Purpose
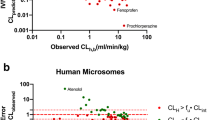
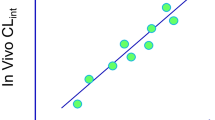
The utility of in vitro metabolism to accurately predict the clearance of hepatically metabolized drugs was evaluated. Three major goals were: (1) to optimize substrate concentration for the accurate prediction of clearance by comparing to K m value, (2) to prove that clearance of drugs by both oxidation and glucuronidation may be predicted by this method, and (3) to determine the effects of nonspecific microsomal binding and plasma protein binding.
Methods
The apparent K m values for five compounds along with scaled intrinsic clearances and predicted hepatic clearances for eight compounds were determined using a substrate loss method. Nonspecific binding to both plasma and microsomal matrices were also examined in the clearance calculations.
Results
The K m values were well within the 2-fold variability expected for between laboratory comparisons. Using both phase I and/or phase II glucuronidation incubation conditions, the predictions of in vivo clearance using the substrate loss method were shown to correlate with published human clearance values. Of particular interest, for highly bound drugs (>95% plasma protein bound), the addition of a plasma protein binding term increased the accuracy of the prediction of in vivo clearance.
Conclusions
The substrate loss method may be used to accurately predict hepatic clearance of drugs.

Similar content being viewed by others
Abbreviations
- HPLC:
-
high-performance liquid chromatography
- K m value:
-
Michaelis constant value
- LC/MS/MS:
-
liquid chromatography tandem mass spectrometry
References
K. Venkatakrishnan L. L. Moltke Particlevon R. S. Obach D. J. Greenblatt (2003) ArticleTitleDrug metabolism and drug interactions: application and clinical value of in vitro models Curr. Drug Metab. 4 423–459 Occurrence Handle10.2174/1389200033489361 Occurrence Handle1:CAS:528:DC%2BD3sXotlGrtLc%3D Occurrence Handle14529374
K. Ito J. B. Houston (2004) ArticleTitleComparison of the use of liver models for predicting drug clearance using in vitro kinetic data from hepatic microsomes and isolated hepatocytes Pharm. Res. 21 785–792 Occurrence Handle10.1023/B:PHAM.0000026429.12114.7d Occurrence Handle1:CAS:528:DC%2BD2cXjs1Kisr8%3D Occurrence Handle15180335
J. C. Kalvass D. A. Tess C. Giragossian M. C. Linhares T. S. Maurer (2001) ArticleTitleInfluence of microsomal concentration on apparent intrinsic clearance: implications for scaling in vitro data Drug Metab. Dispos. 29 1332–1336 Occurrence Handle1:CAS:528:DC%2BD3MXntVGqurk%3D Occurrence Handle11560877
Y. Naritomi S. Terashita S. Kimura A. Suzuki A. Kagayama Y. Sugiyama (2001) ArticleTitlePrediction of human hepatic clearance from in vivo animal experiments and in vitro metabolic studies with liver microsomes from animals and humans Drug Metab. Dispos. 29 1316–1324 Occurrence Handle1:CAS:528:DC%2BD3MXntVGqurs%3D Occurrence Handle11560875
R. S. Obach (1999) ArticleTitlePrediction of human clearance of twenty-nine drugs from hepatic microsomal intrinsic clearance data: an examination of in vitro half-life approach and nonspecific binding to microsomes Drug Metab. Dispos. 27 1350–1359 Occurrence Handle1:CAS:528:DyaK1MXntFSht7c%3D Occurrence Handle10534321
S. Kumar K. Samuel R. Subramanian M. P. Braun R. A. Stearns S. H. Chiu D. C. Evans T. A. Baillie (2002) ArticleTitleExtrapolation of diclofenac clearance from in vitro microsomal metabolism data: role of acyl glucuronidation and sequential oxidative metabolism of the acyl glucuronide J. Pharmacol. Exp. Ther. 303 969–978 Occurrence Handle10.1124/jpet.102.038992 Occurrence Handle1:CAS:528:DC%2BD38Xptlamt7Y%3D Occurrence Handle12438516
M. G. Soars B. Burchell R. J. Riley (2002) ArticleTitleIn vitro analysis of human drug glucuronidation and prediction of in vivo metabolic clearance J. Pharmacol. Exp. Ther. 301 382–390 Occurrence Handle10.1124/jpet.301.1.382 Occurrence Handle1:CAS:528:DC%2BD38Xis1KqsLs%3D Occurrence Handle11907196
R. P. Austin P. Barton S. L. Cockroft M. C. Wenlock R. J. Riley (2002) ArticleTitleThe influence of nonspecific microsomal binding on apparent intrinsic clearance, and its prediction from physicochemical properties Drug Metab. Dispos. 30 1497–1503 Occurrence Handle10.1124/dmd.30.12.1497 Occurrence Handle1:CAS:528:DC%2BD38XptVyisbg%3D Occurrence Handle12433825
R. S. Foti M. B. Fisher (2004) ArticleTitleImpact of incubation conditions on bufuralol human clearance predictions: enzyme lability and nonspecific binding Drug Metab. Dispos. 32 295–304 Occurrence Handle10.1124/dmd.32.3.295 Occurrence Handle1:CAS:528:DC%2BD2cXhslOks7g%3D Occurrence Handle14977863
J. M. Margolis R. S. Obach (2003) ArticleTitleImpact of nonspecific binding to microsomes and phospholipid on the inhibition of cytochrome P4502D6: implications for relating in vitro inhibition data to in vivo drug interactions Drug Metab. Dispos. 31 606–611 Occurrence Handle10.1124/dmd.31.5.606 Occurrence Handle1:CAS:528:DC%2BD3sXjt1Ggt7Y%3D Occurrence Handle12695349
S. Bjorkman D. R. Wada B. M. Berling G. Benoni (2001) ArticleTitlePrediction of the disposition of midazolam in surgical patients by a physiologically based pharmacokinetic model J. Pharm. Sci. 90 1226–1241 Occurrence Handle10.1002/jps.1076 Occurrence Handle1:CAS:528:DC%2BD3MXmvFGjsr0%3D Occurrence Handle11745776
A. J. Wood R. E. Vestal C. L. Spannuth W. J. Stone G. R. Wilkinson D. G. Shand (1980) ArticleTitlePropranolol disposition in renal failure Br. J. Clin. Pharmacol. 10 561–566 Occurrence Handle1:CAS:528:DyaL3MXns1GhsQ%3D%3D Occurrence Handle7470370
L. Z. Benet B. A. Hoener (2002) ArticleTitleChanges in plasma protein binding have little clinical relevance Clin. Pharmacol. Ther. 71 115–121 Occurrence Handle10.1067/mcp.2002.121829 Occurrence Handle1:CAS:528:DC%2BD38XjtFChtbc%3D Occurrence Handle11907485
T. A. Hoeven Particlevan der M. J. Coon (1974) ArticleTitlePreparation and properties of partially purified cytochrome P-450 and reduced nicotinamide adenine dinucleotide phosphate-cytochrome P-450 reductase from rabbit liver microsomes J. Biol. Chem. 249 6302–6310 Occurrence Handle4153601
M. B. Fisher K. Campanale B. L. Ackermann M. Vandenbranden S. A. Wrighton (2000) ArticleTitleIn vitro glucuronidation using human liver microsomes and the pore-forming peptide alamethicin Drug Metab. Dispos. 28 560–566 Occurrence Handle1:CAS:528:DC%2BD3cXivVKgu74%3D Occurrence Handle10772635
M. J. Banker T. H. Clark J. A. Williams (2003) ArticleTitleDevelopment and validation of a 96-well equilibrium dialysis apparatus for measuring plasma protein binding J. Pharm. Sci. 92 967–974 Occurrence Handle10.1002/jps.10332 Occurrence Handle1:CAS:528:DC%2BD3sXjs1ehsrs%3D Occurrence Handle12712416
B. Davies T. Morris (1993) ArticleTitlePhysiological parameters in laboratory animals and humans Pharm. Res. 10 1093–1095 Occurrence Handle10.1023/A:1018943613122 Occurrence Handle1:STN:280:ByyA1M7ivFQ%3D Occurrence Handle8378254
J. B. Houston (1994) ArticleTitleUtility of in vitro drug metabolism data in predicting in vivo metabolic clearance Biochem. Pharmacol. 47 1469–1479 Occurrence Handle10.1016/0006-2952(94)90520-7 Occurrence Handle1:CAS:528:DyaK2cXkt1Kju7c%3D Occurrence Handle8185657
L. S. Goodman L. E. Limbird P. B. Milinoff R. W. Ruddon A. G. Gilman (1996) Goodman and Gilman's The Pharmacological Basis of Therapeutics EditionNumber9th ed. McGraw-Hill New York
J. Mendelson R. A. Upton E. T. Everhart P. Jacob SuffixIII R. T. Jones (1997) ArticleTitleBioavailability of sublingual buprenorphine J. Clin. Pharmacol. 37 31–37 Occurrence Handle1:CAS:528:DyaK2sXhvFKgsbg%3D Occurrence Handle9048270
R. Bort K. Mace A. Boobis M. J. Gomez-Lechon A. Pfeifer J. Castell (1999) ArticleTitleHepatic metabolism of diclofenac: role of human CYP in the minor oxidative pathways Biochem. Pharmacol. 58 787–796 Occurrence Handle10.1016/S0006-2952(99)00167-7 Occurrence Handle1:CAS:528:DyaK1MXltVyjt7o%3D Occurrence Handle10449188
K. Kobayashi T. Yamamoto K. Chiba M. Tani N. Shimada T. Ishizaki Y. Kuroiwa (1998) ArticleTitleHuman buprenorphine N-dealkylation is catalyzed by cytochrome P450 3A4 Drug Metab. Dispos. 26 818–821 Occurrence Handle1:CAS:528:DyaK1cXlsVSqsL0%3D Occurrence Handle9698298
E. Koyama K. Chiba M. Tani T. Ishizaki (1997) ArticleTitleReappraisal of human CYP isoforms involved in imipramine N-demethylation and 2-hydroxylation: a study using microsomes obtained from putative extensive and poor metabolizers of S-mephenytoin and eleven recombinant human CYPs J. Pharmacol. Exp. Ther. 281 1199–1210 Occurrence Handle1:CAS:528:DyaK2sXktVKlu7o%3D Occurrence Handle9190854
M. G. Soars B. J. Ring S. A. Wrighton (2003) ArticleTitleThe effect of incubation conditions on the enzyme kinetics of UDP-glucuronosyltransferases Drug Metab. Dispos. 31 762–767 Occurrence Handle10.1124/dmd.31.6.762 Occurrence Handle1:CAS:528:DC%2BD3sXkt1KrtLg%3D Occurrence Handle12756209
L. L. Moltke Particlevon D. J. Greenblatt J. M. Grassi B. W. Granda K. Venkatakrishnan J. Schmider J. S. Harmatz R. I. Shader (1998) ArticleTitleMultiple human cytochromes contribute to biotransformation of dextromethorphan in vitro: role of CYP2C9, CYP2C19, CYP2D6, and CYP3A J. Pharm. Pharmacol. 50 997–1004
A. Ghosal H. Satoh P. E. Thomas E. Bush D. Moore (1996) ArticleTitleInhibition and kinetics of cytochrome P4503A activity in microsomes from rat, human, and cDNA-expressed human cytochrome P450 Drug Metab. Dispos. 24 940–947 Occurrence Handle1:CAS:528:DyaK28XlvV2itrg%3D Occurrence Handle8886602
G. T. Tucker J. B. Houston S. M. Huang (2001) ArticleTitleOptimizing drug development: strategies to assess drug metabolism/transporter interaction potential—towards a consensus Br. J. Clin. Pharmacol. 52 107–117 Occurrence Handle10.1046/j.0306-5251.2001.temp.1441.x Occurrence Handle1:STN:280:DC%2BD3Mvhs1WqsQ%3D%3D Occurrence Handle11453898
F. D. Boudinot W. J. Jusko (1984) ArticleTitleFluid shifts and other factors affecting plasma protein binding of prednisolone by equilibrium dialysis J. Pharm. Sci. 73 774–780 Occurrence Handle1:CAS:528:DyaL2cXkvVCit74%3D Occurrence Handle6204037
R. S. Obach A. E. Reed-Hagen (2002) ArticleTitleMeasurement of Michaelis constants for cytochrome P450-mediated biotransformation reactions using a substrate depletion approach Drug Metab. Dispos. 30 831–837 Occurrence Handle10.1124/dmd.30.7.831 Occurrence Handle1:CAS:528:DC%2BD38XltVGru7s%3D Occurrence Handle12065442
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohutsky, M.A., Chien, J.Y., Ring, B.J. et al. Predictions of the In Vivo Clearance of Drugs from Rate of Loss Using Human Liver Microsomes for Phase I and Phase II Biotransformations. Pharm Res 23, 654–662 (2006). https://doi.org/10.1007/s11095-006-9663-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9663-4