Abstract
Purpose
Hypopituitarism frequently develops following vasculotoxic snake bite complicated by acute kidney injury (AKI). Well defined prospective studies of prevalence of hypopituitarism and its predictors in vasculotoxic snake bites complicated by AKI are unavailable.
Methods
Fifty-one consecutive patients of AKI following vasculotoxic snake bite were evaluated for various clinical/biochemical parameters (including Free T4, TSH, Cortisol, ACTH, total testosterone, FSH, LH, prolactin, and IGF-1). Diabetes insipidus was evaluated in relevant cases. Twenty minutes whole blood clotting time (WBCT) at presentation was measured in all. MRI of hypothalamo-pituitary region was done at 3 months in subjects with hypopituitarism to rule out structural lesion.
Results
21.6% (11/51) patients developed hypopituitarism at baseline (within 7 days), 39.3% (13 /33) at 3 months developed hypopituitarism. Cortisol deficiency was the commonest abnormality. Subjects who developed hypopituitarism at baseline were younger compared to those without hypopituitarism (35.67 years vs. 46.59 years, p = 0.032) and required more sessions of hemodialysis (8 vs. 3, p = 0.041). Binary logistic regression confirmed that development of hypopituitarism could be predicted by increased number of sessions of hemodialysis (OR 1.51, p = 0.008) and 20 min WBCT (OR 1.2, p = 0.038).
Conclusion
Hypopituitarism is common following vasculotoxic snake bite in subjects who develop AKI requiring hemodialysis. Hypopituitarism can develop as early as 7 days following snake bite and should be evaluated for particularly in younger subjects, especially those requiring increasing number of sessions of hemodialysis and in subjects with abnormal 20 min WBCT at presentation.
Similar content being viewed by others
Introduction
Hypopituitarism is not an uncommon endocrine problem. While sellar/suprasellar tumour, pituitary surgery, radiation and hypophysitis are relatively more common causes, Sheehan’s syndrome, infections of the central nervous system like tuberculosis, HIV and opportunistic fungal infection, thalassemia, hemochromatosis and snake bite are important especially in tropical regions [1, 2].
In India more than 200,000 snake bites are reported annually, which results in 35,000–50,000 deaths. Russell’s viper bites are reported to cause coagulopathy (76%), neurotoxicity (59%), myotoxicity (24%) and renal toxicity (19%) [3]. Despite the wide distribution of Russell’s viper throughout South-east Asia, hypopituitarism following snake bite has been reported from only India, Sri Lanka and Myanmar. Lack of awareness amongst treating physicians and delayed presentation could possibly explain why this entity is often under-reported [4].
The time of onset/presentation of hypopituitarism following snake bite can vary from a few weeks to as long as upto 24 years. Hypopituitarism is thought to occur due to acute damage to the pituitary gland at the time of the precipitating event (envenomation), a gradual/slower progression of pituitary damage may occur over years due to other unknown mechanisms [5].
The predictors of development of hypopituitarism following vasculotoxic snake bite largely remains unknown. Previous studies suggest that acute kidney injury (oliguria) occurs in many (44%) patients of snake bite who subsequently developed hypopituitarism [6].
In the present study patients who developed acute kidney injury following vasculotoxic snake bite were evaluated for hypopituitarism. Pituitary function was evaluated at baseline, 6 weeks and 3 months following snake bite and we attempted to determine any possible predictors of hypopituitarism.
Materials and methods
This was a prospective study conducted at IPGME&R & SSKM Hospital, Kolkata, India. Subjects admitted following vasculotoxic snake bite complicated with acute kidney injury, presenting in the hospital between January 2017 to July 2018 were included in the study. Vasculotoxic snake bites were identified based on the syndromic approach to identifying snake bite as per the WHO guidelines for the management of snake bites [3]. The species of snake was identified by the snake brought by the patient’s relatives or by the description given by them and matching it with photographs of vasculotoxic snakes of the locality.
AKI was defined as any of the following: increase in serum creatinine by ≥ 0.3 mg/dl (≥ 26.5 µmol/l) within 48 h or an increase in creatinine to ≥ 1.5 times baseline (which is presumed to have occurred within the prior 7 days) or urine volume less than 0.5 mL/kg/h for 6 h (KDIGO Clinical Practice Guidelines for Acute Kidney Injury, 2012) [7]. The patients included in the study had AKI requiring haemodialysis and was under the care of a Nephrologist.
Patients who had received injection hydrocortisone before collection of samples for hormone testing were excluded from the study. Patients were studied at baseline (within 1 week of bite) and then at 6 weeks following snake bite and again at 3 months after snake bite. Vital signs, urine output, time to receiving anti-snake venom serum, bleeding manifestations, symptoms suggestive of acute hypopituitarism such as hypoglycemia and hypotension, renal parameters, serum electrolytes and 20 min whole blood clotting time (at baseline) were measured. All patients, irrespective of clinical presentation, underwent hormonal evaluation including free T4 (fT4), TSH, serum cortisol (8–9 am), FSH, LH, prolactin, total testosterone, insulin like growth factor-1 (IGF-1) and anti-thyroid peroxidase antibodies (anti-TPO Ab). Samples were drawn in the fasting state and serum was stored at − 20 °C until testing. Repeat tests (at 6 weeks and 3 months) for serum cortisol (8–9 am) were done after omitting the replacement therapy with hydrocortisone on previous evening, if they were on such therapy from previous evaluation. Repeat tests for central hypothyroidism were done only in those who were not given levothyroxine therapy. Repeat serum testosterone diagnosed initially was interpreted with caution as sepsis in acute stage may affect testosterone level. All assays were done by using the solid phase, competitive chemiluminescent immunometric assay except fT4, cortisol and total testosterone which were done by competitive immunoassays (Immulite-1000, Siemans Healthcare Diagnostics).
Hypopituitarism was defined as deficiency of one or more anterior pituitary cell types i.e. corticotrophs, thyrotrophs, gonadotrophs, lactotrophs and somatotrophs.
Central hypothyroidism was defined by having low fT4 (Normal range (NR) 0.8–1.9 ng/mL) with a low/normal TSH level (NR0.5–6.5 µIU/mL). Adrenal insufficiency was defined as a cortisol level < 3 mcg/dl with low or inappropriately normal ACTH or a cosyntropin stimulated cortisol level < 18 mcg/dl. Hypoprolactinemia was defined as a serum prolactin < 5.0 ng/mL. Hypogonadism was defined as a low serum total testosterone (<3 ng/mL) with a low or inappropriately normal FSH (NR 3–10 IU/l) and LH (NR 3–10 IU/l), keeping in mind that isolated low testosterone may be present in acute illness. IGF1 deficiency was defined as levels lower than the lower limit of the age- specific normal range (lower limit for detection was 25 ng/mL). Insulin tolerance test was not done and low age specific IGF -1 was taken as an evidence of GH deficiency in presence of deficiency of other hormones. In patients suspected to have central diabetes insipidus (symptoms of polyuria and/or hypernatremia), paired serum and urine osmolality were checked and a urine osmolality less than 300 mosm/L in the presence of a serum osmolality greater than 300 mosm/L in the background of deficient anterior pituitary hormones was taken as evidence of central diabetes insipidus. The diagnosis was subsequently confirmed based on the clinical response to desmopressin therapy. A formal water deprivation test was not done. However, as partial nephrogenic diabetes insipidus could not be excluded during the acute phase because of renal tubular dysfunction, the diagnosis was repeated at three months after stopping desmopressin therapy for 3 days. Central hypothyroidism is also often difficult to differentiate from normal reaction to acute sickness. Isolated low TSH with low normal free T4 was not treated immediately with a possible diagnosis of Sick Euthyroid Syndrome and those who did not recover at 6 weeks or 3 months were diagnosed as central hypothyroidism. An MRI of the hypothalamo pituitary region was done in patients with biochemical evidence of hypopituitarism after 3 months post snake bite to exclude any structural lesion.
The 20-min whole blood clotting test (WBCT20) [8, 9] in vasculotoxic snake bites is measured to determine if patients have a clinically significant coagulopathy. The WBCT20 was performed in a clean dry, 10-mL glass test tube glass tube free of preservatives, soaps or other contaminants. 2 mL blood was transferred into the glass tube and left undisturbed for 20 min. Solid clots that remain intact were considered normal. Partial clots and clots that degraded rapidly within 30 s on inversion of the tube were taken as abnormal.
Continuous data are presented as mean ± SD and results of categorical measurements are expressed in terms of frequency and percentage. Mann–Whitney U test and unpaired t-test were used for comparison as appropriate. Kendall’s correlation was applied to study correlation between various categorical and other variables. Binary logistic regression was used to predict development of hypopituitarism from various parameters.
The study was approved by the Institutional Ethics committee of IPGMER and SSKM Hospital, Kolkata, India. Written ‘Informed consent’ was also taken from all either from the patient or legal representative.
Results
A total of 51 patients were included in the study over a period of 18 months. All patients included in our study had AKI and underwent hemodialysis. Most of the patients (76.5%) in our study were farmers who worked bare feet in the fields. All patients were treated appropriately with anti-snake venom. Thirty three patients were available for follow up at 6 weeks and again at 3 months after the snake bite.
The mean age of the patients was 44.8 ± 15.8 years (range 11.5–86 years). All patients were male. Time of presentation to hospital for the bite was less than 12 h in all cases. All had evidence of cellulitis at the site of snake bite. Forty-six (90%) had a bite in the lower limb while 5 (10%) of them had upper limb bites. Generalized bleeding manifestations such as hematuria, hematemesis, gum bleeding, hematochezia was present in 21 (41%) and 11 (22%) had only bleeding from the bite wound. 19 (37%) patients did not present with any bleeding manifestations. One patient needed transfusion of 7 units of fresh frozen plasma. Urine output was normal in 14 (28%), oliguria was there in 24 (47%) and 13 (25%) had anuria. The 20 min whole blood clotting time was abnormal (i.e. did not clot) in 24 (47%) patients and it was normal in 27 (53%). None of the patients had features suggestive of acute hypopituitarism such as recurrent hypoglycemia or hypotension. Table 1 summarizes the clinical and investigation profile of 13 patients who developed hypopituitarism.
Hormonal profile of all 51 patients at baseline irrespective of clinical presentation was checked. At least one deficient anterior pituitary hormone at baseline was found in 21.6% (11/51). Patients diagnosed with hypopituitarism at baseline testing were younger (35.67 years vs. 46.59 years, p = 0.032) and required greater number of sessions of hemodialysis (8 vs. 3, p = 0.041). However, there were no significant differences at baseline in the time to AVS, number of vials of AVS or serum urea and creatinine between patients developing hypopituitarism and those without hypopituitarism. These data are presented in Table 2.
Of 33 patients followed upto 3 months, 13 (39.3%) developed hypopituitarism. Amongst those 13 patients, 11 (84.6%) were diagnosed at baseline while 1 (7.7%) patient developed hypopituitarism at 6 weeks and 1 (7.7%) at 3 months. Twelve of these 13 subjects had cortisol deficiency needing hydrocortisone supplement. At 3 months corticotrophs axis was deficient in 12 (36.4%), followed by lactotrophs in 9 (27.3%), gonadotrophs in 9 (27.3%), somatotrophs in 5 (15.2%) and thyrotrophs in 4 (12.1%). One patient who did not have cortisol deficiecy at baseline developed it subsequently at 6 weeks.Two patients (15.3%) developed central diabetes insipidus. In both of them symptoms of polyuria appeared after cortisol replacement. They were reevaluated for diagnosis at 3 months after stopping desmopressin therapy for 3 days. The chronological biochemical endocrine deficiency is summarized in Table 3.
At initial test, we got two individuals with central hypothyroid. Both of them had very low T4, marginally low T3 (Hence Sick Euthyroid Syndrome was less likely and in such circumstances usually the predominant abnormality is low T3. We didn’t have facilities to check rT3). T3 was done only in selected cases. Hence we treated these two subjects with Levothyroxine. We followed usual protocol for treatment and monitoring of Levothyroxine.
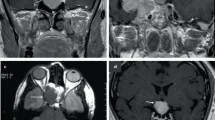
MRI of hypothalamo-pituitary region was done at 3 months in subjects with hypopituitarism to rule out structural lesion. It was abnormal in only 5 out of the 13 patients with hypopituitarism with partial empty sella in 4 patients and old haemorrhage in pituitary in 1 patient and was normal in 8 patients. The persons with Empty Sella Syndrome in MRI were reevaluated for history of any pre-snake bite symptomatology of pituitary dysfunction to exclude its presence before snake bite.
Age, number of vials of AVS, time to AVS, baseline urea and creatinine, bleeding manifestations, urine output did not have any significant correlation with development of hypopituitarism. There was a significant positive correlation between hypopituitarism and number of hemodialysis (Kendal’s Tau-b 0.371, p = 0.015) and 20 min whole blood clotting time (Kendal’s Tau-b 0.291, p = 0.048).
Binary logistic regression using these various parameters showed that the development of hypopituitarism could be predicted by number of hemodialysis (OR 1.51, p = 0.008) and 20 min whole blood clotting time (OR 1.2, p = 0.038).
Discussion
Snake bite is common in India and is an occupational hazard for farmers in rural areas. In our study all subjects were males and 46 (90%) patients had lower limb bites.
The patients attending our hospital were predominantly farmers, working in the paddy fields from rural West Bengal, India, during the rainy seasons. These workers are almost exclusively males. In addition due to social circumstances, in this part of the world often men are often more privileged to be receiving treatment and be transferred to tertiary care set up.
Studies evaluating hypopituitarism as a complication of vascultotoxic snake bite with AKI (SB-AKI) are only handful. Golay at el reported [10] data from 126 patients with SB-AKI (Snake bite related acute kidney injury) of whom 86 were new cases and 40 were those who were still under follow up in nephrology OPD for some duration. 26 patients had died due to complication and 30 of the new cases were lost to follow up after discharge. Thus a total 96 persons were available for evaluation of hypopituitarism. However only 25 of them were biochemically evaluated on the basis of clinical suspicion of pituitary dysfunction. It is a common knowledge that symptoms of hypopituitarism are nonspecific, subtle, insidious and in the elderly can be easily mistaken as age-related physiological changes. These symptoms may also be masked by other chronic ailments with similar manifestions. It is not prudent to check for prevalence of hypopituitarism based on symptoms only, especially in a situation where this happens frequently. Therefore we cannot exclude the possibility of hypopituitarism in the rest of the cohort. Again, though it was evaluated in 25 persons, it was reported to happen in the whole cohort of 96 persons. Thus, the study underestimates the prevalence of hypopituitarism in SB-AKI. As the study population is heterogeneous in terms of inclusion, no conclusions could be drawn regarding time of onset of hypopituitarism following snake bite.
Naik et al. reported [11] data from 76 snake bite patients from multiple centres, of which 60 were evaluated at 6 months. AKI was documented in almost half (n = 20) of the confirmed 41 vasculotoxic snake bite patients. However only six patients were documented to have hypopituitarism. Retrospectively AKI was present in 2 (33.3%) patients. Though it was not reported in the study, 2 out of those 20 who developed AKI early developed hypopituitarism at 6 months. Thus the perspective of this study is completely different from the study conducted by us.
Another recent study [12] by Shivaprasad et al. reported 8 cases of delayed hypopituitarism wherein the aetiology of hypopituitarism was thought to be due to snake bite. However the time interval between the diagnosis of snake bite and hypopituitarism was 8.1 ± 3.6 years. Retrospective analysis revealed 75% (6 out of those 8 hypopituitarism) had AKI. Hence, the perspective of this study also is different from the study conducted by us especially in terms of calculating the prevalence of hypopituitarism as this was not prospective in nature.
In another study [13] by Rajagopala et al. over 17 months, biochemical testing for hormones was done in only those patients with a clinical suspicion of acute hypopituitarism. They identified 9 cases of acute hypopituitarism which occurred after a median of 9 (range 2–14) days after severe envenomation and was associated with multi-organ dysfunction, lower platelet counts, more bleeding and transfusions. All patients had history of acute renal failure.
Most of the cases reported are from the tropical countries of India, Sri Lanka and Myanmar. Patients of our study and their primary caregivers were completely unaware of the possibility of this late complication of snake bite. It is important to recognise hypopituitarism early due to the significant morbidity and cardiovascular mortality associated if untreated [14].
Although many cases of hypopituitarism due to snake bite reported in literature presented with symptoms of acute pituitary insufficiency such as recurrent hypoglycemia and hypotension [5, 13] in our study none of the patients had hypoglycemia or hypotension. All patients included in our study had AKI and underwent hemodialysis. The higher prevalence in our study compared to previous studies could be possibly due to unbiased testing of all patients. This emphasizes the importance of testing for hormone insufficiency despite absence of such symptoms.
Hypopituitarism after snake bite has been reported mostly in the younger population in previous studies [5, 11]. This was probably due the greater outdoor activity in the younger patients and greater likelihood of being bitten by snakes. The mean age of the patients who developed hypopituitarism in our study also was lower than those who did not (35.67 years vs. 46.59 years).
In the study by Naik et al. the common hormone deficiencies were somatotrophs in 83% and gonadotrophs in 50% patients [11]. Hypoprothyroidism and hypoprolactinemia was present in 1 patient. None of the patients who were euhormonal at baseline were diagnosed with new hormone deficiencies at 6 months. Golay et al. did not mention hypoprolactinemia in any of their patients [10]. In our study the most common axis affected was of the corticotrophs which was deficient in 12 (36.4%), followed by lactotrophs in 9 (27.3%) patients, gonadotrophs in 9 (27.3%) patients, somatotrophs in 5 (15.2%) and thyrotrophs in 4 (12.1%) patients. 1 patient who did not have cortisol deficiency at baseline developed it subsequently at 6 weeks post snake bite. This emphasizes the need for testing for other hormone deficiencies on follow up in patients in whom some axes are affected at baseline.
MRI of hypopthalamo-pituitary region was abnormal in only 5 out of the 13 patients with hypopituitarism in our study. It showed a partial empty sella in 4 patients and old haemorrhage in the anterior pituitary in 1 patient. This is similar to previous studies where an MRI abnormality is not a universal finding in chronic hypopituitarism patients post snake bite [11].
Central diabetes insipdus post snake bite is less commonly documented in literature. None of the 6 patients with hypopituitarism had posterior pituitary involvement in the study by Naik et al. [11]. Golay et al. reported central diabetes insipidus in 1 of the 9 patients with hypopituitarism [10]. Grace et al. reported a case of central diabetes insipidus which developed as sequelae to viper bite [15]. In our study central diabetes insipidus was diagnosed in 2 of the 13 patients with hypopititarism which was unmasked after starting cortisol replacement.
In the study by Rajagopala et al. hypotension predicted the development of acute hypopituitarism in patients with Russell’s viper envenomation [13]. Golay et al. concluded that development of chronic kidney disease on follow up of snake bite patients with acute renal failure predicted subsequent development of hypopituiarism in these patients [10]. Renal failure, coagulopathy or any clinical score of severity of illness did not predict development of hypopituitarism in the study by Naik et al. [11].
In our study, at baseline, the requirement of number of sessions of hemodialysis was significantly greater in those who developed hypopituitarism compared to those who did not. There was a statistically significant positive correlation between the number of hemodialysis received by the patient and development of hypopituitarism. However in binary logistic regression analysis of various parameters such as age, bleeding manifestations, 20 min whole blood clotting time, time to receiving anti-venom serum, number of vials of anti-venom serum, number of hemodialysis, baseline urea and creatinine, the only two predictors were the number of hemodialysis and 20 min whole blood clotting time.
Conclusion
The prevalence of hypopituitarism secondary to vasculotoxic snake bite with acute kidney injury requiring hemodialysis is much higher than the prevalence in the previous studies in literature relating SB-AKI and hypopituitarism. The most common axis to be involved in hypopituitarism post snake bite is corticotrophs followed by the gonadotrophs and the thyrotrophs. Pituitary damage secondary to vasculotoxic snake bite with acute kidney injury may be progressive with later development of new hormone deficiencies not present at initial diagnosis of hypopituitarism. Hence it is important to follow up patients and do appropriate tests for various hormone axes which may unmask new hormone insufficiencies. Most importantly, the development of hypopituitarism could be predicted by increased number of hemodialysis and abnormal 20 min whole blood clotting time.
Limitations of the study
All patients included in the study were male. Also, we have a relatively small sample size and this may limit the interpretation of the results in the perspective of a larger sample size. We believe that the chances of reversibility is very low after 3 months and repeat tests after a several months or years may yield more hormonal deficiency as suggested by other studies. Though we have followed stringent criteria, it is sometimes very difficult to differentiate central hypothyroidism and hypogonadism in acute clinical illness and results must be interpreted with extra caution.
References
Kalra S, Dhanwal D, Khadilkar V (2011) Hypopituitarism in the tropics. Indian J Endocrinol Metab 15(3):151
Gundgurthi A, Garg MK, Bhardwaj R, Brar KS, Kharb S, Pandit A (2012) Clinical spectrum of hypopituitarism in India: a single center experience. Indian J Endocrinol Metab 16:803–808
Warrel DA (2010) Guidelines for the management of snake. World Health Organization. https://apps.searo.who.int/PDS_DOCS/B4508.pdf. Accessed 13 Oct 2019
Antonypillai CN, Wass JA, Warrell DA, Rajaratnam HN. Hypopituitarism following envenoming by Russell's vipers (Daboia siamensis and D. russelii) resembling Sheehan's syndrome: first case report from Sri Lanka, a review of the literature and recommendations for endocrine management.QJM. 2011; 104 (2):97–108.
Pe T, Phillips RE, Warrell DA, Moore RA, Swe TN, M Lwin, Burke CW. Acute and chronic pituitary failure resembling Sheehan's syndrome following bites by Russell's viper in Burma. Lancet. 1987; 2(8562):763–7.
Lwin M, Warrell DA, Phillips RE, Swe TN, Pe T, Lay MM (1985) Bites by Russell's viper (Vipera russelli siamensis) in Burma: haemostatic, vascular, and renal disturbances and response to treatment. Lancet 2(8467):1259–1264
KDIGO Clinical Practice Guideline for Acute Kidney Injury. (2012); 2, Suppl 1. https://kdigo.org/wp-content/uploads/2016/10/KDIGO-2012-AKI-Guideline-English.pdf. Accessed 13 Oct 2019
Isbister GK, Maduwage K, Shahmy S, Mohamed F, Abeysinghe C, Karunathilake H, Ariaratnam CA, Buckley NA (2013) Diagnostic 20-min whole blood clotting test in Russell's viper envenoming delays antivenom administration. QJM 106(10):925–932
Benjamin JM, Chippaux JP, Sambo BT, Massougbodji A (2018) Delayed double reading of whole blood clotting test (WBCT) results at 20 and 30 minutes enhances diagnosis and treatment of viper envenomation. J Venom Anim Toxins Incl Trop Dis 24:14
Golay V, Roychowdhary A, Dasgupta S, Pandey R (2014) Hypopituitarism in patients with vasculotoxic snake bite envenomation related acute kidney injury: a prospective study on the prevalence and outcomes of this complication. Pituitary 17(2):125–131
Naik BN, Bhalla A, Sharma N, Mokta J, Singh S, Gupta P, Rai A, Subbiah S, Bhansali A, Dutta P (2018) Pituitary dysfunction in survivors of Russell's viper snake bite envenomation: A prospective study. Neurol India 66(5):1351
Shivaprasad C, Aiswarya Y, Sridevi A, Anupam B, Amit G, Rakesh B, Annie PA, Anish K (2019) Delayed hypopituitarism following Russell's viper envenomation: a case series and literature review. Pituitary 22(1):4–12
Rajagopala S, Thabah MM, Ariga KK, Gopalakrishnan M (2015) Acute hypopituitarism complicating Russell’s viper envenomation: case series and systematic review. QJM 108(9):719–728
Bates AS, Van't Hoff W, Jones PJ, Clayton RN (1996) The effect of hypopituitarism on life expectancy. J Clin Endocrinol Metab 81(3):1169–1172
Grace M, Shanoj KC (2014) An unusual complication of snake bite. Egypt J Intern Med 26:91–92
Acknowledgements
We acknowledge the research grant provided by Endocrine Society of Bengal, a state affiliate of Endocrine Society of India for conducting the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bhat, S., Mukhopadhyay, P., Raychaudhury, A. et al. Predictors of hypopituitarism due to vasculotoxic snake bite with acute kidney injury. Pituitary 22, 594–600 (2019). https://doi.org/10.1007/s11102-019-00990-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-019-00990-8