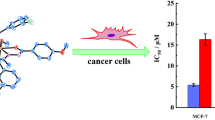
Abstract
Antiproliferative activity of twenty one Michael adducts of aroylacrylic acids and cyclic amines (\(N\)-Me-piperazine, imidazole, 2-Me-imidazole, and indole) was tested toward five human tumor cell lines (HeLa, LS174, K562, FemX, MDA-MB-361) in vitro. Compounds exerted antiproliferative activity in the high to the single-digit micromolar concentrations, causing increase of the cell population fraction in S phase and apoptosis. \(N\)-Me-piperazine and imidazole derivatives of aroylacrylic acids substituted with bulky alkyl substituents (2,4-di-\(i\)-Pr-Ph-, 2,4,6-tri-Et-Ph-, or \(\beta \)-tetrahydronaphthyl-) showed the best potency, while indole adducts were proved as the inferior antiproliferative agents. Few compounds showed significant selectivity, tumor versus healthy cells, with selectivity index \({\sim }60\) for the most selective congener. An unbiased in silico distinction between more and less potent compounds was obtained from 3D QSAR models derived by alignment-independent GRIND-2 descriptors.
Graphical Abstract




Similar content being viewed by others
References
Bougault J (1908) Sur l‘acide benzoylacrylique. Ann Chim Phys 15:491–515
Cromwell NH, Creger PL, Cook KE (1956) Studies with the amine adducts of para-benzoylacrylic acid and its methyl ester. J Am Chem Soc 78:4412–4415. doi:10.1021/ja01598a058
Li X, Liu N, Zhang H, Knudson SE, Li H-J, Lai C-T, Simmerling C, Slayden RA, Tonge PJ (2011) CoA adducts of 4-oxo-4-phenylbut-2-enoates: inhibitors of MenB from the M. tuberculosis menaquinone biosynthesis pathway. ACS Med Chem Lett 2:818–823. doi:10.1021/ml200141e
Hayashi K, Neve J, Hirose M, Kuboki A, Shimada Y, Kepinski S, Nozaki H (2012) Rational design of an auxin antagonist of the SCF\(^{TIR1}\) auxin receptor complex. ACS Chem Biol 7:590–598. doi: 10.1021/cb200404c
PubChem’s BioAssay, AID 376: HTS Discovery of chemical inhibitors of HePTP, a leukemia target. Burnham Center for Chemical Genomics
Wang Y, Xiao J, Suzek TO, Zhang J, Wang J, Zhou Z, Han L, Karapetyan K, Dracheva S, Shoemaker BA, Bolton E, Gindulyte A, Bryant SH (2012) PubChem’s BioAssay database. Nucleic Acids Res 40:D400–D412. doi:10.1093/nar/gkr1132
Couquelet J, Couquelet J, Cluzel R, Tronche P (1969) Antibacterial \(\alpha \)-amino-\(\beta \)-aroylpropionic acids. Chim Ther 4:127–130
Gieldanowski J, Steuden W (1981) Screening system for the evaluation of preparations with potential cytostatic and immunotropic activity. II. Derivatives of propionic and hydroxybutyric acids and of tetrahydrofuran-2-one. Arch Immunol Ther Exp 29:257–265
Drakulić BJ, Stanojković TP, Žižak ŽS, Dabović MM (2011) Antiproliferative activity of aroylacrylic acids. Structure–activity study based on molecular interaction fields. Eur J Med Chem 46:3265–3273. doi:10.1016/j.ejmech.2011.04.043
Drakulić BJ, Juranić ZD, Stanojković TP, Juranić IO (2005) 2-[(Carboxymethyl) sulfanyl]-4-oxo-4-arylbutanoic acids selectively suppressed proliferation of neoplastic human HeLa cells. A SAR/QSAR study. J Med Chem 48:5600–5603. doi:10.1021/jm0502889
Vitorović-Todorović MD, Erić-Nikolić A, Kolundžija B, Hamel E, Ristić S, Juranić IO, Drakulić BJ (2013) (\(E)\)-4-Aryl-4-oxo-2-butenoic acid amides, chalcone-aroylacrylic acid chimeras: design, antiproliferative activity and inhibition of tubulin polymerization. Eur J Med Chem 62:40–50. doi: 10.1016/j.ejmech.2013.01.006
Papa D, Schwenk E, Villani F, Klingsberg E (1948) \(\beta \)-Aroylacrylic acids. J Am Chem Soc 70:3356–3360. doi: 10.1021/ja01190a041
Abubshait SA (2007) An efficient synthesis and reactions of novel indolylpyridazinone derivatives with expected biological activity. Molecules 12:25–42. doi:10.3390/12010025
Tamam GH, Soliman EA, El-Gendy AM, Al-Kady AY (1987) Alkylation of indole with \(\beta \)-aroylacrylic acids and some studies on the alkylated products. J Chem Soc Pak 9:1–10
Goodford PJ (1985) A computational procedure for determining energetically favorable binding sites on biologically important macromolecules. J Med Chem 28:849–857. doi:10.1021/jm00145a002
GRID 22c (2004) Molecular Discovery Ltd. http://www.moldiscovery.com. Accessed 20 May 2014
Pentacle 1.0.6 (2010) Molecular Discovery Ltd. http://www.moldiscovery.com/. Accessed 20 May 2014
Durán A, Comesaña G, Pastor M (2008) Development and validation of AMANDA, a new algorithm for selecting highly relevant regions in molecular interaction fields. J Chem Inf Model 48:1813–1823. doi:10.1021/ci800037t
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 6:55–63
Ohno M, Abe TJ (1991) Rapid colorimetric assay for the quantification of leukemia inhibitory factor (LIF) and interleukin-6 (IL-6). J Immunol Methods 145:199–203
Clothier RH (1995) The FRAME cytotoxicity test. Methods Mol Biol 43:109–118
Hawkins PCD, Skillman AG, Warren GL, Ellingson BA, Stahl MT (2010) Conformer generation with OMEGA: algorithm and validation using high quality structures from the Protein Databank and Cambridge Structural Database. J Chem Inf Model 50:572–584. doi:10.1021/ci100031x
Hawkins PCD, Nicholls A (2012) Conformer generation with OMEGA: learning from the data set and the analysis of failures. J Chem Inf Model 52:2919–2936. doi:10.1021/ci300314k
OMEGA 2.5.1.4 (2013) OpenEye Scientific Software, Santa Fe, NM. http://www.eyesopen.com. Accessed 20 May 2014
Halgren TA (1999) MMFF VII. Characterization of MMFF94, MMFF94s and other widely available force fields for conformational energies and for intermolecular interaction energies and geometries. J Comput Chem 20:730–748. doi:10.1002/(SICI)1096-987X
Gaillard P, Carrupt PA, Testa B, Boudon A (1994) Molecular lipophilicity potential, a tool in 3D QSAR: method and applications. J Comput Aided Mol Des 8:83–96
Pedretti A, Villa L, Vistoli G (2004) Vega—an open platform to develop chemo-bio-informatics applications, using plug-in architecture and script programming. J Comput Aided Mol Des 18:167–173
Acknowledgments
This work is supported by the Ministry of Education, Science and Technological Development of Serbia, under Grant 172035. Authors gratefully acknowledge OpenEye Scientific Software, Santa Fe, NM, for the free academic licensing of software tools.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Juranić, I.O., Tošić, A.V., Kolundžija, B. et al. Antiproliferative activity of the Michael adducts of aroylacrylic acids and cyclic amines. Mol Divers 18, 577–592 (2014). https://doi.org/10.1007/s11030-014-9528-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-014-9528-4