Abstract
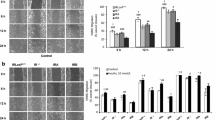
After confirmation of the presence of adiponectin (ADPN) receptors and intra-cellular binding proteins in coronary artery smooth muscle cells (VSMC), we tested the hypotheses that, in acute insulin resistance: (i) the activation/inactivation of metabolic and mitogenic insulin signaling pathways are inversely affected by ADPN and, (ii) changes in VSMC migration/proliferation rates correlate with signal activity/inactivity. In primary cultures of VSMC exposed to high glucose and palmitate plus insulin, the expression of PI-3 kinase (Akt and m-TOR), MAP-Kinase (Erk and p-38) molecules, and inflammatory markers (TLR-4 and IkB-α) were assessed with Western blot, in the absence/presence of AdipoRon (AR). Migration and proliferation rates were measured in similar experimental conditions. There were decreases of ~ 25% (p-Akt) and 40–60% (p-mTOR) expressions with high glucose/palmitate, which reversed when AR was added were. Elevations in p-Erk and p-p38 expressions were obliterated by AR. Although, no changes were detected with high glucose and palmitate, when AR was added, a decline in inflammatory activity was substantiated by a ~ 50% decrease in TLR-4 and 40–60% increase in IkBα expression. Functional assays showed 10–20% rise in VSMC proliferation with high glucose and palmitate, but addition of AR lead to 15–25% decline. The degree of VSMC migration was reduced with AR addition by ~ 15%, ~ 35% and 55%, in VSMC exposed to 5 mM, 25 mM glucose and 25 mM + 200 µM palmitate, respectively. Changes in intracellular molecular messaging in experiments mimicking acute insulin resistance suggest that anti-inflammatory and anti-atherogenic actions of ADPN in VSMC are mediated via insulin signaling pathways.



Similar content being viewed by others
References
American Heart Association (2019) Heart disease and stroke statistics— update. AHA, Dallas. https://professional.heart.org/professional/ScienceNews/UCM_503383_Heart-Disease-and-Stroke-Statistics. Accessed 14 August 2019
Cecilia C, Wang L, Hess CN, Hiatt WR, Goldfine AB (2016) Atherosclerotic cardiovascular disease and heart failure in type 2 diabetes mellitus—mechanisms, management, and clinical considerations. Circulation 133:2459–2502. https://doi.org/10.1161/CIRCULATIONAHA.116.022194
Einarson TR, Acs A, Ludwig C, Panton UH (2018) Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc Diabetol 17(83):2–19. https://doi.org/10.1186/s12933-018-0728-6
DeFronzo RA (2008) Claude Bernard Lecture, European Association for the Study of Diabetes Annual Meeting, Rome, Italy
Cersosimo E, DeFronzo RA (2006) Insulin resistance and endothelial dysfunction: the road map for cardiovascular diseases. Diabetes Metab Res Rev 22:423–436
Van Gaal LF, Mertens IL, De Block CE (2006) Mechanisms linking obesity with cardiovascular disease. Nature 444:875–880
Cersosimo E, XiaoJing X, Musi N (2015) Role of insulin signaling in vascular smooth muscle cell migration, proliferation and inflammation. Am J Physiol Cell Physiol 302(4):C652–C657
Cersosimo E, XiaoJing X, Upala S, Triplitt C, Musi N (2014) Acute insulin resistance stimulates and insulin sensitization attenuates vascular smooth muscle cell migration and proliferation. Physiol Rep 2(8):e12123
Davidson M, Meyer PM, Haffner S, Feinstein S, D’Agostino R Sr, Kondos GT, Perez A, Chen Z, Mazzone T (2008) Increased high-density lipoprotein cholesterol predicts the pioglitazone-mediated reduction of carotid intima-media thickness progression in patients with type 2 diabetes mellitus. Circulation 117:2123–2130
Mazzone T, Meyer PM, Feinstein SB, Davidson MH, Kondos GT, D`gostino RB, Perez A, Provost JC, Haffner SM (2006) Effect of pioglitazone compared with glimepiride on carotid intima-media thickness in type 2 diabetes: a randomized trial. JAMA 296:2572–2581
Nissen SE, Nichols SJ, Wolski K, Nesto R, Kupfer S, Perez A, Jure H, Larchelliere R, Staniloae CS, Mavromatis K, Saw J, Hu B, Lincoff AM, Tuzcu EM (2008) Comparison of pioglitazone vs. glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes (The Periscope Randomized Controlled Trial). JAMA 299:1561–1573
Dormandy J, Charbonnel B, Erdmann E, Massi-Benedetti M, Skene A, PROactive Study Group (2005) Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet 366(9493):1279–1289
Kernan WN, Viscoli CM, Furie KL et al (2016) Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med 374:1321–1331. https://doi.org/10.1056/NEJMoa1506930
Wajcberg E, Fernandez M, DeFronzo RA, Cersosimo E (2007) Relationship between improvements in metabolic control, lipids, and vascular reactivity in Mexican American with Type 2 diabetes mellitus treated with Pioglitazone. J Clin Endo Metabol 92:1256–1262
Kizer JR (2013) Tangled threesome: adiponectin, insulin sensitivity, and adiposity. Can Mendelian randomization sort out causality? Diabetes 62(4):1007–1009. https://doi.org/10.2337/db12-1673
Han SH, Quon MJ, Kim J, Koh KK (2007) Adiponectin and cardiovascular disease response to therapeutic interventions. JACC 49(5):531–538. https://doi.org/10.1016/j.jacc.2006.08.061
Ruan H, Dong LQ (2016) Adiponectin signaling and function in insulin target tissues. J Mol Cell Biol 8(2):101–109
Okada-Iwabu M, Yamauchi T, Iwabu M, Honma T, Hamagami K, Matsuda K, Yamaguchi M, Tanabe H, Kimura-Someya T, Shirouzu M, Ogata H, Tokuyama K, Ueki K, Nagano T, Tanaka A, Yokoyama S, Kadowaki T (2013) A small-molecule AdipoR agonist for type 2 diabetes and short life in obesity. Nature 503(7477):493–499. https://doi.org/10.1038/nature12656
Esfandiarei M, Larn JT, Yazdi SA, Kariminia A, Dorado JN, Kuzeljevic B, Syyong HT, Hu K, van Breemen C (2011) Diosgenin modulates vascular smooth muscle cell function by regulating cell viability, migration and calcium homeostasis. J Phamacol Exp Ther 336(3):925–939
Pan H, Chen J, Xu J, Ma R (2009) Anti-fibrotic effect by activation of peroxisome proliferator-activated receptor gamma in corneal fibroblasts. Mol Vis 15:2279–2286
Belfort R, Mandarino L, Kashyap S, Wirfel K, Pratipanawatr T, Berria R, DeFronzo RA, Cusi K (2005) Dose-response effect of elevated plasma free fatty acid on insulin signaling. Diabetes 54:1640–1648
Pajvani UB, Hawkins M, Combs TP, Rajala MW, Doebber T, Berger JP, Wagner JA, Wu M, Knopps A, Xiang AH (2004) Complex distribution, not absolute amount of adiponectin, correlates with thiazolidinedione-mediated improvement in insulin sensitivity. J Biol Chem 279:12152–12162. https://doi.org/10.1074/jbc.M311113200
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-(Delta-Delta C (T) Method. Methods 25:402–408
Reyna SM, Ghosh S, Tantiwong P, Meka CS, Eagan P, Jenkinson CP, Cersosimo E, DeFronzo RA, Coletta RK, Sriwijitkamol A, Musi N (2008) Elevated toll-like receptor 4 expression and signaling in muscle from insulin-resistant subjects. Diabetes 57:2595–2602
Prasad KM, Jin X, Wissan A, Abou A, Naudi SM (2014) Real-time vascular mechanosensation through ex vivo artery perfusion. Biol Proced 16:60–73
White PW, Abularrage CJ, Weiswasser JM, Kellicut DC, Arora S, Sidawa AN (2006) Hypoxia attenuates insulin-induced proliferation and migration of human diabetic infra-popliteal vascular smooth muscle cells. Ann Vasc Surg 20(3):381–386
Tong NW, Wang Q, Liu XJ, Wu X (2005) In vitro study to the effects of pioglitazone on endothelial cell functions of human umbilical vein and the mechanism involved. J Sichuan Univ Med Sci Edit 36(4):525–528 (Original in Chinese)
Mao X, Kikani CK, Riojas RA, Langlais P, Wang L, Ramos FJ, Fang Q, Christ-Roberts CY, Hong JY, Kim RY et al (2006) APPL1 binds to adiponectin receptors and mediates adiponectin signaling and function. Nat Cell Biol 8:516–523. https://doi.org/10.1038/ncb1404
Xin X, Zhou L, Reyes CM, Liu F, Dong LQ (2011) Appl1 mediates adiponectin-stimulated p38 mapk activation by scaffolding the tak1-mkk3-p38 mapk pathway. Am J Physiol Endocrinol Metab 2011(300):E103–E110. https://doi.org/10.1152/ajpendo.00427.2010
Wang C, Xin X, Xiang R, Ramos FJ, Liu M, Lee HJ, Chen H, Mao X, Kikani CK, Liu F et al (2009) “Yin-Yang” regulation of adiponectin signaling by APPL isoforms in muscle cells. J Biol Chem 284:31608–31615. https://doi.org/10.1074/jbc.M109.010355
Ryu AK, Galan XX, Feng D, Abdul-Ghani MA, Zhou L, Wang C, Li C, Holmes BM, Sloane LB, Austad SN, Guo S, Musi N, DeFronzo RA, Deng C, White MF, Liu F, Dong LQ (2014) APPL1 potentiates insulin sensitivity by facilitating the binding of IRS1/2 to the insulin receptor. Cell Rep 7:1227–1238
Yoon MJ, Lee GY, Chung J-J, Ahn YH, Hong SH, Kim JB (2006) Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of amp-activated protein kinase, p38 mitogen-activated protein kinase, and peroxisome proliferator-activated receptor α. Diabetes 55:2562–2570. https://doi.org/10.2337/db05-1322
Lu X, Kassab GS (2004) Nitric oxide is significantly reduced in ex vivo porcine arteries during reverse flow because of increased superoxide production. J Physiol 561:575–582
Pajvani UB, Du X, Combs TP, Berg AH, Rajala MW, Schulthess T, Engel JR, Brownlee M, Scherer PE (2003) Structure-function studies of the adipocyte-secreted hormone acrp30/adiponectin implications for metabolic regulation and bioactivity. J Biol Chem 278:9073–9085. https://doi.org/10.1074/jbc.M207198200
Steinberg HO, Chaker H, Leaming R et al (1996) Obesity/insulin resistance is associated with endothelial dysfunction. J Clin Invest 97:2601–2610
Cusi K, Maezono K, Osman A, Pendergrass M, Patti ME, Pratipanawatr T, DeFronzo RA, Kahn RC, Mandarino L (2000) Insulin resistant differentially affects PI-3 kinase and MAP-kinase-mediated signaling in human muscle. J Clin Invest 105:311–320
Matsuda M, Shimomura I, Sata M, Arita Y, Nishida M, Maeda N, Kumada M, Okamoto Y, Nagaretani H, Nishizawa H (2002) Role of adiponectin in preventing vascular stenosis the missing link of adipo-vascular axis. J Biol Chem 277:37487–37491. https://doi.org/10.1074/jbc.M206083200
Wang C, Mao X, Wang L, Liu M, Wetzel MD, Guan KL, Dong LQ, Liu F (2007) Adiponectin sensitizes insulin signaling by reducing p70 S6Kinase-mediated serine phosphorylation of IRS-1. J Biol Chem 282(11):7991–7996. https://doi.org/10.1074/jbc.M700098200
Gogg S, Smith U, Jansson PA (2009) Increased MAPK activation and impaired insulin signaling in subcutaneous microvascular endothelial cells in type 2 diabetes: the role of endothelin-1. Diabetes 58:2238–2245
Okamoto Y, Kihara S, Ouchi N, Nishida M, Arita Y, Kumada M, Ohashi K, Sakai N, Shimomura I, Kobayashi H (2002) Adiponectin reduces atherosclerosis in apolipoprotein e-deficient mice. Circulation 106:2767–2770. https://doi.org/10.1161/01.CIR.0000042707.50032.19
Acknowledgements
This work was supported in part by funds from the Kronkosky Foundation (E.C.), The University of Texas Health and the Texas Diabetes Institute, University Health System, San Antonio, Texas; and by National Institutes of Health R01 Grant DK102965 (L. Q. D.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no potential conflicts of interests for all the authors in this manuscript.
Informed consent
The Institutional Review Board at the UTHSCSA granted full exception for a consenting process/informed consent, in view of the nature of these in vitro experiments.
Research involving human and animal rights
All data presented in this manuscript were generated in experiments conducted exclusively with frozen cell cultures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cersosimo, E., Xu, X., Terasawa, T. et al. Anti-inflammatory and anti-proliferative action of adiponectin mediated by insulin signaling cascade in human vascular smooth muscle cells. Mol Biol Rep 47, 6561–6572 (2020). https://doi.org/10.1007/s11033-020-05707-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05707-w