphism site. Clin Endocrinol (Oxf) 49:115–123
Davis JR, McNeilly JR, Norris AJ et al (2006) Fetal gonadotrope cell origin of FSH-secreting pituitary adenoma - insight into human pituitary tumour pathogenesis. Clin Endocrinol (Oxf) 65:648–654. https://doi.org/10.1111/j.1365-2265.2006.02644.x
Martínez-Fuentes AJ, Molina M, Vázquez-Martínez R et al (2011) Expression of functional KISS1 and KISS1R system is altered in human pituitary adenomas: evidence for apoptotic action of kisspeptin-10. Eur J Endocrinol 164:355–362. https://doi.org/10.1530/EJE-10-0905
Richard N, Corvaisier S, Camacho E et al (2009) KiSS-1 and GPR54 at the pituitary level: overview and recent insights. Peptides 30:123–129. https://doi.org/10.1016/j.peptides.2008.09.015
Isojima T, Shimatsu A, Yokoya S et al (2012) Standardized centile curves and reference intervals of serum insulin-like growth factor-I (IGF-I) levels in a normal Japanese population using the LMS method. Endocr J 59:771–780. https://doi.org/10.1507/endocrj.ej12-0110
Zhu H, Xu Y, Gong F et al (2017) Reference ranges for serum insulin-like growth factor I (IGF-I) in healthy Chinese adults. PLoS ONE 12:e0185561. https://doi.org/10.1371/journal.pone.0185561
Kawaguchi T, Ogawa Y, Ito K et al (2013) Follicle-stimulating hormone-secreting pituitary adenoma manifesting as recurrent ovarian cysts in a young woman–latent risk of unidentified ovarian hyperstimulation: a case report. BMC Res Notes 6:408. https://doi.org/10.1186/1756-0500-6-408
Kolnes AJ, Øystese K, Olarescu NC et al (2020) FSH levels are related to E-cadherin expression and subcellular location in nonfunctioning pituitary tumors. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa281
Di Rocco C, Maira G, Borrelli P (1982) Pituitary microadenomas in children. Childs Brain 9:165–178. https://doi.org/10.1159/000120052
Moses N, Goldberg V, Gutman R et al (1986) Combined FSH and LH secreting pituitary adenoma in a young fertile woman without primary gonadal failure. Acta Endocrinol (Copenh) 112:58–63. https://doi.org/10.1530/acta.0.1120058
Spertini F, Deruaz JP, Perentes E et al (1986) Luteinizing hormone (LH) and prolactin-releasing pituitary tumor: possible malignant transformation of the LH cell line. J Clin Endocrinol Metab 62:849–854. https://doi.org/10.1210/jcem-62-5-849
Blanco C, Lucas T, Alcañiz J et al (1994) Usefulness of thyrotropin-releasing hormone test, SMS 201–995, and bromocriptine in the diagnosis and treatment of gonadotropin-secreting pituitary adenomas. J Endocrinol Invest 17:99–104. https://doi.org/10.1007/BF03347693
Djerassi A, Coutifaris C, West VA et al (1995) Gonadotroph adenoma in a premenopausal woman secreting follicle-stimulating hormone and causing ovarian hyperstimulation. J Clin Endocrinol Metab 80:591–594. https://doi.org/10.1210/jcem.80.2.7852525
Christin-Maitre S, Rongières-Bertrand C, Kottler ML et al (1998) A spontaneous and severe hyperstimulation of the ovaries revealing a gonadotroph adenoma. J Clin Endocrinol Metab 83:3450–3453. https://doi.org/10.1210/jcem.83.10.5182
Catargi B, Felicie-Dellan E, Tabarin A (1999) Comment on gonadotroph adenoma causing ovarian hyperstimulation. J Clin Endocrinol Metab 84:3404. https://doi.org/10.1210/jcem.84.9.6011-6
Välimäki MJ, Tiitinen A, Alfthan H et al (1999) Ovarian hyperstimulation caused by gonadotroph adenoma secreting follicle-stimulating hormone in 28-year-old woman. J Clin Endocrinol Metab 84:4204–4208. https://doi.org/10.1210/jcem.84.11.6138
Pentz-Vidovíc I, Skorić T, Grubisić G et al (2000) Evolution of clinical symptoms in a young woman with a recurrent gonadotroph adenoma causing ovarian hyperstimulation. Eur J Endocrinol 143:607–614
Tashiro H, Katabuchi H, Ohtake H et al (2000) An immunohistochemical and ultrastructural study of a follicle-stimulating hormone-secreting gonadotroph adenoma occurring in a 10-year-old girl. Med Electron Microsc 33:25–31. https://doi.org/10.1007/s007950000004
Saveanu A, Morange-Ramos I, Gunz G et al (2001) A luteinizing hormone-, alpha-subunit- and prolactin-secreting pituitary adenoma responsive to somatostatin analogs: in vivo and in vitro studies. Eur J Endocrinol 145:35–41. https://doi.org/10.1530/eje.0.1450035
Shimon I, Rubinek T, Bar-Hava I et al (2001) Ovarian hyperstimulation without elevated serum estradiol associated with pure follicle-stimulating hormone-secreting pituitary adenoma. J Clin Endocrinol Metab 86:3635–3640. https://doi.org/10.1210/jcem.86.8.7766
Castelbaum AJ, Bigdeli H, Post KD et al (2002) Exacerbation of ovarian hyperstimulation by leuprolide reveals a gonadotroph adenoma. Fertil Steril 78:1311–1313. https://doi.org/10.1016/s0015-0282(02)04342-x
Murata Y, Ando H, Nagasaka T et al (2003) Successful pregnancy after bromocriptine therapy in an anovulatory woman complicated with ovarian hyperstimulation caused by follicle-stimulating hormone-producing plurihormonal pituitary microadenoma. J Clin Endocrinol Metab 88:1988–1993. https://doi.org/10.1210/jc.2002-021820
Murakami T, Higashitsuji H, Yoshinaga K et al (2004) Management of ovarian hyperstimulation due to follicle-stimulating hormone-secreting gonadotroph adenoma. BJOG 111:1297–1300. https://doi.org/10.1111/j.1471-0528.2004.00409.x
Benito M, Asa SL, Livolsi VA et al (2005) Gonadotroph tumor associated with multiple endocrine neoplasia type 1. J Clin Endocrinol Metab 90:570–574. https://doi.org/10.1210/jc.2004-1373
Mor E, Rodi IA, Bayrak A et al (2005) Diagnosis of pituitary gonadotroph adenomas in reproductive-aged women. Fertil Steril 84:757. https://doi.org/10.1016/j.fertnstert.2005.02.050
Roberts JE, Spandorfer S, Fasouliotis SJ et al (2005) Spontaneous ovarian hyperstimulation caused by a follicle-stimulating hormone-secreting pituitary adenoma. Fertil Steril 83:208–210. https://doi.org/10.1016/j.fertnstert.2004.06.061
Sugita T, Seki K, Nagai Y et al (2005) Successful pregnancy and delivery after removal of gonadotrope adenoma secreting follicle-stimulating hormone in a 29-year-old amenorrheic woman. Gynecol Obstet Invest 59:138–143. https://doi.org/10.1159/000083087
Kihara M, Sugita T, Nagai Y et al (2006) Ovarian hyperstimulation caused by gonadotroph cell adenoma: a case report and review of the literature. Gynecol Endocrinol 22:110–113. https://doi.org/10.1080/09513590600581665
Knoepfelmacher M, Danilovic DL, Rosa Nasser RH et al (2006) Effectiveness of treating ovarian hyperstimulation syndrome with cabergoline in two patients with gonadotropin-producing pituitary adenomas. Fertil Steril 86:719.e15–18. https://doi.org/10.1016/j.fertnstert.2006.01.055
Sicilia V, Earle J, Mezitis SG (2006) Multiple ovarian cysts and oligomenorrhea as the initial manifestations of a gonadotropin-secreting pituitary macroadenoma. Endocr Pract 12:417–421. https://doi.org/10.4158/EP.12.4.417
Ghayuri M, Liu JH (2007) Ovarian hyperstimulation syndrome caused by pituitary gonadotroph adenoma secreting follicle-stimulating hormone. Obstet Gynecol 109:547–549. https://doi.org/10.1097/01.AOG.0000247316.60573.4a
Cooper O, Geller JL, Melmed S (2008) Ovarian hyperstimulation syndrome caused by an FSH-secreting pituitary adenoma. Nat Clin Pract Endocrinol Metab 4:234–238. https://doi.org/10.1038/ncpendmet0758
Kajitani T, Liu S, Maruyama T et al (2008) Analysis of serum FSH bioactivity in a patient with an FSH-secreting pituitary microadenoma and multicystic ovaries: a case report. Hum Reprod 23:435–439. https://doi.org/10.1093/humrep/dem374
Baba T, Endo T, Kitajima Y et al (2009) Spontaneous ovarian hyperstimulation syndrome and pituitary adenoma: incidental pregnancy triggers a catastrophic event. Fertil Steril 92:390.e1–3. https://doi.org/10.1016/j.fertnstert.2009.02.071
Castelo-Branco C, del Pino M, Valladares E (2009) Ovarian hyperstimulation, hyperprolactinaemia and LH gonadotroph adenoma. Reprod Biomed Online 19:153–155. https://doi.org/10.1016/s1472-6483(10)60065-x
Gryngarten MG, Braslavsky D, Ballerini MG et al (2010) Spontaneous ovarian hyperstimulation syndrome caused by a follicle-stimulating hormone-secreting pituitary macroadenoma in an early pubertal girl. Horm Res Paediatr 73:293–298. https://doi.org/10.1159/000284395
Garmes HM, Grassiotto OR, Fernandes YB et al (2012) A pituitary adenoma secreting follicle-stimulating hormone with ovarian hyperstimulation: treatment using a gonadotropin-releasing hormone antagonist. Fertil Steril 97:231–234. https://doi.org/10.1016/j.fertnstert.2011.10.015
Kanaya M, Baba T, Kitajima Y et al (2012) Continuous follicle-stimulating hormone exposure from pituitary adenoma causes periodic follicle recruitment and atresia, which mimics ovarian hyperstimulation syndrome. Int J Womens Health 4:427–431. https://doi.org/10.2147/IJWH.S33386
Karapanou O, Tzanela M, Tamouridis N et al (2012) Gonadotroph pituitary macroadenoma inducing ovarian hyperstimulation syndrome: successful response to octreotide therapy. Hormones (Athens) 11:199–202. https://doi.org/10.14310/horm.2002.1347
Macchia E, Simoncini T, Raffaelli V et al (2012) A functioning FSH-secreting pituitary macroadenoma causing an ovarian hyperstimulation syndrome with multiple cysts resected and relapsed after leuprolide in a reproductive-aged woman. Gynecol Endocrinol 28:56–59. https://doi.org/10.3109/09513590.2011.588758
Halupczok J, Bidzińska-Speichert B, Lenarcik-Kabza A et al (2014) Gonadotroph adenoma causing ovarian hyperstimulation syndrome in a premenopausal woman. Gynecol Endocrinol 30:774–777. https://doi.org/10.3109/09513590.2014.934668
Cote DJ, Smith TR, Sandler CN et al (2016) Functional gonadotroph adenomas: case series and report of literature. Neurosurgery 79:823–831. https://doi.org/10.1227/NEU.0000000000001188
Caretto A, Lanzi R, Piani C et al (2017) Ovarian hyperstimulation syndrome due to follicle-stimulating hormone-secreting pituitary adenomas. Pituitary 20:553–560. https://doi.org/10.1007/s11102-017-0817-7
Vargas G, Balcazar-Hernandez LJ, Melgar V et al (2017) An FSH and TSH pituitary adenoma, presenting with precocious puberty and central hyperthyroidism. Endocrinol Diabetes Metab Case Rep. https://doi.org/10.1530/EDM-17-0057
Broughton C, Mears J, Williams A et al (2018) A clinically functioning gonadotroph adenoma presenting with abdominal pain, ovarian hyperstimulation and fibromatosis. Endocrinol Diabetes Metab Case Rep. https://doi.org/10.1530/EDM-18-0123
Eisenberg A, Mersereau J, Buckley AF et al (2019) Multiple pituitary adenomas with functional follicle-stimulating hormone secretion leading to ovarian hyperstimulation syndrome. AACE Clin Case Rep 5:159–163. https://doi.org/10.4158/ACCR-2018-0474
Graillon T, Castinetti F, Chabert-Orsini V et al (2019) Functioning gonadotroph adenoma with severe ovarian hyperstimulation syndrome: a new emergency in pituitary adenoma surgery? Surgical considerations and literature review. Ann Endocrinol (Paris) 80:122–127. https://doi.org/10.1016/j.ando.2018.11.007
Hirano M, Wada-Hiraike O, Miyamamoto Y et al (2019) A case of functioning gonadotroph adenoma in a reproductive aged woman. Endocr J 66:653–656. https://doi.org/10.1507/endocrj.EJ19-0066
Ichijo M, Tsuchiya K, Kasai T et al (2020) Follicle-stimulating hormone-secreting pituitary adenoma accompanied by painful thyroiditis. Intern Med 59:545–550. https://doi.org/10.2169/internalmedicine.3667-19
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C (T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Murray PG, Higham CE, Clayton PE (2015) 60 YEARS OF NEUROENDOCRINOLOGY: the hypothalamo-GH axis: the past 60 years. J Endocrinol 226:T123-140. https://doi.org/10.1530/JOE-15-0120
Meinhardt UJ, Ho KK (2006) Modulation of growth hormone action by sex steroids. Clin Endocrinol (Oxf) 65:413–422. https://doi.org/10.1111/j.1365-2265.2006.02676.x
Guarda FJ, Yu X, Shiraliyeva N et al (2021) GnRH agonist-associated pituitary apoplexy: a case series and review of the literature. Pituitary 24:681–689. https://doi.org/10.1007/s11102-021-01143-6
Javed Z, Qamar U, Sathyapalan T (2015) The role of kisspeptin signalling in the hypothalamic-pituitary-gonadal axis–current perspective. Endokrynol Pol 66:534–547. https://doi.org/10.5603/EP.2015.0066
Tena-Sempere M (2006) The roles of kisspeptins and G protein-coupled receptor-54 in pubertal development. Curr Opin Pediatr 18:442–447. https://doi.org/10.1097/01.mop.0000236396.79580.cc
Gahete MD, Vázquez-Borrego MC, Martínez-Fuentes AJ et al (2016) Role of the Kiss1/Kiss1r system in the regulation of pituitary cell function. Mol Cell Endocrinol 438:100–106. https://doi.org/10.1016/j.mce.2016.07.039
Navarro VM, Castellano JM, Fernández-Fernández R et al (2004) Developmental and hormonally regulated messenger ribonucleic acid expression of KiSS-1 and its putative receptor, GPR54, in rat hypothalamus and potent luteinizing hormone-releasing activity of KiSS-1 peptide. Endocrinology 145:4565–4574. https://doi.org/10.1210/en.2004-0413
Navarro VM, Castellano JM, Fernández-Fernández R et al (2005) Characterization of the potent luteinizing hormone-releasing activity of KiSS-1 peptide, the natural ligand of GPR54. Endocrinology 146:156–163. https://doi.org/10.1210/en.2004-0836
Navarro VM, Castellano JM, Fernández-Fernández R et al (2005) Effects of KiSS-1 peptide, the natural ligand of GPR54, on follicle-stimulating hormone secretion in the rat. Endocrinology 146:1689–1697. https://doi.org/10.1210/en.2004-1353
Park JW, Kim JH, Kwon JY (2020) Effects of Kiss2 on the expression of gonadotropin genes in the pituitary of Nile Tilapia (Oreochromis niloticus). Dev Reprod 24:149–158. https://doi.org/10.12717/DR.2020.24.3.149
Richard N, Galmiche G, Corvaisier S et al (2008) KiSS-1 and GPR54 genes are co-expressed in rat gonadotrophs and differentially regulated in vivo by oestradiol and gonadotrophin-releasing hormone. J Neuroendocrinol 20:381–393. https://doi.org/10.1111/j.1365-2826.2008.01653.x
Bernard V, Young J, Chanson P et al (2015) New insights in prolactin: pathological implications. Nat Rev Endocrinol 11:265–275. https://doi.org/10.1038/nrendo.2015.36
Acknowledgements
We thank Dr. Yuelun Zhang for his help in statistical analysis.
Funding
This work was supported by Chinese Academy of Medical Sciences Innovation Fund for Medical Science (CAMS-2016-I2M-1-002, 2017-I2M-1-011).
Author information
Authors and Affiliations
Contributions
Conception and design: All authors. Administrative support: HZ and YY. Collection and assembly of data: LW. RT-qPCR: HL. Data analysis and interpretation: All authors. Manuscript writing: LW. Final approval of manuscript: All authors. Accountable for all aspects of the work: All authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
This study was approved by PUMCH's medical ethics committee (Ethical audit number: ZS-1083). Written informed consent was obtained from participants prior to the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, L., Liang, H., Deng, C. et al. Functioning gonadotroph adenomas in premenopausal women: clinical and molecular characterization and review of the literature. Pituitary 25, 454–467 (2022). https://doi.org/10.1007/s11102-021-01205-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-021-01205-9