Abstract
Purpose
Patients who have been diagnosed with prostate cancer (PCa) typically have a dismal outlook and few therapeutic choices available to them, because the precise pathogenesis of the disease is not yet fully understood. The presence of HP1α, also known as the heterochromatin protein 1α, is required for the creation of higher-order chromatin structures. However, little is known about HP1α that serves roles in the pathogenesis of PCa. The primary purpose of our research was to investigate alterations in the levels of HP1α expression and to plan a series of tests to validate the function of HP1α in PCa.
Method
Information on HP1α expression in PCa and benign prostatic hyperplasia (BPH) tissues were gathered using the Cancer Genome Atlas (TCGA) and Gene Expression Profiling Interactive Analysis (GEPIA) databases. RT-qPCR, western blotting, and immunohistochemistry (IHC) were used to assess HP1α mRNA and protein expression in several human PCa tissues and cell lines. The CCK8 assay, clone formation assay, and transwell assay were used to examine biological activities including cell proliferation, migration, and invasion. The expression of proteins connected to apoptosis and the epithelial-mesenchymal transition (EMT) was examined using Western blot. The tumorigenic effect of HP1α was also verified by in vivo experiments.
Result
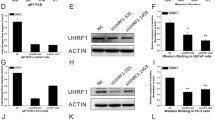
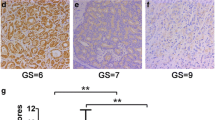
HP1α expression was much higher in PCa than in BPH tissues and cells, and was positively correlated with the Gleason score of PCa. In vitro experiments showed that HP1α knockdown could inhibit the ability of proliferation, invasion, and migration of PC3 and LNCaP cells, and promote cell apoptosis and EMT. In vivo experiments showed that HP1α knockdown inhibited tumorigenesis in mice.
Conclusion
Our findings indicate that HP1α expression promotes PCa development and might be a novel therapeutic target for the diagnosis or treatment of PCa.





Similar content being viewed by others
Data Availability
All data included in this study are available upon request by contact with the corresponding author.
References
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73:17–48. https://doi.org/10.3322/caac.21763
Nyame YA, Gore JL (2020) What goes up must come down: identifying truth from global prostate Cancer Epidemiology. Eur Urol 77:53–54. https://doi.org/10.1016/j.eururo.2019.09.018
Center MM, Jemal A, Lortet-Tieulent J, Ward E, Ferlay J, Brawley O, Bray F (2012) International variation in prostate cancer incidence and mortality rates. Eur Urol 61:1079–1092. https://doi.org/10.1016/j.eururo.2012.02.054
Choi E, Buie J, Camacho J, Sharma P, de Riese WTW (2022) Evolution of Androgen Deprivation Therapy (ADT) and its new emerging modalities in prostate Cancer: an update for practicing urologists, Clinicians and Medical Providers. Res Rep Urol 14:87–108. https://doi.org/10.2147/RRU.S303215
Mills IG (2014) Maintaining and reprogramming genomic androgen receptor activity in prostate cancer. Nat Rev Cancer 14:187–198. https://doi.org/10.1038/nrc3678
Zhong S, Peng S, Chen Z, Chen Z, Luo JL (2022) Choosing kinase inhibitors for Androgen Deprivation Therapy-Resistant prostate Cancer. Pharmaceutics 14. https://doi.org/10.3390/pharmaceutics14030498
Ku SY, Rosario S, Wang Y, Mu P, Seshadri M, Goodrich ZW, Goodrich MM, Labbe DP, Gomez EC, Wang J, Long HW, Xu B, Brown M, Loda M, Sawyers CL, Ellis L, Goodrich DW (2017) Rb1 and Trp53 cooperate to suppress prostate cancer lineage plasticity, metastasis, and antiandrogen resistance. Science 355:78–83. https://doi.org/10.1126/science.aah4199
Cucchiara V, Yang JC, Mirone V, Gao AC, Rosenfeld MG, Evans CP (2017) Epigenomic regulation of androgen receptor signaling: potential role in prostate Cancer therapy. Cancers (Basel) 9. https://doi.org/10.3390/cancers9010009
Sekhoacha M, Riet K, Motloung P, Gumenku L, Adegoke A, Mashele S (2022) Prostate Cancer Review: Genetics, diagnosis, Treatment Options, and alternative approaches. Molecules 27. https://doi.org/10.3390/molecules27175730
Kumar R, Sena LA, Denmeade SR, Kachhap S (2022) The testosterone paradox of advanced prostate cancer: mechanistic insights and clinical implications. Nat Rev Urol doi. https://doi.org/10.1038/s41585-022-00686-y
Castellon EA, Indo S, Contreras HR (2022) Cancer Stemness/Epithelial-Mesenchymal Transition Axis Influences Metastasis and Castration Resistance in prostate Cancer: potential therapeutic target. Int J Mol Sci 23. https://doi.org/10.3390/ijms232314917
Akova Olken E, Aszodi A, Taipaleenmaki H, Saito H, Schonitzer V, Chaloupka M, Apfelbeck M, Bocker W, Saller MM (2022) SFRP2 overexpression induces an osteoblast-like phenotype in prostate Cancer cells. Cells 11. https://doi.org/10.3390/cells11244081
Eguchi T, Csizmadia E, Kawai H, Sheta M, Yoshida K, Prince TL, Wegiel B, Calderwood SK (2022) SCAND1 reverses epithelial-to-mesenchymal transition (EMT) and suppresses prostate Cancer Growth and Migration. Cells 11. https://doi.org/10.3390/cells11243993
Rezaeian A, Khatami F, Heidari Keshel S, Akbari MR, Mirzaei A, Gholami K, Mohammadi Farsani R, Aghamir SMK (2022) The effect of mesenchymal stem cells-derived exosomes on the prostate, bladder, and renal cancer cell lines. Sci Rep 12:20924. https://doi.org/10.1038/s41598-022-23204-x
Zhang J, Hu Z, Horta CA, Yang J (2022) Regulation of epithelial-mesenchymal transition by tumor microenvironmental signals and its implication in cancer therapeutics. Semin Cancer Biol 88:46–66. https://doi.org/10.1016/j.semcancer.2022.12.002
Vad-Nielsen J, Nielsen AL (2015) Beyond the histone tale: HP1alpha deregulation in breast cancer epigenetics. Cancer Biol Ther 16:189–200. https://doi.org/10.1080/15384047.2014.1001277
Her C, Phan TM, Jovic N, Kapoor U, Ackermann BE, Rizuan A, Kim YC, Mittal J, Debelouchina GT (2022) Molecular interactions underlying the phase separation of HP1alpha: role of phosphorylation, ligand and nucleic acid binding. Nucleic Acids Res 50:12702–12722. https://doi.org/10.1093/nar/gkac1194
Ci X, Hao J, Dong X, Choi SY, Xue H, Wu R, Qu S, Gout PW, Zhang F, Haegert AM, Fazli L, Crea F, Ong CJ, Zoubeidi A, He HH, Gleave ME, Collins CC, Lin D, Wang Y (2018) Heterochromatin protein 1alpha mediates Development and aggressiveness of neuroendocrine prostate Cancer. Cancer Res 78:2691–2704. https://doi.org/10.1158/0008-5472.CAN-17-3677
Wang Y, Wang Y, Ci X, Choi SYC, Crea F, Lin D, Wang Y (2021) Molecular events in neuroendocrine prostate cancer development. Nat Rev Urol 18:581–596. https://doi.org/10.1038/s41585-021-00490-0
Vad-Nielsen J, Jakobsen KR, Daugaard TF, Thomsen R, Brugmann A, Sorensen BS, Nielsen AL (2016) Regulatory dissection of the CBX5 and hnRNPA1 bi-directional promoter in human breast cancer cells reveals novel transcript variants differentially associated with HP1alpha down-regulation in metastatic cells. BMC Cancer 16:32. https://doi.org/10.1186/s12885-016-2059-x
Kim JM, Shin Y, Lee S, Kim MY, Punj V, Shin HI, Kim K, Koh JM, Jeong D, An W (2018) MacroH2A1.2 inhibits prostate cancer-induced osteoclastogenesis through cooperation with HP1alpha and H1.2. Oncogene 37:5749–5765. https://doi.org/10.1038/s41388-018-0356-3
Jeon YH, Kim GW, Kim SY, Yi SA, Yoo J, Kim JY, Lee SW, Kwon SH (2022) Heterochromatin protein 1: a multiplayer in Cancer Progression. Cancers (Basel) 14. https://doi.org/10.3390/cancers14030763
Zhang K, Myllymaki SM, Gao P, Devarajan R, Kytola V, Nykter M, Wei GH, Manninen A (2017) Oncogenic K-Ras upregulates ITGA6 expression via FOSL1 to induce anoikis resistance and synergizes with alphav-class integrins to promote EMT. Oncogene 36:5681–5694. https://doi.org/10.1038/onc.2017.177
Gasparics A, Sebe A (2022) Forward Genetic Screens as Tools to investigate role and mechanisms of EMT in Cancer. Cancers (Basel) 14. https://doi.org/10.3390/cancers14235928
Zhang P, Wei Y, Wang L, Debeb BG, Yuan Y, Zhang J, Yuan J, Wang M, Chen D, Sun Y, Woodward WA, Liu Y, Dean DC, Liang H, Hu Y, Ang KK, Hung MC, Chen J, Ma L (2014) ATM-mediated stabilization of ZEB1 promotes DNA damage response and radioresistance through CHK1. Nat Cell Biol 16:864–875. https://doi.org/10.1038/ncb3013
Funding
The author declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
ZSY: project development, data collection, data analysis, manuscript writing. LHR: data analysis, manuscript writing. SC: data collection, data analysis. CFH: data analysis, manuscript reviewing. KSS: data analysis, manuscript reviewing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Ethics statement
The study was approved and consented by the Ethics Committee of the Second Hospital of Tianjin Medical University, Tianjin, China. (No. KY2019K077 and No. YN2019Y70).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, S., Li, H., Shen, C. et al. HP1α promotes the progression of prostate cancer. Mol Biol Rep 50, 4459–4468 (2023). https://doi.org/10.1007/s11033-023-08373-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08373-w