Abstract
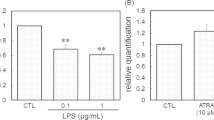
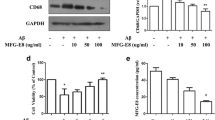
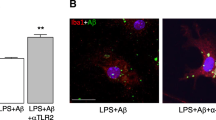
Activation of glial cells has been observed in neurodegenerative diseases including Alzheimer’s disease (AD). Aggregation of amyloid β (Aβ) is profusely observed as characteristic pathology in AD brain. In our previous study using microglial cell line BV-2, tissue-type transglutaminase (TG2) was found to be involved in phagocytosis (Kawabe et al., in Neuroimmunomodulation 22(4):243–249, 2015; Kawabe et al., Neurochem Res 2017). In the present study, we examined whether TG2 and milk fat globule EGF factor 8 protein (MFG-E8), an adaptor protein promotes macrophage to engulf apoptotic cells, were involved in Aβ endocytosis. When the neuronal/glial mixed culture was stimulated freshly prepared Aβ1−42 for 3 days, the incorporation of Aβ was observed by immunofluorescence staining technique in Iba-1-positive microglia. Cystamine, a broad competitive inhibitor of TGs, suppressed it. When aggregated Aβ was added to the mixed culture, the immunoreactivity of MFG-E8 surrounding Aβ was observed, and then followed by microglial endocytosis. Using western blotting technique, MFG-E8 was detected in cell lysate of astrocyte culture, and was also detected in the medium. When microglia culture was incubated with astrocyte conditioned medium, MFG-E8 levels in microglia tended to increase. It is likely that microglia might utilize MFG-E8 released from astrocytes as well as that expressed in themselves in order to endocytose Aβ aggregation. Furthermore, we confirmed that MFG-E8 could bind with TG2 in microglia culture by immunoprecipitate technique. These results suggest that microglia might uptake Aβ as a complex of aggregated Aβ/MFG-E8/TG2.





Similar content being viewed by others
Abbreviations
- Aβ:
-
Amyloid beta
- ACM:
-
Astrocytes conditioned medium;
- AD:
-
Alzheimer’s disease
- BSA:
-
Bovine serum albumin
- CBB:
-
Coomassie brilliant blue
- CNS:
-
Central nervous system
- DMEM:
-
Dulbecco’s modified Eagle medium
- DMSO:
-
Dimethyl sulfoxide
- EGF:
-
Epidermal growth factor
- FBS:
-
Fetal bovine serum
- FITC:
-
Fluorescein isothiocyanate
- GFAP:
-
Glial fibrillary acidic protein
- HRP:
-
Horseradish peroxidase
- MFG-E8:
-
Milk fat globule EGF factor 8 protein
- LPS:
-
Lipopolysaccharide
- NO:
-
Nitric oxide
- PBS:
-
Phosphate-buffered saline
- PS:
-
Phosphatidylserine
- TG:
-
Transglutaminase
- VR:
-
Vitronectin receptor.
References
Kreutzberg GW (1996) Microglia: a sensor for pathological events in the CNS. Trends Neurosci 19:312–318
Nakamura Y (2002) Regulating factors for microglia activation. Biol Pharm Bull 25:945–953
Neniskyte U, Neher JJ, Brown GC (2011) Neuronal death induced by nanomolar amyloid β is mediated by primary phagocytosis of neurons by microglia. J Biol Chem 286(46):39904–39913
Anderson CM, Swanson RA (2000) Astrocyte glutamate transport: review of properties, regulation, and physiological functions. Glia 32:1–14
Abbott NJ (2002) Astrocyte-endothelial interactions and blood-brain barrier permeability. J Anat 200:629–638
Correale J, Villa A (2009) Cellular elements of the blood-brain barrier. Neurochem Res 34:2067–2077
Pratten MK, Lloyd JB (1986) Pinocytosis and phagocytosis: the effect of size of a particulate substrate on its mode of capture by rat peritoneal macrophages cultured in vitro. Biochim Biophys Acta 881:307–313
Shanbhag AS, Jacobs JJ, Black J, Galante JO, Glant TT (1994) Macrophage/particle interactions: effect of size, composition and surface area. J Biomed Mat Res 28:81–90
Aukunuru JV, Kompella UB (2002) In vitro delivery of nano- and micro-particles to human retinal pigment epithelial (ARPE-19) cells. Drug Del Technol 2:50–57
Fadok VA, Chimini G (2001) The phagocytosis of apoptotic cells. Semin Immunol 13:365–372
Ravichandran KS (2011) Beginnings of a good apoptotic meal: the find-me and eat-me signaling pathways. Immunity 35(4):445–455
Oshima K, Aoki N, Kato T, Kitajima K, Matsuda T (2002) Secretion of a peripheral membrane protein, MFG-E8, as a complex with membrane vesicles. Eur J Biochem 269:1209–1218
Hanayama R, Tanaka M, Miwa K, Shinohara A, Iwamatsu A, Nagata S (2002) Identification of a factor that links apoptotic cells to phagocytes. Nature 417(6885):182–187
Hanayama R, Nagata S (2005) Impaired involution of mammary glands in the absence milk fat globule EGF factor 8. Proc Natl Acad Sci USA 102(46):16886–16891
Neher JJ, Neniskyte U, Zhao JW, Bal-Price A, Tolkovsky AM, Brown GC (2011) Inhibition of microglial phagocytosis is sufficient to prevent inflammatory neuronal death. J Immunol 186:4973–4983
Fricker M, Neher JJ, Zhao JW, Théry C, Tolkovsky AM, Brown GC (2012) MFG-E8 mediates primary phagocytosis of viable neurons during neuroinflammation. J Neurosci 32(8):2657–2666
Griffin M, Casadio R, Bergamini CM (2002) Transglutaminases: Nature’s biological glues. Biochem J 368:377–396
Jeitner TM, Muma NA, Battaile KP, Cooper, AJL (2009) Transglutaminase activation in neurodegenerative diseases. Future Neurol 4:449–467
Basso M, Berlin J, Xia L, Sleiman SF, Ko B, Haskew-Layton R, Kim E, Antonyak MA, Cerione RA, Iismaa S, Willis D, Cho S, Ratan RR (2012) Transglutaminase inhibition protects against oxidative stress-induced neuronal death downstream of pathological ERK activation. J Neurosci 32(19):6561–6569
Belkin AM (2011) Extracellular TG2: emerging functions and regulation. FEBS J 278:4704–4716
Grosso H, Mouradian MM (2012) Transglutaminase 2: biology, relevance to neurodegenerative diseases and therapeutic implications. Pharmacol Ther 133:392–410
Szondy Z, Sarang Z, Molnár P, Németh T, Piacentini M, Mastroberardino PG, Falasca L, Aeschlimann D, Kovács J, Kiss I, Syegeydi E, Lakos G, Rajnavölgyi É, Birckbichler PJ, Melino G, Fésüs L (2003) Tranglutaminase 2-/-mice reveal a phagocytosis-associated crosstalk between macrophages and apoptotic cells. Proc Natl Acad Sci USA 100(13):7812–7817
Tóth B, Garabuczi, É., Sarang Z, Vereb G, Vámosi G, Aeschimann D, Blaskó B, Bécsi B, Erdõdi B, Lacy-Hulbert A, Zhang A, Falasca L, Birge RB, Balajthy Z, Melino G, Fésüs L, Szondy Z (2009) Transglutaminase 2 is needed for the formation of an efficient phagocyte portal in macrophage engulfing apoptosis cells. J Immunol 182(4):2084–2092
Kawabe K, Takano K, Moriyama M, Nakamura Y (2015) Lipopolysaccharide-stimulated transglutaminase 2 expression enhances endocytosis activity in mouse microglial cell line BV-2. Neuroimmunomodulation 22(4):243–249
Kawabe K, Takano K, Moriyama M, Nakamura Y (2017) Amphotericin B increases transglutaminase 2 expression associated with upregulation of endocytotic activity in mouse microglial cell line BV-2. Neurochem Res 42:1488–1495
Takano K, Shiraiwa M, Moriyama M, Nakamura Y (2010) Transglutaminase 2 expression induced by lipopolysaccharide stimulation together with NO synthase induction in cultured astrocytes. Neurochem Int 57(7):812–818
Nakamura Y, Si QS, Kataoka K (1999) Lipopolysaccharide-induced microglial activation in culture: temporal profiles of morphological change and release of cytokines and nitric oxide. Neurosci Res 35:95–100
Watanabe T, Totsuka R, Miyatani S, Kurata S, Sato S, Katoh I, Kobayashi S, Ikawa Y (2005) Production of the long and short forms of MFG-E8 by epidermal keratinocytes. Cell Tissue Res 321(2):185–193
Akiyama H, Mori M, Saido T (1999) Occurrence of the diffuse amyloid β-protein (Aβ) deposits with numerous Aβ-containing glial cells in the cerebral cortex of patients with Alzheimer’s disease. Glia 25:324–331
Mitrasinovic OM, Murphy GM Jr (2003) Microglial overexpression of the M-CSF receptor augments phagocytosis of opsonized Abeta. Neurobiol Aging 24(6):807–815
Wang Z, Collighan RJ, Gross SR, Danen EH, Orend G, Telci D, Griffin M (2010) RGD-independent cell adhesion via a tissue transglutaminase-fibronectin matrix promotes fibronectin fibril deposition and requires syndecan-4/2 and α5β1 integrin co-signaling. J Biol Chem 285(51):40212–40229
Acknowledgements
This work was supported in part by JSPS KAKENHI Grant No. JP15J12259 to K.K., JP26850209 to K.T., JP26450447 to M.M., and JP15K07768 to Y.N.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kawabe, K., Takano, K., Moriyama, M. et al. Microglia Endocytose Amyloid β Through the Binding of Transglutaminase 2 and Milk Fat Globule EGF Factor 8 Protein. Neurochem Res 43, 41–49 (2018). https://doi.org/10.1007/s11064-017-2284-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-017-2284-y