Abstract
Introduction: Gabexate mesylate is a drug marketed only in Italy and Japan and it is considered an essential drug in the treatment of acute pancreatitis. A periodic revision and evaluation of drug utilization in our hospital demonstrated that the dosages and indications of Gabexate mesilate (GM) did not follow those settled by the National Health System and by our Hospital Formulary. We therefore promoted and conducted a program of drug information and meetings with the physicians in order to improve the correct utilisation of GM and identify potential problems.
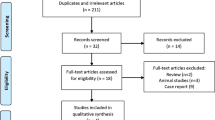
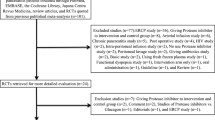
Methods: GM prescriptions in 1999 were analysed for indications and dosages. Scheduled meetings with the medical staff were successively started in order to debate current therapies and treatments of acute pancreatitis using the criteria of evidence- based medicine (EBM). Follow-up was done in 2000, 2001 and 2002 to evaluate the efficacy of our intervention.
Results: The hospital board and 20 physicians were involved in this program of evidence-based drug information. The follow-up and the monitoring of GM prescriptions showed a reduction in the utilisation of GM in the subsequent period.
Conclusions: GM is one of the most expensive drugs in our hospital, but its benefits are not very evident. Moreover, the international guidelines and literature ascribe to GM only a marginal role in the treatment of acute pancreatitis. Evidence-based medicine strengthens clinical experience with the evidence from the literature and underlines that essential drugs are used worldwide.
Similar content being viewed by others
References
M Buchler P Malfertheiner W Uhl J Scholmerich F Stockmann G. Adler et al. (1993) ArticleTitleGabexate mesilate in human acute pancreatitis. Gastroenterology 104 1165–70 Occurrence Handle1:STN:280:ByyB3Mngt10%3D Occurrence Handle8462805
H. Goebell (1988) ArticleTitleMulticenter double-blind study of Gabexate-mesilate (Foy), given intravenously in low dose in acute pancreatitis Digestion 40 83
R Valderrama M Perez-Mateo S Navarro N Vazquez L Sanjose MJ. Adriàn et al. (1992) ArticleTitleMulticenter double-blind trial of Gabexate mesylate (FOY) in unselected patients with acute pancreatitis Digestion 51 65–70 Occurrence Handle1:STN:280:By2A28zgsl0%3D Occurrence Handle1499875
P Pederzoli G Cavallini M Falconi C. Bassi (1993) ArticleTitleGabexate mesilate vs aprotinine in human acute pancreatitis (GA ME. P.A.): a prospective randomized double-blind multicenter study. Int J Pancreatol 14 117–24 Occurrence Handle1:STN:280:ByuC3M%2Fms1M%3D Occurrence Handle7506742
CY Yang CS Chang-Chien YF. Liaw (1987) ArticleTitleControlled trial of protease inhibitor gabexate mesylate (Foy) in the treatment of acute pancreatitis. Pancreas 2 IssueID(6 698–700 Occurrence Handle1:STN:280:BieC2cvkt1I%3D Occurrence Handle3125546
A Messori R Rampazzo G Scroccaro R Olivato C Bassi M. Falconi et al. (1995) ArticleTitleEffectiveness of Gabexate mesilate in acute pancreatitis, a metaanalysis Digest Dis Sci 40 IssueID(4 734–8 Occurrence Handle1:STN:280:ByqB3snovVY%3D Occurrence Handle7720462
A Andriulli G Leandro R Clemente V Festa n Caruso V. Annese et al. (1998) ArticleTitleMeta-analysis of somatostatin, octeotride and gabexate mesilate in the terapy of acute pancreatitis Aliment Pharmacol Ther 12 237–45 Occurrence Handle10.1046/j.1365-2036.1998.00295.x Occurrence Handle1:CAS:528:DyaK1cXislejsrY%3D Occurrence Handle9570258
United Kingdom guidelines for the management of acute pancreatitis (1998). Gut 42 (2):S1–S13
H Friess HG Beger U Sulkowski H Becker B Hofbauer HJ. Dennler et al. (1995) ArticleTitlecontrolled multicentre study of the prevention of complications by octreotide in patients undergoing surgery for chronic pancreatitis Br J Surg 82 1270–3 Occurrence Handle1:STN:280:BymD3s%2FksVQ%3D Occurrence Handle7552016
F Carballo E Domininguez L. Fernandez-Calvet et al. (1991) ArticleTitleIs somatostatin useful in the treatment of acute pancreatitis? A meta-analysis Digestion 49 12–3
H Paran D Neufeld A Mayo I Shwartz P Singer O. Kaplan et al. (1995) ArticleTitlePreliminary report of a prospective randomized study of octreotide in the treatment of severe acute pancreatitis J Am Coll Surg 181 121–4 Occurrence Handle1:STN:280:ByqA2MzltV0%3D Occurrence Handle7627383
F Fiedler G Jauernig V Keim A Richter HJ. Bender (1996) ArticleTitleOctreotide treatment in patients with necrotising pancreatitis and pulmonary failure Intens Care Med 22 909–15 Occurrence Handle10.1007/s001340050186 Occurrence Handle1:STN:280:ByiD287jvFE%3D
C McKay J Baxter C. Imrie (1997) A randomized controlled trial of octeotride in the management of patients with acute pancreatitis. Int J Pancreatol 21 13–9 Occurrence Handle1:CAS:528:DyaK2sXjtFemsL0%3D
CW Imrie IS Benjamin JC Ferguson AJ McKay I Mackenzie J ONeill et al. (1978) ArticleTitleA single-centre, double-blind trial of trasylol therapy in primary acute pancreatitis Br J Surg 65 337–40 Occurrence Handle1:STN:280:CSeC28nkt1Y%3D Occurrence Handle348250
R Berling S Genell K. Ohlsson (1994) ArticleTitleHigh-dose intraperitoneal aprotinin treatment of acute severe pancreatitis : a double-blind randomized multi-centre trial J Gastroenterol 29 479–85 Occurrence Handle1:STN:280:ByqD3sblsFw%3D Occurrence Handle7524945
DL. Wyncoll (1999) ArticleTitleThe management of severe acute necrotising pancreatitis: an evidence-based review of the literature Intens Care Med 25 146–56 Occurrence Handle10.1007/s001340050808 Occurrence Handle1:STN:280:DyaK1M3gslyrsQ%3D%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banfi, R., Borselli, G., Cappelletti, S. et al. Gabexate mesilate and acute pancreatitis: an experience of evidence based drug information for improving rational drug use. Pharm World Sci 27, 121–123 (2005). https://doi.org/10.1007/s11096-004-5692-4
Issue Date:
DOI: https://doi.org/10.1007/s11096-004-5692-4