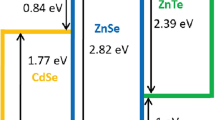
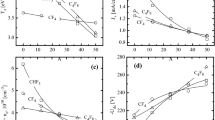
Abstract
Calculated values of viscosity, thermal and electrical conductivities of plasma formed in mixtures of silver (Ag) and silica (SiO2) are presented. The calculations, which assume local thermodynamic equilibrium, are performed for three pressures (1, 10 and 30 atm) in the temperature range from 4,000 to 30,000 K. All the data for the potential interactions and the necessary formulations to obtain values of transport coefficients are given in details. For atmospheric pressure, five mixtures (100% Ag, 75% Ag and 25% SiO2, 50% Ag and 50% SiO2, 25% Ag and 75% SiO2, 100% SiO2) in weight percentage are studied. In order to analyse the pressure influence on the transport coefficients, three samples of Ag–SiO2 mixtures (100% Ag, 50% Ag and 50% SiO2, 100% SiO2) in weight percentage are discussed for pressures of 1, 10 and 30 atm. In addition for the test case of oxygen plasma, we compare the computation code results with values obtained by other authors: discrepancies are found and explained.





Similar content being viewed by others
References
http://www.profuseinternational.com/sciencetech/index.html
Rochette D, Bussière W (2004) Plasma Sources Sci Technol 13:293
Rochette D, Clain S (2005) Int J Heat Fluid Flow 26(2):322
Coufal O (1998) J Phys D Appl Phys 31:2025
Bussière W, André P (2001) J Phys D Appl Phys 34:1657
Rochette D, Bussière W, André P (2004) Plasma Chem Plasma Process 24(3):475
Murphy AB (1995) Plasma Chem Plasma Process 15(2):279
Murphy AB, Arundell CJ (1994) Plasma Chem Plasma Process 14(4):451
Aubreton J, Fauchais P (1983) Revue Phys Appl 18:51
Hirschfelder JO, Curtiss CF, Byron Bird R (1964) Molecular theory of gases and liquids. Wiley, New york
Chapmann S, Cowling TG (1939) The mathematical Theory of Non-Uniform Gases, Cambridge University Press, Cambridge
Hulburt HM, Hirschfelder JO (1941) J Chem Phys 9:61
Hulburt HM, Hirschfelder JO (1961) J Chem Phys 35:1901
Rainwater JC, Biolsi L, Biolsi KJ, Holland PM (1983) J Chem Phys 79(3):1462
Zhang H-X, Balasubramanian K (1993) J Chem Phys 98(9):7092
Lide DR (eds) (2001) CRC handbook of chemistry and physics, 82nd edn. CRC Press, New York
Gurvich LV, Veyts IV, Alcock CB (1991) Thermodynamic properties of individual substances, 4th edn, vol 2, part 1. edited by Hemisphere Publishing Corporation
Peyerimhoff SD, Buenker RJ (1982) Chem Phys 72:111
Capitelli M, Ficocelli E (1972) J Phys B Atom Mol Phys 5:2066
Huber KP, Herzberg G (1979) Molecular spectra and molecular structure IV. Constants of diatomic molecules. Van Nostrand Reinhold Co.
Monchick L (1959) Phys Fluids 2(6):695
Sourd B, Aubreton J, Elchinger M-F, Labrot M, Michon U (2006) J Phys D Appl Phys 39:1105
Levin E, Partridge H, Stallcop JR (1989) Collision integrals and high temperature transport properties for N–N, O–O and N–O. Research Institute for Advanced Computer Science NASA Ames Research Center Technical Report 89.43
Sourd B, André P, Aubreton J, Elchinger M-F (2007) Plasma Chem Plasma Process 27:35
O’Hara H, Smith FJ (1971) Comput Phys Commun 2:47
Rainwater JC, Holland PM, Biolsi L (1982) J Chem Phys 77(1): 434
Mexmain JM (1983) Coefficients de transport dans un plasma à deux temperatures-Application au mélange Argon Oxygène. PhD, Limoges University
Aubreton J (1985) Etude des propriétés thermodynamiques et de transport dans des plasmas thermiques à l’équilibre et hors d’équilibre thermodynamiques: applications aux plasmas de mélange Ar-H2 et Ar-O2 (in French). Thèse d’état, Université de Limoges, France
Klein M, Smith F (1968) J Res Nat Bureau Stand-A Phys Chem 72 A(4):359
Kappen K (1994) Berechnung der Materialfunktionen von Hochdruckplasmen am Beispiel von Methanol. Lehrstuhl, Technische Universität München
Kee RJ, Lewis GD, Warnatz J, Coltrin ME, Miller JA, Moffat HK (1998) A Fortran computer code package for the evaluation of gas-phase, multicomponent tranport properties. Supercedes SAND86–8246B, http://www.osc.edu/PET/CCM/skeleton/software/chemkin/transport.pdf
Capitelli M, Gorse C, Longo S, Giordano D (1998) Transport properties of high temperature air species. AIAA 1–13
Timpel D, Scheerschmidt K, Garofolini SH (1997) J Non-Cryst Solids 221:187
http://xplor.csb.yale.edu/xplor/xploron-web/toppar/parameter.elements
Koalaga Z (1991) Contribution à l’étude expérimentale et théorique des plasmas d’arcs électriques laminés (in French). Thèse d’Université, No343, Université Blaise Pascal, France
Andre P, Brunet L, Bussière W, Caillard J, Lombard J, Picard J (2004) Eur Phys J Appl Phys 25:169
Diaz Penã M, Pando C, Renuncio JAR (1982) J Chem Phys 76(1):325
Diaz Penã M, Pando C, Renuncio JAR (1982) J Chem Phys 76(1):333
Ronchi C, Losilevski IL, Yakub E (2004) Equation of state of uranium dioxide. Data Collection. Springer-Verlag, Berlin
Dalgarno A (1958) Phil Trans Roy Soc Lond 250 A:426
Devoto RS (1967) Phys Fluids 10(2):354
Rapp D, Francis WE (1962) J Chem Phys 37(11):2631
Kihara T, Taylor MH, Hirschfelder JO (1960) Phys Fluids 3(5):715
Murphy AB (2000) Transport coefficients of hydrogen and argon-hydrogen plasmas 20(3):279
Devoto RS (1973) Phys Fluids 16(5):616
Mason EA, Munn RJ, Smith FJ (1967) Phys Fluids 10(8):1827
Itikawa Y (1974) Atomic Data Nuclear Data Tables 14(1):1
Spencer FE, Phelps AV (1976) Momentum transfer cross-sections and conductivity integrals for gases of MHD interest. In: Proc XVth Symp Eng Asp of MDH, Philadelphia PA, IX.9.1-IX.9.12
Robinson EJ, Geltmann (1967) Single- and double-quantum photodetachment of negative ions. Phys Rev 153(1):153_4
Fricke B (1986) J Chem Phys 84(2):862
Mayol R, Salvat F (1997) Atomic Data Nuclear Data Tables 65(1):55
André P (2005) Propriétés de transport pour l’étude de l’arc électrique. Exemple le cuivre. Colloque sur les arcs électriques (in French), 37–42, http://hal.ccsd.cnrs.fr
Aubreton A, Elchinger MF (2003) J Phys D Appl Phys 36(15):1798
Trajmar S, Williams W, Srivastava SK (1977) J Phys B Molec Phys 10(16):3323
Msezane AZ, Henry RJW (1986) Phys Rev A 33(3):1631
Scheibner KF, Hazi AU (1987) Phys Rev A 35(11):4869
Chervy B, Gleizes A (1998) Electrical conductivity in SF6 thermal plasma at low temperature (1000–5000 K). J Phys D Appl Phys 2557
Boulos MI, Fauchais P, Pfender E (1994) Thermal plasmas. Plenum Press, New York
Aubreton J, Bonnefoi C, Mexmain JM (1986) Revue Phys Appl 21:365
Devoto RS (1967) Phys Fluids 10(10):2105
Bonnefoi C (1975) Contribution au calcul théorique des coefficients de transport d’un plasma d’azote par la méthode de Chapman–Enskog à la 4ème approximation (in French). Thèse de 3ème, Université de Limoges, France, No. 75.9
Chervy B, Gleizes A, Razafinimanana M (1994) J Phys D Appl Phys 27:1193
Vanderslice JT, Weismann Stanley, Mason EA, Fallon RJ (1962) The Physics of Fluids 5(2):155
Muckenfuss C, Curtiss CF (1958) J Chem Phys 29(6):1273
Butler JN, Brokaw RS (1957) J Chem Phys 26(6):1636
Capitelli M, Gorse C, Longo S, Giordano D (2000) J Thermophys Heat Trans 14(2):259
Acknowledgements
The authors acknowledge many interesting and friendly discussions with Mr. Aubreton and Mrs. Elchinger from SPCTS at Limoges University. The authors also acknowledge, both for their financial support and their interest through many discussions, Mr. T. Rambaud and Mr. J.L. Gelet from Ferraz Shawmut, Mr. J.C. Perez Quesada from Mesa, Mr. F. Gentils from Schneider Electric.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A. Collision Integrals
The reduced collision integral is given by:
with
and χ (E, b) the scattering angle:
where r m is the minimum relative distance between the two atoms following the chosen potential V(r). b is the impact parameter. E is the relative total energy.
The reduced temperature T * is given by : T * = kT/ɛ where T is the temperature, k is Boltzmann constant and ɛ is the depth potential (Table 1).
Then the integral collision can be calculated from the reduced integral collision Ω *(l,s) ij (T * ) versus the reduced temperature: \(\overline{\Omega}_{_{ij} }^{(l,s)} \left(T \right)=\sigma ^{2}\overline{{\Omega }}_{ij}^{\ast(l,s)} \left({kT/\varepsilon } \right)\).
With σ the value of r for which V(r) = 0.
Appendix B. Formulation of Transport Coefficient
Electrical Conductivity
To calculate the electrical conductivity σ we used the third-order approximation of the Chapman–Enskog method [60, 61]:
where e is the electronic charge, n 1 the electron number density, k the Boltzmann constant, m 1 mass of an electron and T is the temperature. The parameters q ij are given in [60, 61].
Coefficient of Viscosity
To calculate the viscosity assuming that it is due to the heavy chemical species, we used the formulation given in [10, 62]:
where
with \(A_{ij}^\ast =\frac{\overline{\Omega}_{i j}^{(2,2)}}{\overline{\Omega}_{i j}^{(1,1)} }\), \(\eta _i =\frac{5}{16}\frac{1}{\overline{\Omega}_{i,i}^{(2,2)} }\sqrt\frac{k T}{N_a \pi }\sqrt{M_i }\), \(\eta _{ij} =\frac{5}{16}\frac{1}{\overline{\Omega}_{i,j}^{(2,2)} }\sqrt{\frac{k T}{N_a \pi }}\sqrt{\frac{2 M_i M_j }{M_i +M_j }}\)
x i and M i are the molar fraction and molar mass of i chemical species, N a is the Avogadro number, \(\overline{\Omega}_{i j}^{(l,m)} \) are collision integrals given in Appendix A and υ is the number of heavy chemical species.
Total Thermal Conductivity
The total thermal conductivity λ tot can be separated into four terms with a good accuracy [60, 63]:
where λ etr is the translational thermal conductivity of electrons, λ htr the translational thermal conductivity of heavy species particles, λint the internal thermal conductivity and λreact the chemical reaction thermal conductivity.
Thermal conductivity of electrons at the third approximation order is given by [60, 61]:
The translational thermal conductivity due to the heavy species in the second-order approximation can be written as [62, 64]:
where
with \(k_{ij} =\frac{75}{64}k^{3/2}\sqrt{\frac{N_a T}{\pi }}\sqrt{\frac{M_i +M_j }{2 M_i M_j }}\frac{1}{\overline{\Omega}_{ij}^{(2,2)} }\), \(A_{ik}^\ast =\frac{\overline{\Omega}_{ik}^{(2,2)} }{\overline{\Omega}_{ik}^{(1,1)} }\) and \(B_{ik}^\ast =\frac{5 \overline{\Omega}_{ik}^{\left({1,2} \right)} -4 \overline{\Omega}_{ik}^{(1,3)} }{\overline{\Omega}_{ik}^{(1,1)} }\)
The internal conductivity due to the effect of internal degrees of freedom is taken into account with the Eucken correction [10, 61, 62]:
with the internal conductivity of the i chemical species \(\lambda _{{\rm int} i} =\frac{PD_{ii} \left(1 \right)}{T}\left({\frac{C_{p i} }{R}-\frac{5}{2}} \right)\) and the use of the first approximation for the binary diffusion coefficients:
The formulation of the chemical reaction thermal conductivity was developed by Butler and Brokaw [65] and is written as:
with \(A_{ ij} =\sum_{k=1}^{\upsilon-1} {\sum_{l=k+1}^\upsilon {\frac{R T}{ P D_{kl} \left(1 \right)}} } x_k x_l \left({\frac{a_{ik} }{x_k }-\frac{a_{il} }{x_l }} \right)\left({\frac{a_{jk} }{x_k }-\frac{a_{jl} }{x_l }} \right)\) i reaction can be written as \(\sum_{k=1}^N {a_{ik} B^{k}} =0\) where B k is the symbol of the k chemical species. The μ reaction that we have to take into account must be linearly independent. For i reaction the enthalpy variation ΔH i is ΔH i = ∑ ν j=1 a ij H j where H j is the specific enthalpy of the j chemical species.
Appendix C. Chemical Composition
Rights and permissions
About this article
Cite this article
André, P., Bussière, W. & Rochette, D. Transport Coefficients of Ag–SiO2 Plasmas. Plasma Chem Plasma Process 27, 381–403 (2007). https://doi.org/10.1007/s11090-007-9086-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-007-9086-y
Keywords
- Transport coefficients
- Transport properties
- Electrical conductivity
- Viscosity
- Thermal conductivity
- Silver
- Silica
- Oxygen
- Plasma
- Fuses