Abstract
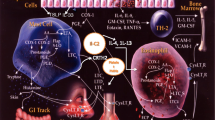
Aspirin-exacerbated respiratory disease (AERD) is characterized by immune cells dysfunction. This study aimed to investigate the molecular mechanisms involved in AERD pathogenesis. Relevant literatures were identified by a PubMed search (2005–2019) of english language papers using the terms “Aspirin-exacerbated respiratory disease”, “Allergic inflammation”, “molecular mechanism” and “mutation”. According to the significant role of inflammation in AERD development, ILC-2 is known as the most important cell in disease progression. ILC-2 produces cytokines that induce allergic reactions and also cause lipid mediators production, which activates mast cells and basophils, ultimately. Finally, Monoclonal antibody and Aspirin desensitization in patients can be a useful treatment strategy for prevention and treatment.
Similar content being viewed by others
Data availability
The datasets used analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ARED:
-
Aspirin Exacerbated Respiratory Disease
- NSAIDS:
-
Nonsteroidal anti-inflammatory drugs
- BAG-1:
-
BCL-2 associated athanogenet
- TLR:
-
Toll Like Receptor
References
Renjiao L, Fengming L (2020) The safety and efficacy of aspirin desensitization combined with long-term aspirin therapy in Aspirin-exacerbated respiratory disease. J Investigational AllergolClinImmunol 30(5):327–333
Taniguchi M, Mitsui C, Hayashi H, Ono E, Kajiwara K, Mita H et al (2019) Aspirin-exacerbated respiratory disease (AERD): current understanding of AERD. Allergol Inter 68(3):289–295
Kowalski ML, Makowska J, Blanca M, Bavbek S, Bochenek G, Bousquet J et al (2011) Hypersensitivity to nonsteroidal anti-inflammatory drugs (NSAIDs)–classification, diagnosis and management: review of the EAACI/ENDA# and GA2LEN/HANNA. Allergy 66(7):818–829
Stevenson DD, Szczeklik A (2006) Clinical and pathologic perspectives on aspirin sensitivity and asthma. J Allergy ClinImmunol 118(4):773–786
Pleskow WW, Stevenson DD, Mathison DA, Simon RA, Schatz M, Zeiger RS (1982) Aspirin desensitization in aspirin-sensitive asthmatic patients: clinical manifestations and characterization of the refractory period. J Allergy ClinImmunol 69(1):11–19
Laidlaw TM (2018) Pathogenesis of NSAID-induced reactions in aspirin-exacerbated respiratory disease. World J Otorhinolaryngol-Head Neck Surg 4(3):162–168
Aksu K, Kurt E, Alatas Ö, Gülbas Z, editors. Effect of aspirin desensitization on T-cell cytokines and plasma lipoxins in aspirin-exacerbated respiratory disease. Allergy & Asthma Proceedings; 2014.
Seyedian SS, Nokhostin F, Malamir MD (2019) A review of the diagnosis, prevention, and treatment methods of inflammatory bowel disease. Journal of medicine and life 12(2):113
Lassila R, Editor (2016) Platelet function tests in bleeding disorders. Seminars in thrombosis and hemostasis. Thieme Medical Publishers, NY
Liu T, Barrett NA, Kanaoka Y, Buchheit K, Laidlaw TM, Garofalo D et al (2019) Cysteinyl leukotriene receptor 2 drives lung immunopathology through a platelet and high mobility box 1-dependent mechanism. Mucosal Immunol 12(3):679–690
Maugeri N, Evangelista V, Celardo A, Dell’Elba G, Martelli N, Piccardoni P et al (1994) Polymorphonuclear leukocyte-platelet interaction: role of P-selectin in thromboxane B2 and leukotriene C4 cooperative synthesis. ThrombHaemost 72(09):450–456
Liu T, Laidlaw TM, Katz HR, Boyce JA (2013) Prostaglandin E2 deficiency causes a phenotype of aspirin sensitivity that depends on platelets and cysteinylleukotrienes. ProcNatlAcadSci 110(42):16987–16992
Rashidi H, Shahbazian H, Nokhostin F, Bahadoram M, Payami SP (2015) The association between concentration of Uric Acid and metabolic syndrome among adolescents. Tehran University Medical Journal TUMS Publications 73(8):600–607
Laidlaw TM, Cahill KN, Cardet JC, Murphy K, Cui J, Dioneda B et al (2019) A trial of type 12 purinergic (P2Y12) receptor inhibition with prasugrel identifies a potentially distinct endotype of patients with aspirin-exacerbated respiratory disease. J Allergy ClinImmunol. 143(1):316–324
Tunjungputri RN, van der Ven AJ, Riksen N, Rongen G, Tacke S, van den Berg T et al (2015) Differential effects of platelets and platelet inhibition by ticagrelor on TLR2-and TLR4-mediated inflammatory responses. ThrombHaemost 113(05):1035–1045
Dinkla S, van Cranenbroek B, van der Heijden WA, He X, Wallbrecher R, Dumitriu IE et al (2016) Platelet microparticles inhibit IL-17 production by regulatory T cells through P-selectin. Blood 127(16):1976–1986
Bandeira-Melo C, Bozza PT, Weller PF (2002) The cellular biology of eosinophil eicosanoid formation and function. J Allergy ClinImmunol 109(3):393–400
Rajaei E, Jalali MT, Pezeshki SMS, Rezaeeyan H, Maniati M, Elyasi M et al (2020) Dose HLA-B5, 7, 8, 27, and 51 Antigens Associated to Behcet’s disease? A Study in Southwestern Iran. Curr Rheumatology Rev 16(2):120–124
Choi M-R, Xu J, Lee S, Yeon S-H, Park S-K, Rha K-S et al (2020) Chloroquine treatment suppresses mucosal inflammation in a mouse model of eosinophilic chronic rhinosinusitis. Allergy Asthma Immunol Res 12(6):994
Wong C, Wang C, Ip W, Tian Y, Lam C (2005) Role of p38 MAPK and NF-kB for chemokine release in coculture of human eosinophils and bronchial epithelial cells. ClinExpImmunol 139(1):90–100
El-Shazly A, Roncarati P, Lejeune M, Lefebvre P, Delvenne P (2017) Tyrosine kinase inhibition is an important factor for gene expression of CRTH2 in human eosinophils and lymphocytes: A novel mechanism for explaining eosinophils recruitment by the neuro-immune axis in allergic rhinitis. IntImmunopharmacol 45:180–186
Steinke JW, Negri J, Liu L, Payne SC, Borish L (2014) Aspirin activation of eosinophils and mast cells: implications in the pathogenesis of aspirin-exacerbated respiratory disease. J Immunol 193(1):41–47
Minshall EM, Leung DY, Martin RJ, Song YL, Cameron L, Ernst P et al (1997) Eosinophil-associated TGF-β1 mRNA expression and airways fibrosis in bronchial asthma. Am J Respir Cell MolBiol 17(3):326–333
Wang D, Yan Z, Bu L, An C, Deng B, Zhang J et al (2019) Protective effect of peptide DR8 on bleomycin-induced pulmonary fibrosis by regulating the TGF-β/MAPK signaling pathway and oxidative stress. ToxicolApplPharmacol 382:114703
Esmaeilzadeh H, Nabavi M, Amirzargar AA, Aryan Z, Arshi S, Bemanian MH et al (2015) HLA-DRB and HLA-DQ genetic variability in patients with aspirin-exacerbated respiratory disease. Am J Rhinol Allergy 29(3):e63–e69
Lin D, Lin H, Xiong X (2014) Expression and role of BAG-1 in eosinophilic and non-eosinophilic chronic rhinosinusitis with nasal polyps. Inflammation 37(6):1912–1918
Kikuchi K, Abe S, Kodaira K, Yukawa T, Hozawa S, Mochizuki H et al (2013) Heat shock protein 70 gene polymorphisms in Japanese patients with aspirin-exacerbated respiratory disease. J Investig Med 61(4):708–714
Doherty TA, Broide DH (2018) Lipid regulation of group 2 innate lymphoid cell function: moving beyond epithelial cytokines. J Allergy ClinImmunol 141(5):1587–1589
Doherty TA (2015) At the bench: understanding group 2 innate lymphoid cells in disease. J LeukocBiol 97(3):455–467
von Moltke J, O’Leary CE, Barrett NA, Kanaoka Y, Austen KF, Locksley RM (2017) Leukotrienes provide an NFAT-dependent signal that synergizes with IL-33 to activate ILC2s. J Exp Med 214(1):27–37
Rozsasi A, Heinemann A, Keck T (2016) Release of cyclooxygenase-2 and lipoxin A4 from blood leukocytes in aspirin-exacerbated respiratory disease. Allergy Rhinology 7(3):0172
MacLean Scott E, Solomon LA, Davidson C, Storie J, Palikhe NS, Cameron L (2018) Activation of Th2 cells downregulates CRTh2 through an NFAT1 mediated mechanism. PLoS ONE 13(7):e0199156
Bachert C, Zhang L, Gevaert P (2015) Current and future treatment options for adult chronic rhinosinusitis: focus on nasal polyposis. J Allergy ClinImmunol 136(6):1431–1440
Buchheit KM, Cahill KN, Katz HR, Murphy KC, Feng C, Lee-Sarwar K et al (2016) Thymic stromal lymphopoietin controls prostaglandin D2 generation in patients with aspirin-exacerbated respiratory disease. J Allergy ClinImmunol 137(5):1566–1576
Mitchell PD, O’Byrne PM (2017) Biologics and the lung: TSLP and other epithelial cell-derived cytokines in asthma. PharmacolTher 169:104–112
Toki S, Goleniewska K, Reiss S, Zhang J, Bloodworth MH, Stier MT et al (2018) Glucagon-like peptide 1 signaling inhibits allergen-induced lung IL-33 release and reduces group 2 innate lymphoid cell cytokine production in vivo. J Allergy ClinImmunol 142(5):1515–1528
Cho YJ, Kim S, Orimo K, Saito H, Matsumoto K, Morita H et al (2020) Does the Difference in Microbial Patterns in the Airways Induce Distinct Endotypes of Asthma? Allergy, asthma & immunology research 12(3):375–377
Liang Y, Yi P, Yuan DMK, Jie Z, Kwota Z, Soong L et al (2019) IL-33 induces immunosuppressive neutrophils via a type 2 innate lymphoid cell/IL-13/STAT6 axis and protects the liver against injury in LCMV infection-induced viral hepatitis. Cell MolImmunol 16(2):126–137
Kim H, Yu S, Choi H, Effects of particulate air pollution on tuberculosis development in seven major cities of Korea from, (2010) to 2016: methodological considerations involving long-term exposure and time lag. Epidemiology and health 2020:42
Chang H, Shin S, Lee T, Bae D, Park J, Kim Y et al (2015) Development of a genetic marker set to diagnose aspirin-exacerbated respiratory disease in a genome-wide association study. Pharmacogenomics J 15(4):316–321
Kurosawa M, Yukawa T, Hozawa S, Mochizuki H (2015) Recent advance in investigation of gene polymorphisms in Japanese patients with aspirin-exacerbated respiratory disease. AllergolImmunopathol 43(1):92–100
Song Y-S, Yang E-M, Kim S-H, Jin HJ, Park H-S (2012) Effect of genetic polymorphism of ALOX15 on aspirin-exacerbated respiratory disease. Int Arch Allergy Immunol 159(2):157–161
Losol P, Kim S-H, Shin YS, Ye YM, Park H-S (2013) A genetic effect of IL-5 receptor α polymorphism in patients with aspirin-exacerbated respiratory disease. Experimental Mol Med 45(3):e14
Claesson H-E (2009) On the biosynthesis and biological role of eoxins and 15-lipoxygenase-1 in airway inflammation and Hodgkin lymphoma. Prostaglandins Other Lipid Mediat 89(3–4):120–125
Pasaje CFA, Kim JH, Park BL, Cheong HS, Chun JY, Park TJ et al (2010) Association of SLC6A12 variants with aspirin-intolerant asthma in a Korean population. Ann Hum Genet 74(4):326–334
Corry DB, Kheradmand F (2007) A new link to airway obstruction in asthma. Nat Med 13(7):777–778
Lu WY, Inman M (2009) γ-aminobutyric acid nurtures allergic asthma. ClinExp Allergy 39(7):956–961
Haybar H, Shahrabi S, Rezaeeyan H, Jodat H, Saki N (2019) Strategies to inhibit arsenic trioxide-induced cardiotoxicity in acute promyelocyticleukemia. J Cell Physiol 234(9):14500–14506
Park BL, Kim T-H, Kim J-H, Bae JS, Pasaje CFA, Cheong HS et al (2013) Genome-wide association study of aspirin-exacerbated respiratory disease in a Korean population. Hum Genet 132(3):313–321
Shin S-W, Park J, Kim Y-J, Uh S-t, Choi BW, Kim M-k et al (2012) A highly sensitive and specific genetic marker to diagnose aspirin-exacerbated respiratory disease using a genome-wide association study. DNA Cell Biol 31(11):1604–1609
Kim J-H, Park B-L, Pasaje CFA, Bae JS, Park JS, Park SW et al (2012) Association of FANCC polymorphisms with FEV1 decline in aspirin exacerbated respiratory disease. MolBiol Rep 39(3):2385–2394
Pasaje CFA, Bae JS, Park B-L, Kim J-H, Cheong HS, Uh S-T et al (2012) Genetic variations in KIFC1 and the risk of aspirin exacerbated respiratory disease in a Korean population: an association analysis. MolBiol Rep 39(5):5913–5919
Kobuke K, Furukawa Y, Sugai M, Tanigaki K, Ohashi N, Matsumori A et al (2001) ESDN, a novel neuropilin-like membrane protein cloned from vascular cells with the longest secretory signal sequence among eukaryotes, is up-regulated after vascular injury. J BiolChem 276(36):34105–34114
Park T-J, Kim J-H, Park B-L, Cheong HS, Bae JS, Pasaje CF et al (2012) Potential association of DCBLD2 polymorphisms with fall rates of FEV1 by aspirin provocation in Korean asthmatics. J Korean Med Sci 27(4):343
Narayanankutty A, Palma-Lara I, Pavón-Romero G, Pérez-Rubio G, Camarena Á, Teran LM et al (2016) Association of TRPM3 polymorphism (rs10780946) and aspirin-exacerbated respiratory disease (AERD). Lung 194(2):273–279
Kim J-H, Park B-L, Pasaje CFA, Kim Y, Bae JS, Park JS et al (2012) Contribution of the OBSCN nonsynonymous variants to aspirin exacerbated respiratory disease susceptibility in Korean population. DNA Cell Biol 31(6):1001–1009
Losol P, Palikhe NS, Lee JW, Palikhe S, Kim M-A, Yang E-M et al (2015) Association of P2RY12 polymorphisms with eosinophil and platelet activation in patients with aspirin-exacerbated respiratory disease. Annals Allergy Asthma Immunol 114(5):423–424
Losol P, Shin YS (2013) A genetic effect of IL-5 receptor a polymorphism in patients with aspirin-exacerbated respiratory disease. ExpMol Med 45(3):1–7
Kim S-H, Yang E-M, Lee H-N, Choi G-S, Ye Y-M, Park H-S (2010) Association of the CCR3 gene polymorphism with aspirin exacerbated respiratory disease. Respir Med 104(5):626–632
Palikhe N, Kim SH, Cho BY, Ye YM, Choi GS, Park HS (2010) Genetic variability in CRTH2 polymorphism increases eotaxin-2 levels in patients with aspirin exacerbated respiratory disease. Allergy 65(3):338–346
Cook KA, Stevenson DD (2016) Current complications and treatment of aspirin-exacerbated respiratory disease. Expert Rev Respiratory Med 10(12):1305–1316
Yeung WYW, Park HS (2020) Update on the management of nonsteroidal anti-inflammatory drug hypersensitivity. Yonsei Med J 61(1):4
Education NA, Program P (2007) Expert panel report 3 (EPR-3): guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy ClinImmunol 120(5 Suppl):S94–S138
Rodríguez-Jiménez JC, Moreno-Paz FJ, Terán LM, Guaní-Guerra E (2018) Aspirin exacerbated respiratory disease: current topics and trends. Respir Med 135:62–75
Simon RA, Dazy KM, Waldram JD (2015) Aspirin-exacerbated respiratory disease: characteristics and management strategies. Expert Rev ClinImmunol 11(7):805–817
Choi J-H, Kim J-H, Park H-S (2015) Upper airways in aspirin-exacerbated respiratory disease. CurrOpin Allergy ClinImmunol 15(1):21–26
Kennedy JL, Stoner AN, Borish L (2016) Aspirin-exacerbated respiratory disease: Prevalence, diagnosis, treatment, and considerations for the future. Am J RhinolAller 30(6):407–413
Berges-Gimeno MP, Simon RA, Stevenson DD (2002) The natural history and clinical characteristics of aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol 89(5):474–478
Berges-Gimeno M, Simon R, Stevenson D (2002) The effect of leukotriene-modifier drugs on aspirin-induced asthma and rhinitis reactions. ClinExp Allergy 32(10):1491–1496
Steinke JW, Wilson JM (2016) Aspirin-exacerbated respiratory disease: pathophysiological insights and clinical advances. Journal of asthma and allergy 9:37
Dahlén S-E, Malmstrom K, Nizankowska E, Dahlén B, Kuna P, Kowalski M et al (2002) Improvement of aspirin-intolerant asthma by montelukast, a leukotriene antagonist: a randomized, double-blind, placebo-controlled trial. Am J RespirCrit Care Med 165(1):9–14
Berges-Gimeno MP, Simon RA, Stevenson DD (2003) Early effects of aspirin desensitization treatment in asthmatic patients with aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol 90(3):338–341
Spies JW, Valera FCP, Cordeiro DL, Mendonça TNd, Leite MGJ, Tamashiro E et al (2016) The role of aspirin desensitization in patients with aspirin-exacerbated respiratory disease (AERD). Brazilian J Otorhinolaryngol 82(3):263–268
Woo S-D, Luu QQ, Park H-S (2020) NSAID-exacerbated respiratory disease (NERD): from pathogenesis to improved care. Frontiers in Pharmacology 11:1147
Bobolea I, Barranco P, Fiandor A, Cabañas R, Quirce S (2010) Omalizumab: a potential new therapeutic approach for aspirin-exacerbated respiratory disease. J InvestigAllergolClinImmunol 20(5):448–449
Hayashi H, Mitsui C, Nakatani E, Fukutomi Y, Kajiwara K, Watai K et al (2016) Omalizumab reduces cysteinyl leukotriene and 9 [alpha], 11 [beta]-prostaglandin F2overproduction in aspirin-exacerbated respiratory disease. J Allergy ClinImmunol 137(5):1585
Gevaert P, Calus L, Van Zele T, Blomme K, De Ruyck N, Bauters W et al (2013) Omalizumab is effective in allergic and nonallergic patients with nasal polyps and asthma. J Allergy ClinImmunol 131(1):110–116
Gevaert P, Van Bruaene N, Cattaert T, Van Steen K, Van Zele T, Acke F et al (2011) Mepolizumab, a humanized anti–IL-5 mAb, as a treatment option for severe nasal polyposis. J Allergy ClinImmunol 128(5):989–995
Wenzel S, Ford L, Pearlman D, Spector S, Sher L, Skobieranda F et al (2013) Dupilumab in persistent asthma with elevated eosinophil levels. N Engl J Med 368(26):2455–2466
Mahdavinia M, Batra PS, Codispoti C (2019) Mepolizumab utility in successful aspirin desensitization in aspirin-exacerbated respiratory disease in a refractory case. Ann Allergy Asthma Immunol 123(3):311–312
Varricchi G, Bagnasco D, Borriello F, Heffler E, Canonica GW (2016) Interleukin-5 pathway inhibition in the treatment of eosinophilic respiratory disorders: evidence and unmet needs. CurrOpin Allergy ClinImmunol 16(2):186
Buchheit KM, Laidlaw TM (2016) Update on the management of aspirin-exacerbated respiratory disease. Allergy, asthma & immunology research 8(4):298
Thaçi D, Simpson EL, Beck LA, Bieber T, Blauvelt A, Papp K et al (2016) Efficacy and safety of dupilumab in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical treatments: a randomised, placebo-controlled, dose-ranging phase 2b trial. The Lancet 387(10013):40–52
Laidlaw TM, Boyce JA (2015) Platelets in patients with aspirin-exacerbated respiratory disease. Journal of Allergy and Clinical Immunology 135(6):1407–1414
Uller L, Mathiesen JM, Alenmyr L, Korsgren M, Ulven T, Högberg T et al (2007) Antagonism of the prostaglandin D 2 receptor CRTH2 attenuates asthma pathology in mouse eosinophilic airway inflammation. Respir Res 8(1):1–10
Walgama ES, Hwang PH (2017) Aspirin-exacerbated respiratory disease. OtolaryngolClin North Am 50(1):83–94
Spies JW, Valera FCP, Cordeiro DL, de Mendonça TN, Leite MGJ, Tamashiro E et al (2016) The role of aspirin desensitization in patients with aspirin-exacerbated respiratory disease (AERD). Brazilian journal of otorhinolaryngology 82(3):263–268
Maleknia M, Valizadeh A, Pezeshki S, Saki N. Immunomodulation in leukemia: cellular aspects of anti-leukemic properties. Clinical and Translational Oncology. 2019:1–10.
Uller L, Mathiesen JM, Alenmyr L, Korsgren M, Ulven T, Högberg T et al (2007) Antagonism of the prostaglandin D 2 receptor CRTH2 attenuates asthma pathology in mouse eosinophilic airway inflammation. Respir Res 8(1):16
Acknowledgements
We wish to thank all our colleagues in Vali-e-Asr Hospital and Allied Health Sciences School, Zanjan University of Medical Sciences.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
MN has conceived the manuscript and revised it. HH and NS wrote the manuscript. MM and AB design the tables. M,M conducted revised.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hybar, H., Saki, N., Maleknia, M. et al. Aspirin exacerbated respiratory disease (AERD): molecular and cellular diagnostic & prognostic approaches. Mol Biol Rep 48, 2703–2711 (2021). https://doi.org/10.1007/s11033-021-06240-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06240-0