Abstract
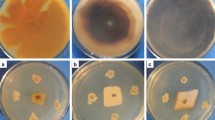
In this work, we identified a gene from Theobroma cacao L. genome and cDNA libraries, named TcGlu2, that encodes a β-1,3-1,4-glucanase. The TcGlu2 ORF was 720 bp in length and encoded a polypeptide of 239 amino acids with a molecular mass of 25.58 kDa. TcGlu2 contains a conserved domain characteristic of β-1,3-1,4-glucanases and presented high protein identity with β-1,3-1,4-glucanases from other plant species. Molecular modeling of TcGlu2 showed an active site of 13 amino acids typical of glucanase with β-1,3 and 1,4 action mode. The recombinant cDNA TcGlu2 obtained by heterologous expression in Escherichia coli and whose sequence was confirmed by mass spectrometry, has a molecular mass of about 22 kDa (with His-Tag) and showed antifungal activity against the fungus Moniliophthora perniciosa, causal agent of the witches’ broom disease in cacao. The integrity of the hyphae membranes of M. perniciosa, incubated with protein TcGlu2, was analyzed with propidium iodide. After 1 h of incubation, a strong fluorescence emitted by the hyphae indicating the hydrolysis of the membrane by TcGlu2, was observed. To our knowledge, this is the first study of a cacao β-1,3-1,4-glucanase expression in heterologous system and the first analysis showing the antifungal activity of a β-1,3-1,4-glucanase, in particular against M. perniciosa.






Similar content being viewed by others
Abbreviations
- ORF:
-
Open reading frame
- PB:
-
Phosphate buffer
- PI:
-
Propidium iodide
References
Minic Z (2008) Physiological roles of plant glycoside hydrolases. Planta 227:723–740
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The Carbohydrate-Active EnZymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37:D233–D238
Hrmova M, Fincher GB (2001) Structure-function relationships of β-d-glucan endo- and exohydrolases from higher plants. Plant Mol Biol 47:73–91
P Antoni (2000) Bacterial 1,3-1,4-β-glucanases: structure, function and protein engineering. Biochim Biophys Acta 1543:361–382
Nakatani Y, Lamont IL, Cutfield JF (2010) Discovery and characterization of a distinctive Eex-1,3/1,4-β-glucanase from the marine bacterium Pseudoalteromonas sp. strain BB1. Appl Environ Microbiol 76:6760–6768
Niu D, Zhou X-x, Yuan T-y, Lin Z-w, Ruan H, Li W-f (2010) Effect of the C-terminal domains and terminal residues of catalytic domain on enzymatic activity and thermostability of lichenase from Clostridium thermocellum. Biotechnol Lett 32:963–967
Huang H, Yang P, Luo H, Tang H, Shao N, Yuan T, Wang Y, Bai Y, Yao B (2008) High-level expression of a truncated 1,3-1,4-β-d-glucanase from Fibrobacter succinogene in Pichia pastoris by optimization of codons and fermentation. Appl Microbiol Biotechnol 78:95–103
Takeda T, Takahashi M, Nakanishi-Masuno T, Nakano Y, Saitoh H, Hirabuchi A, Fujisawa S, Terauchi R (2010) Characterization of endo-1,3-1,4-β-glucanases in GH family 12 from Magnaporthe oryzae. Appl Microbiol Biotechnol 88:1113–1123
Yang S, Qiaojuan Y, Jiang Z, Fan G, Wang L (2008) Biochemical characterization of a novel thermostable β-1,3–1,4-glucanase (lichenase) from Paecilomyces thermophila. J Agric Food Chem 56:5345–5351
Celestino K, Cunha R, Felix C (2006) Characterization of a beta-glucanase produced by Rhizopus microsporus var. microsporus, and its potential for application in the brewing industry. BMC Biochem 7:23
Slakeski N, Fincher GB (1992) Developmental regulation of (1 → 3,1 → 4)-β-glucanase gene expression in barley : tissue-specific expression of individual isoenzymes. Plant Physiol 99:1226–1231
Takeda H, Sugahara T, Kotake T, Nakagawa N, Sakurai N (2010) Sugar treatment inhibits IAA-induced expression of endo-1,3:1,4-β-glucanase EI transcripts in barley coleoptile segments. Physiol Plant 139:413–420
Akiyama T, Jin S, Yoshida M, Hoshino T, Opassiri R, Ketudat Cairns JR (2009) Expression of an endo-(1,3;1,4)-β-glucanase in response to wounding, methyl jasmonate, abscisic acid and ethephon in rice seedlings. J Plant Physiol 166:1814–1825
Pungartnik C, da Silva AC, de Melo SA, Gramacho KP, de Mattos Cascardo JC, Brendel M, Micheli F, da Silva Gesteira A (2009) High-affinity copper transport and Snq2 export permease of Saccharomyces cerevisiae modulate cytotoxicity of PR-10 from Theobroma cacao. Mol Plant Microbe Interact 22:39–51
Maximova S, Marelli J-P, Young A, Pishak S, Verica J, Guiltinan M (2006) Over-expression of a cacao class I chitinase gene in Theobroma cacao L. enhances resistance against the pathogen Colletotrichum gloeosporioides. Planta 224:740–749
Menezes SP, dos Santos JL, Cardoso THS, Pirovani CP, Micheli F, Noronha FSM, Alves AC, Faria AMC, da Silva Gesteira A (2012) Evaluation of the allergenicity potential of TcPR-10 protein from Theobroma cacao. PLoS ONE 7:e37969
Micheli F, Guiltinan M, Gramacho KP, Wilkinson MJ, Figueira AVdO, Cascardo JCdM, Maximova S, Lanaud C (2010) Functional genomics of cacao. In: Jean-Claude K, Michel D (eds) Advances in botanical research, Chapt 3. Academic Press, London, pp 119–177
Gesteira AS, Micheli F, Carels N, Da Silva A, Gramacho K, Schuster I, Macedo J, Pereira G, Cascardo J (2007) Comparative analysis of expressed genes from cacao meristems infected by Moniliophthora perniciosa. Ann Bot 100:129–140
Argout X, Salse J, Aury J-M, Guiltinan MJ, Droc G, Gouzy J, Allegre M, Chaparro C, Legavre T, Maximova SN, Abrouk M, Murat F, Fouet O, Poulain J, Ruiz M, Roguet Y, Rodier-Goud M, Barbosa-Neto JF, Sabot F, Kudrna D, Ammiraju JSS, Schuster SC, Carlson JE, Sallet E, Schiex T, Dievart A, Kramer M, Gelley L, Shi Z, Berard A, Viot C, Boccara M, Risterucci AM, Guignon V, Sabau X, Axtell MJ, Ma Z, Zhang Y, Brown S, Bourge M, Golser W, Song X, Clement D, Rivallan R, Tahi M, Akaza JM, Pitollat B, Gramacho K, D’Hont A, Brunel D, Infante D, Kebe I, Costet P, Wing R, McCombie WR, Guiderdoni E, Quetier F, Panaud O, Wincker P, Bocs S, Lanaud C (2011) The genome of Theobroma cacao. Nat Genet 43:101–108
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Blom N, Gammeltoft S, Brunak S (1999) Sequence and structure-based prediction of eukaryotic protein phosphorylation sites. J Mol Biol 294:1351–1362
Quevillon E, Silventoinen V, Pillai S, Harte N, Mulder N, Apweiler R, Lopez R (2005) InterProScan: protein domains identifier. Nucleic Acids Res 33:W116–W120
Henikoff S, Henikoff JG (1992) Amino acid substitution matrices from protein blocks. Proc Natl Acad Sci USA 89:10915–10919
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Bordoli L, Kiefer F, Arnold K, Benkert P, Battey J, Schwede T (2008) Protein structure homology modeling using SWISS-MODEL workspace. Nat Protocols 4:1–13
Notredame C, Higgins DG, Heringa J (2000) T-coffee: a novel method for fast and accurate multiple sequence alignment. J Mol Biol 302:205–217
Tsai L-C, Chen Y-N, Shyur L-F (2008) Structural modeling of glucanase–substrate complexes suggests a conserved tyrosine is involved in carbohydrate recognition in plant 1,3-1,4-β-d-glucanases. J Comput Aided Mol Des 22:915–923
Weiner SJ, Kollman PA, Case DA, Singh UC, Ghio C, Alagona G, Profeta S, Weiner P (1984) A new force field for molecular mechanical simulation of nucleic acids and proteins. J Am Chem Soc 106:765–784
Weiner SJ, Kollman PA, Nguyen DT, Case DA (1986) An all atom force field for simulations of proteins and nucleic acids. J Comput Chem 7:230–252
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26:283–291
Melo F, Feytmans E (1997) Novel knowledge-based mean force potential at atomic level. J Mol Biol 267:207–222
Melo F, Feytmans E (1998) Assessing protein structures with a non-local atomic interaction energy. J Mol Biol 277:1141–1152
Filho DF, Pungartnik C, Cascardo JCM, Brendel M (2006) Broken hyphae of the basidiomycete Crinipellis perniciosa allow quantitative assay of toxicity. Curr Microbiol 52:407–412
Durek P, Schmidt R, Heazlewood JL, Jones A, MacLean D, Nagel A, Kersten B, Schulze WX (2010) PhosPhAt: the Arabidopsis thaliana phosphorylation site database. An update. Nucleic Acids Res 38:D828–D834
Lin J, Xie Z, Zhu H, Qian J (2010) Understanding protein phosphorylation on a systems level. Brief Funct Genomics 9:32–42
Park C-J, Kim K-J, Shin R, Park JM, Shin Y-C, Paek K-H (2004) Pathogenesis-related protein 10 isolated from hot pepper functions as a ribonuclease in an antiviral pathway. Plant J 37:186–198
Song W, Henquet MGL, Mentink RA, van Dijk AJ, Cordewener JHG, Bosch D, America AHP, van der Krol AR (2011) N-glycoproteomics in plants: perspectives and challenges. J Proteomics 74:1463–1474
Wolf N (1991) Complete nucleotide sequence of a Hordeum vulgare gene encoding (1 → 3, 1 → 4)-β-glucanase isoenzyme II. Plant Physiol 96:1382–1384
Bailey B, Bae H, Strem M, Antunezdemayolo G, Guiltinan M, Verica J, Maximova S, Bowers J (2005) Developmental expression of stress response genes in leaves and their response to Nep1 treatment and a compatible infection by Phytophthora megakarya. Plant Physiol Biochem 43:611–622
Ji C, Kuc J (1996) Antifungal activity of cucumber beta-1,3-glucanase and chitinase. Physiol Mol Plant Pathol 49:257–265
Sela-Buurlage MB, Ponstein AS, Bres-Vloemans SA, Melchers LS, van den Elzen PJM, Cornelissen BJC (1993) Only specific tobacco (Nicotiana tabacum) chitinases and [beta]-1,3-glucanases exhibit antifungal activity. Plant Physiol 101:857–863
Jach G, Görnhardt B, Mundy J, Logemann J, Pinsdorf E, Leah R, Schell J, Maas C (1995) Enhanced quantitative resistance against fungal disease by combinatorial expression of different barley antifungal proteins in transgenic tobacco. Plant J 8:97–109
Schaefer S, Gasic K, Cammue B, Broekaert W, van Damme E, Peumans W, Korban S (2005) Enhanced resistance to early blight in transgenic tomato lines expressing heterologous plant defense genes. Planta 222:858–866
Acknowledgments
This research was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil) and the International Foundation for Science (IFS). The work of DSB and TPS was supported by the Fundação de Amparo à Pesquisa da Bahia (FAPESB, Brazil). We thank Dr. Alan Pomella (Grupo Farroupilha, Brazil) for kindly providing the M. perniciosa strain and Dr. Claudia Fortes Ferreira (Embrapa, Cruz das Almas-BA, Brazil) for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated in memory of Júlio Cezar M. Cascardo.
Rights and permissions
About this article
Cite this article
Britto, D.S., Pirovani, C.P., Andrade, B.S. et al. Recombinant β-1,3-1,4-glucanase from Theobroma cacao impairs Moniliophthora perniciosa mycelial growth. Mol Biol Rep 40, 5417–5427 (2013). https://doi.org/10.1007/s11033-013-2640-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2640-1