Abstract
Background
Genetic variations in immune signaling genes may have regulatory effect on phenotypic heterogeneity of immune cells and immune functions, hence promoting tumor growth.
Purpose
We compared the frequencies of potentially functional CD38 gene single nucleotide polymorphisms rs1130169 (T > C) in 86 healthy controls and 90 colorectal cancer (CRC) cases to assess their association with cancer risk and CD38 gene expression.
Results
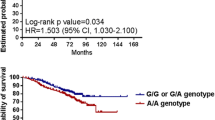
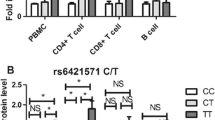
The association between allele C rs1130169 and CRC risk was observed. Allele C was also significantly correlated with an increased CD38 mRNA level and CD38 positive cell percentages in peripheral blood of healthy controls that could be a possible explanation for CRC risk in C allele carriers. In peripheral blood of CRC patients CD38 mRNA and serum soluble CD38 protein levels significantly differed from those in healthy controls. Calculation of the CD38 full-length and with the third exon deletion mRNA ratio in corresponding samples showed that the mRNA isoform ratio was significantly higher in CRC cases than in controls. It suggests that alternative splicing regulates elevation of CD38 full-length mRNA level in peripheral blood of CRC patients. We also have observed higher expression levels of CD38 full-length mRNA in peripheral blood of CRC patients with lymph node metastases compared to patients without metastases.
Conclusion
This study indicated biological significance of rs1130169 variations that can alter differences in CRC risk by regulating CD38 gene expression.


Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Chang CM, Chia VM, Gunter MJ et al (2013) Innate immunity gene polymorphisms and the risk of colorectal neoplasia. Carcinogenesis 34:2512–2520. https://doi.org/10.1093/carcin/bgt228
Cho YA, Lee J, Oh JH, Chang HJ, Sohn DK, Shin A, Kim J (2019) Genetic risk score, combined lifestyle factors and risk of colorectal cancer. Cancer Res Treat: Off J Korean Cancer Assoc 51(3):1033–1040. https://doi.org/10.4143/crt.2018.447
Hogan KA, Chini CC, Chini EN (2019) The multi-faceted ecto-enzyme CD38: roles in immunomodulation, cancer, aging, and metabolic diseases. Front Immunol 10:1187. https://doi.org/10.3389/fimmu.2019.01187
Takasawa S (2022) CD38–Cyclic ADP-ribose signal system in physiology, biochemistry, and pathophysiology. Int J Mol Sci 23(8):4306. https://doi.org/10.3390/ijms23084306
Higashida H, Yokoyama S, Huang JJ, Liu L, Ma W-J, Akther S, Higashida C, Kikuchi M, Minabe Y, Munesue T (2012) Social memory, amnesia, and autism: brain oxytocin secretion is regulated by NAD+ metabolites and single nucleotide polymorphisms of CD38. Neurochem Int 61:828–838. https://doi.org/10.1016/j.neuint.2012.01.030
Shahrabi S, Ghanavat M, Behzad MM, Purrahman D, Saki N (2020) CD markers polymorphisms as prognostic biomarkers in hematological malignancies. Oncol Rev 14(2):99–100. https://doi.org/10.4081/oncol.2020.466
Jamroziak K, Szemraj Z, Grzybowska-Izydorczyk O et al (2009) CD38 gene polymorphisms contribute to genetic susceptibility to B-cell chronic lymphocytic leukemia: evidence from two case-control studies in polish caucasians. Cancer Epidemiol Biomarkers Prev 18(3):945–953. https://doi.org/10.1158/1055-9965
Hartman WR, Pelleymounter LL, Moon I, Kalari K, Liu M, Wu TY, Escande C, Nin V, Chini EN, Weinshilboum RM (2010) CD38 expression, function, and gene resequencing in a human lymphoblastoid cell line-based model system. Leuk Lymphoma 51:1315–1325. https://doi.org/10.3109/10428194.2010.483299
Lebedev M, Egorova NI, Sholkina MN, Vilkov SA, Baryshnikov AJ, Novikov VV (2004) Serum levels of different forms of soluble CD38 antigen in burned patients. Burns 30:552–556. https://doi.org/10.1016/j.burns.2004.01.029
Cikos S, Bukovska A, Koppel J (2007) Relative quantification of mRNA: comparison of methods currently used for real-time PCR data analysis. BMC Mol Biol 8:113. https://doi.org/10.1186/1471-2199-8-113
Mallone R, Ferrua S, Morra M, Zocchi E, Mehta K, Notarangelo LD, Malavasi F (1998) Characterization of a CD38 like 78 kilodalton soluble protein released from B cell lines derived from patients with X linked agammaglobulinemia. J Clin Invest 101:2821–2830. https://doi.org/10.1172/JCI1068
Perenkov AD, Novikov DV, Sakharnov NA, Aliasova AV, Utkin OV, Alu B, Novikov VV (2012) Heterogeneous expression of CD38 gene in tumor tissue in patients with colorectal cancer. Mol Biol (Mosk) 46:786–791
Ogino S, Nosho K, Irahara N, Shima K, Baba Y, Kirkner GJ, Mino-Kenudson M, Giovannucci EL, Meyerhardt JA, Fuchs CS (2010) Negative lymph node count is associated with survival of colorectal cancer patients, independent of tumoral molecular alterations and lymphocytic reaction. Am J Gastroenterol 105:420–433. https://doi.org/10.1038/ajg.2009.578
Bahri R, Bollinger A, Bollinger T, Orinska Z, Bulfone-Paus S (2012) Ectonucleotidase CD38 demarcates regulatory, memory-like CD8 + T cells with IFN-c-mediated suppressor activities. PLoS ONE 7:e45234. https://doi.org/10.1371/journal.pone.0045234
Deaglio S, Robson SC (2011) Ectonucleotidases as regulators of purinergic signaling in thrombosis, inflammation, and immunity. Adv Pharmacol 61:301–332. https://doi.org/10.1016/B978-0-12-385526-8.00010-2
Masuda K, Kornberg A, Miller J, Lin S, Suek N, Botella T, Secener KA, Bacarella AM, Cheng L, Ingham M, Rosario V, Al-Mazrou AM, Lee-Kong SA, Kiran RP, Stoeckius M, Smibert P, Del Portillo A, Oberstein PE, Sims PA, Yan KS, Han A (2022) Multiplexed single-cell analysis reveals prognostic and nonprognostic T cell types in human colorectal cancer. JCI Insight 7(7):e154646. https://doi.org/10.1172/jci.insight.154646
Acknowledgements
We would like to thank all participants and Alexander V. Kalugin for their help in samples collection. Research was supported by the Ministry of education and science of Russian Federation to perform public works in the field of scientific activities: task 2014/134(2575). This work was supported by the Strategic Academic Leadership Program of Kazan Federal University.
Funding
No funding was obtained for this study.
Author information
Authors and Affiliations
Contributions
ADP, VVN conceived and designed the experiments. DVN, ADP, SVS, NIK made contributions to the data collection and analysis and wrote the manuscript. ADP, DVN prepared Figs. 1 and 2. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no potential conflict of interest associated with this manuscript.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki and this research was reviewed and approved by the Ethics Committee of the Nizhny Novgorod I.N. Blokhina Research Institute of Epidemiology and Microbiology November 25, 2021, protocol No. 6.
Consent to participate
All subjects gave written informed consent to the use of their blood samples for research purposes.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Novikov, D.V., Perenkov, A.D., Shumilova, S.V. et al. CD38 gene polymorphism rs1130169 contribution to the increased gene expression and risk of colorectal cancer (pilot study). Mol Biol Rep 51, 63 (2024). https://doi.org/10.1007/s11033-023-09034-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-023-09034-8