Abstract
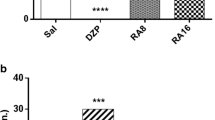
We synthesized a series of novel indole compounds containing aroylhydrazone moieties and evaluated them in mice to check their anticonvulsant activity. In the present study the most potent C3-modified derivative 3e, containing 2-furyl fragment was evaluated in kainate (KA)-induced status epilepticus (SE) and the consequences on oxidative stress and inflammation in the hippocampus in mice were explored. Melatonin was used as positive control while the melatonin receptor antagonist Luzindol was studied alone or in combination with melatonin or 3e, respectively. After intraperitoneal (i.p.) pre-treatment with melatonin 3e, Luzindol + melatonin and Luzindol + 3e for 7 days (melatonin and 3e—30 mg kg−1 or 60 mg kg−1, Luzindol 10 mg kg−1) the animals were i.p. injected with KA (30 mg kg−1, i.p.). The 3e decreased the SE-induced seizure intensity while melatonin suppressed seizures at the higher dose of 60 mg kg−1. Luzindol blocked the anticonvulsant effect of both Mel and 3e. The dose-dependent antioxidant effect of 3e measured by reduced glutathione (GSH) and total GSH in the hippocampus, was comparable to the effect of melatonin. Luzindol fully blocked the effect of melatonin but affected partially the antioxidant activity of 3e. The KA-induced increased amplifier of neuroinflammation high-mobility group box protein 1 (HMGB1) was neither alleviated by melatonin, nor by 3e. The activation by this DNA-binding protein receptor for advanced glycation end products (RAGE) was not affected by SE, melatonin and 3e pre-treatment. Our results suggest that the novel indole derivate 3e, containing 2-furyl fragment, might be clinically useful as an adjunct therapy against SE and concomitant oxidative stress.




Similar content being viewed by others
References
Agrawal N, Govender S (2011) Epilepsy and neuropsychiatric comorbidities. Adv Psychiatric Treat 17:44–53. https://doi.org/10.1192/apt.bp.108.006510
Trinka E, Cock H, Hesdorffer D, Rossetti AO, Scheffer IE, Shinnar S, Shorvon S, Lowenstein DH (2015) A definition and classification of status epilepticus: report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia 56:1515–1523. https://doi.org/10.1111/epi.13121
Kaneko Y, Pappas C, Malapira T, Vale F, Tajiri N, Borlongan CV (2017) Extracellular HMGB1 modulates glutamate metabolism associated with Kainic acid-induced epilepsy-like hyperactivity in primary rat neural cells. Cell Physiol Biochem 41(3):947–959. https://doi.org/10.1159/000460513
Baxevanis AD, Landsman D (1995) The HMG-1 box protein family: classification and functional relationships. Nucleic Acids Res 23:1604–1613. https://doi.org/10.1093/nar/23.9.1604
Ravizza T, Vezzani A (2013) Receptor for advanced glycation endproducts is upregulated in temporal lobe epilepsy and contributes to experimental seizures. Neurobiol Dis 58:102–114. https://doi.org/10.1016/j.nbd.2013.03.006
Lin L, Huang QX, Yang SS, Chu J, Wang JZ, Tian Q (2013) Melatonin in Alzheimer’s disease. Int J Mol Sci 14(7):14575–14593. https://doi.org/10.3390/ijms140714575
Molina-Carballo A, Muñoz-Hoyos A, Reiter RJ, Sánchez-Forte M, Moreno-Madrid F, Rufo-Campos M, Molina-Font JA, Acuna-Castroviejo D (1997) The utility of high doses of melatonin as adjunctive anticonvulsant therapy in a child with severe myoclonic epilepsy: two years’ experience. J Pineal Res 23:97–105. https://doi.org/10.1111/j.1600-079X.1997.tb00341.x
Petkova Zl, Tchekalarova J, Pechlivanova D, Moyanova Sl, Kortenska L, Mitreva R, Popov D, Markova P, Lozanov V, Atanasova D, Lazarov N, Stoynev A (2014) Treatment with melatonin after status epilepticus attenuates seizure activity and neuronal damage but does not prevent the disturbance in diurnal rhythms and behavioral alterations in spontaneously hypertensive rats in kainate model of temporal lobe epilepsy. Epilepsy Behav 31:198–208. https://doi.org/10.1016/j.yebeh.2013.12.013
Rocha AKA, Cipolla-Neto J, Amado D (2018) Epilepsy: neuroprotective, anti-inflammatory, and anticonvulsant effects of melatonin. Melatonin: medical uses and role in health and disease, Chap. 8, Nova Science Publishers, Inc. ISBN: 978-1-53612-987-8
Srinivasan V, Cardinali DP, Srinivasan US et al (2011) Therapeutic potential of melatonin and its analogs in Parkinson’s disease: focus on sleep and neuroprotection. Ther Adv Neurol Disord 4(5):297–317. https://doi.org/10.1177/1756285611406166
Tchekalarova J, Petkova Z, Pechlivanova D, Sl Moyanova, Kortenska L, Mitreva R, Lozanov V, Atanasova D, Lazarov N, Al Stoynev (2013) Prophylactic treatment with melatonin after status epilepticus: effects on epileptogenesis, neuronal damage and behavioral changes in kainate model of temporal lobe epilepsy. Epilepsy Behav 27:174–187. https://doi.org/10.1016/j.yebeh.2013.01.009
Tchekalarova J, Moyanova S, De Fusco A, Ngomba RT (2015) The role of the melatoninergic system in epilepsy and comorbid psychiatric disorders. Brain Res Bull 119:80–92. https://doi.org/10.1016/j.brainresbull.2015.08.006
Yeganeh-Salehpour M, Mollica A, Momtaz S, Sanadgol N, Farzaei MH (2019) Melatonin and multiple sclerosis: from plausible neuropharmacological mechanisms of action to experimental and clinical evidence. Clin Drug Investig 39(7):607–624. https://doi.org/10.1007/s40261-019-00793-6
Atanasova M, Petkova Zl, Pechlivanova D, Dragomirova P, Blazhev, Tchekalarova J (2013) Strain differences in the effect of long-term treatment with melatonin on kainic acid-induced status epilepticus, oxidative stress and the expression of heat shock proteins. Pharmacol Biochem Behav 111:44–50. https://doi.org/10.1016/j.pbb.2013.08.006
Angelova V, Rangelov M, Todorova N, Dangalov M, Andreeva-Gateva P, Kondeva-Burdina M, Karabeliov V, Shivachev B, Tchekalarova J (2019) Discovery of novel indole-based aroylhydrazones as anticonvulsants: pharmacophore-based design. Bioorg Chem 90:103028. https://doi.org/10.1016/j.bioorg.2019.103028
Marchev S, Andreeva-Gateva P, Tzoneva R, Surcheva S, Tzonev A, Kamenova K, Angelova VT, Tchekalarova J, Vlaskovska M (2019) Analgesic activity of some aroylhydrazone-based molecular hybrids with antiseizure activity: in vivo and in silico evaluations. Biotechnol Biotechnol Equip 33:98–107. https://doi.org/10.1080/13102818.2018.1555009
Andreeva-Gateva P, Tchekalarova J, Kamenova K, Angelova VT, Surcheva SI (2021) Aryl hydrocarbon receptors in indole derivative treated mice: Neuropharmacological perspectives. Acta Medica Bulg 48(1):25–33. https://doi.org/10.2478/amb-2021-0004
Tchekalarova J, Angelova V, Todorova N, Andreeva-Gateva P, Rangelov M (2019) Evaluation of the anticonvulsant effect of novel melatonin derivatives in the intravenous pentylenetetrazol seizure test in mice. Eur J Pharmacol 20:863:172684. https://doi.org/10.1016/j.ejphar.2019.172684
Tchekalarova J, Ivanova N, Nenochovska Z, Tzoneva R, Stoyanova T, Uzunova V, Surcheva S, Tzonev A, Angelova V, Andreeva-Gateva P (2020) Evaluation of neurobiological and antioxidant effects of novel melatonin analogs in mice. Saudi Pharm J 28(12):1566–1579. https://doi.org/10.1016/j.jsps.2020.10.004
Zeng L, Zhang H, Xu C, Bian Y, Xu X, Xi Q, Zhang RJ (2013) Zhejiang Univ-Sci B Biomed Biotechnol 14(11):1004–1012. https://doi.org/10.1631/jzus.B1300138
Chen Y, Huang X-J, Yu N, Xie Y, Zhang K, Wen F et al (2015) HMGB1 contributes to the expression of P-glycoprotein in mouse epileptic brain through toll-like receptor 4 and receptor for advanced glycation end products. PLoS ONE 10:e0140918. https://doi.org/10.1371/journal.pone.0140918
Fu L, Liu K, Wake H, Teshigawara K, Yoshino T, Takahashi H et al (2017) Therapeutic effects of anti-HMGB1 monoclonal antibody on pilocarpineinduced status epilepticus in mice. Sci Rep 7:1179. https://doi.org/10.1038/s41598-017-01325-y
Maroso M, Balosso S, Ravizza T, Liu J, Aronica E, Iyer AM, Rossetti C, Molteni M, Casalgrandi M, Manfredi A, Bianchi ME, Vezzani A (2010) Nat Med 16:413–419. https://doi.org/10.1038/nm.2127
Iori V, Maroso M, Rizzi M, Iyer AM, Vertemara R, Carli M, Vezzani A (2013) Receptor for advanced glycation endproducts is upregulated in temporal lobe epilepsy and contributes to experimental seizures. Neurobiol Dis 58:102–114. https://doi.org/10.1016/j.nbd.2013.03.006
Ito M, Takahashi H, Yano H, Shimizu YI, Yano Y, Ishizaki Y et al (2017) High mobility group box 1 enhances hyperthermia-induced seizures and secondary epilepsy associated with prolonged hyperthermia-induced seizures in developing rats. Metab Brain Dis 32:2095–2104. https://doi.org/10.1007/s11011-017-0103-4
Vezzani A, Granata T (2005) Brain inflammation in epilepsy: experimental and clinical evidence. Epilepsia 46:1724–1743. https://doi.org/10.1111/j.1528-1167.2005.00298.x
Acknowledgements
This work was supported by the National Science Fund of Bulgaria, Grant DN13/16 21.12.2017.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11064_2021_3447_MOESM2_ESM.xls
Raw O.D. values for all standards and experimental samples for reduced GSH measurements. Supplementary file1 (XLS 66 kb)
Rights and permissions
About this article
Cite this article
Tchekalarova, J., Stoyanova, T., Tzoneva, R. et al. The Anticonvulsant Effect of a Novel Indole-Related Compound in the Kainate-Induced Status Epilepticus in Mice: The Role of the Antioxidant and Anti-inflammatory Mechanism. Neurochem Res 47, 327–334 (2022). https://doi.org/10.1007/s11064-021-03447-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03447-2