Abstract
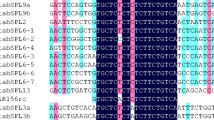
Three coding region cDNAs of duplicated PISTILLATA-like (PI-like) MADS-box genes, BnPI-1, BnPI-2 and BnPI-3, were isolated from B. napus by RT-PCR. The sequence analysis showed that the three PI cDNAs possessed 627, 627 and 625 nucleotides, respectively, and their nucleotide sequences had 96.49–98.72% similarity. Due to a deletion of two nucleotides, the protein sequence in the downstream of the frameshift site was altered in BnPI-3. Therefore, there were only 171 amino acids coded by BnPI-3, while there were 208 ones coded by BnPI-1 or BnPI-2. The deduced amino acid identity between BnPI-1 and BnPI-2 was 97.6% and the amino acid sequence of BnPI-1 and BnPI-2 shared 72.6% identity with BnPI-3. The deduced amino acid sequences of the coded proteins indicated high homology with the members of the PI family of MADS-box proteins. RT-PCR analysis showed that BnPI transcription was only detectable in petals and stamens. The yeast two-hybrid assays results showed that the three BnPI proteins exhibited different dimerization affinities with three BnAP3. BnPI-1 and BnPI-2 could form strong heterodimers with BnAP3. The dimerization affinity of BnPI-1 with BnAP3-4 is the strongest in all the combinations, while the affinity of BnPI-3 with BnAP3-4 is the weakest. The dimerization affinity to BnAP3-4 of BnPI-1 is 3.5 times of that of BnPI-3. The distinguished weak interaction to AP3 of BnPI-3 is probably due to the loss of the PI motif. The divergences of sequence and affinity of protein interaction might reflect some functional divergence of the three PI genes in B. napus.




Similar content being viewed by others
References
Bowman JL, Smyth DR, Meyerowitz EM (1991) Genetic interactions among floral homeotic genes of Arabidopsis. Development 112:1–20
Coen ES, Meyerowitz EM (1991) The war of the whorls: genetic interactions controlling flower development. Nature 353:31–37
Davies B, Schwarz-Sommer Z (1994) Control of floral organ identity by homeotic MADS-box transcription factors. Results Probl Cell Differ 20:235–258
Weigel D, Meyerowitz EM (1994) The ABCs of floral homeotic genes. Cell 78:203–209
Angenent GC, Colombo L (1996) Molecular control of ovule development. Trends Plant Sci 1:228–232
Theissen G (2001) Development of floral organ identity: stories from the MADS house. Curr Opin Plant Biol 4:75–85
Theissen G, Becker A, Di Rosa A, Kanno A, Kim JT, Münster T, Winter KU, Saedler H (2000) A short history of MADS-box genes in plants. Plant Mol Biol 42:115–149
Theissen G, Kim JT, Saedler H (1996) Classification and phylogeny of the MADS-box multigene family suggest defined roles of the MADS-box gene subfamilies in the morphological evolution of eukaryotes. J Mol Evol 43:484–516
Yang Y, Fanning L, Jack T (2003) The K domain mediates heterodimerization of the Arabidopsis floral organ identity proteins, APETALA3 and PISTILLATA. Plant J 33:47–59
Bowman JL, Smyth DR, Meyerowitz EM (1989) Genes directing flower development in Arabidopsis. Plant Cell 1:37–52
Krizek BA, Meyerowitz EM (1996) The Arabidopsis homeotic genes APETALA3 and PISTILLATA are sufficient to provide the B class organ identity function. Development 122:11–22
Samach A, Kohalmi SE, Motte P, Datla R, Haughn GW (1997) Divergence of function and regulation of class B floral organ identity genes. Plant Cell 9:559–570
Jack T, Brockman LL, Meyerowitz EM (1992) The homeotic gene APETALA3 of Arabidopsis thaliana encodes a MADS box and is expressed in petals and stamens. Cell 68:683–697
Jack T, Fox GL, Meyerowitz EM (1994) Arabidopsis homeotic gene APETALA3 ectopic expression: transcriptional and posttranscriptional regulation determine floral organ identity. Cell 76:703–716
Goto K, Meyerowitz EM (1994) Function and regulation of the Arabidopsis floral homeotic gene PISTILLATA. Genes Dev 8:1548–1560
Honma T, Goto K (2000) The Arabidopsis floral homeotic gene PISTILLATA is regulated by discrete cis-elements responsive to induction and maintenance signals. Development 127:2021–2030
Lynch M, Conery JS (2000) The evolutionary fate and consequences of duplicate genes. Science 290:1151–1155
Blanc G, Wolfe KH (2004) Widespread paleopolyploidy in model plant species inferred from age distributions of duplicate genes. Plant Cell 16:1667–1678
Cui L, Wall PK, Leebens-Mack JH et al (2006) Widespread genome duplications throughout the history of flowering plants. Genome Res 16:738–749
Bodt S, Maere S, Peer YV (2005) Genome duplication and the origin of angiosperms. Trends Ecol Evol 20:591–597
Duarte JM, Cui L, Wall PK et al (2005) Expression pattern shifts following duplication indicative of subfunctionalization and neofunctionalization in regulatory genes of Arabidopsis. Mol Biol Evol 23:469–478
Irish VF, Litt A (2005) Flower development and evolution: gene duplication, diversification and redeployment. Curr Opin Genet Dev 15:454–460
Wagner A (1998) The fate of duplicated genes: loss or new function? Bioessays 20:785–788
Nagahara U (1935) Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilisation. J Japan Bot 7:389–452
Palmer JD, Shields CR, Cohen DB, Orton TJ (1983) Chloroplast DNA evolution and the origin of amphidiploid Brassica species. Theor Appl Genet 65:181–189
Parkin IAP, Sharpe AG, Keith DJ, Lydiate DJ (1995) Identification of the A and C genomes of the amphidiploid Brassica napus (oilseed rape). Genome 38:1122–1131
Schmidt R, Acarkan A, Boivin K (2001) Comparative structural genomics in the Brassicaceae family. Plant Physiol Biochem 39:253–262
Pylatuik JD, Lindsay DL, Davis AR, Bonham-Smith PC (2003) Isolation and characterization of a Brassica napus cDNA corresponding to a B-class floral development gene. J Exp Bot 54:2385–2387
Zhou YT, Wang HY, Zhou L, Wang MP, Li HP, Wang ML, Zhao Y (2007) Analyses of the floral organ morphogenesis and the differentially expressed genes of an apetalous flower mutant in Brassica napus. Plant Cell Rep 27:9–20
Zhao Y, Wang ML (2004) Inheritance and agronomic performance of an apetalous flower mutant in Brassica napus L. Euphytica 137:381–386
Kramer EM, Dorit RL, Irish VF (1998) Molecular evolution of genes controlling petal and stamen development: duplication and divergence within the APETALA3 and PISTILLATA MADS-box gene lineages. Genetics 149:765–783
Lamb RS, Irish VF (2003) Functional divergence within the APETALA3/PISTILLATA floral homeotic gene lineages. Proc Natl Acad Sci USA 100:6558–6563
Vandenbussche M, Zethof J, Royaert S, Weterings K, Gerats T (2004) The duplicated B-class heterodimer model: Whorl specific effects and complex genetic interactions in Petunia hybrida flower development. Plant Cell 16:741–754
Yadav SR, Prasad K, Vijayraghavan U (2007) Divergent regulatory OsMADS2 functions control size, shape and differentiation of the highly derived rice floret second whorl organ. Genetics 176:283–294
Kramer EM, Di Stilio VS, Schlu¨ter PM (2003) Complex patterns of gene duplication in the APETALA3 and PISTILLATA lineages of the Ranunculaceae. Int J Plant Sci 164:1–11
Litt A, Irish VF (2003) Duplication and diversification in the APETALA1/FRUITFULL floral homeotic gene lineage: implications for the evolution of floral development. Genetics 165:821–833
Vandenbussche M, Theissen G, Van der Peer Y, Gerats T (2003) Structural diversification and neo-functionalization during floral MADS-box gene evolution by C-terminal frameshift mutations. Nucleic Acids Res 31:4401–4409
Acknowledgement
This work was supported by the National Natural Science Foundation of China (Grant Nos. 30571174 and 30871540).
Author information
Authors and Affiliations
Corresponding author
Additional information
The sequences reported in this paper have been deposited in GenBank with the accession numbers, EU159431, EU159432 and EU159433, respectively.
Wei Deng and Lin Zhou are committed as the first author.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Deng, W., Zhou, L., Zhou, Y. et al. Isolation and characterization of three duplicated PISTILLATA genes in Brassica napus . Mol Biol Rep 38, 3113–3120 (2011). https://doi.org/10.1007/s11033-010-9981-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-9981-9