Abstract
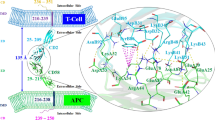
Macrophage migration inhibitory factor (MIF) is a key cytokine in autoimmune and inflammatory diseases that attracts and then retains activated immune cells from the periphery to the tissues. MIF exists as a homotrimer and its effects are mediated through its primary receptor, CD74 (the class II invariant chain that exhibits a highly structured trimerization domain), present on class II expressing cells. Although a number of binding residues have been identified between MIF and CD74 trimers, their spatial orientation has not been established. Using a docking program in silico, we have modeled binding interactions between CD74 and MIF as well as CD74 and a competitive MIF inhibitor, RTL1000, a partial MHC class II construct that is currently in clinical trials for multiple sclerosis. These analyses revealed 3 binding sites on the MIF trimer that each were predicted to bind one CD74 trimer through interactions with two distinct 5 amino acid determinants. Surprisingly, predicted binding of one CD74 trimer to a single RTL1000 antagonist utilized the same two 5 residue determinants, providing strong suggestive evidence in support of the MIF binding regions on CD74. Taken together, our structural modeling predicts a new MIF(CD74)3 dodecamer that may provide the basis for increased MIF potency and the requirement for ~3-fold excess RTL1000 to achieve full antagonism.



Similar content being viewed by others
Abbreviations
- MIF:
-
macrophage migration inhibitory factor
- RTL1000:
-
recombinant T cell receptor ligand
References
Benedek G et al. (2013) Partial MHC class II constructs inhibit MIF/CD74 binding and downstream effects. Eur J Immunol 43:1309–1321. doi:10.1002/eji.201243162
Cox GM et al. (2013) Macrophage migration inhibitory factor potentiates autoimmune-mediated neuroinflammation. J Immunol 191:1043–1054. doi:10.4049/jimmunol.1200485
El-Turk F et al. (2008) The conformational flexibility of the carboxy terminal residues 105-114 is a key modulator of the catalytic activity and stability of macrophage migration inhibitory factor. Biochemistry 47:10740–10756. doi:10.1021/bi800603x
El-Turk F et al. (2012) Characterization of molecular determinants of the conformational stability of macrophage migration inhibitory factor: leucine 46 hydrophobic pocket. PLoS One 7:e45024. doi:10.1371/journal.pone.0045024
Grieb G, Kim BS, Simons D, Bernhagen J, Pallua N (2014) MIF and CD74 - suitability as clinical biomarkers. Mini Rev Med Chem 14:1125–1131
Jasanoff A, Wagner G, Wiley DC (1998) Structure of a trimeric domain of the MHC Class II-associated chaperonin and targeting protein Ii. EMBO J 17:6812–6818 doi:10.1093/emboj/17.23.6812
Landsverk OJ, Bakke O, Gregers TF (2009) MHC II and the Endocytic pathway: regulation by invariant chain. Scand J Immunol 70:184–193 doi:10.1111/j.1365-3083.2009.02301.x
Leng L et al. (2003) MIF signal transduction initiated by binding to CD74. J Exp Med 197:1467–1476. doi:10.1084/jem.20030286
Meza-Romero R et al. (2014) HLA-DRalpha1 constructs block CD74 expression and MIF effects in experimental autoimmune encephalomyelitis. J Immunol 192:4164–4173. doi:10.4049/jimmunol.1303118
Morandi E, Tarlinton RE, Gran B (2015) Multiple Sclerosis between Genetics and Infections: human endogenous retroviruses in monocytes and macrophages. Front Immunol 6:647 doi:10.3389/fimmu.2015.00647
Pantouris G et al. (2015) An analysis of MIF structural features that control functional activation of CD74. Chem Biol 22:1197–1205. doi:10.1016/j.chembiol.2015.08.006
Pierce BG, Hourai Y, Weng Z (2011) Accelerating protein docking in ZDOCK using an advanced 3D convolution library. PLoS One 6:e24657. doi:10.1371/journal.pone.0024657
Pierce BG, Wiehe K, Hwang H, Kim BH, Vreven T, Weng Z (2014) ZDOCK Server: interactive docking prediction of protein-protein complexes and symmetric multimers bioinformatics. 30:1771–1773 doi:10.1093/bioinformatics/btu097
Powell ND, Papenfuss TL, McClain MA, Gienapp IE, Shawler TM, Satoskar AR, Whitacre CC, (2005) Cutting Edge: macrophage migration inhibitory factor is necessary for progression of experimental autoimmune encephalomyelitis. J Immunol 175:5611–5614
Shi X et al. (2006) CD44 is the signaling component of the macrophage migration inhibitory factor-CD74 receptor complex. Immunity 25:595–606. doi:10.1016/j.immuni.2006.08.020
Starlets D et al. (2006) Cell-surface CD74 initiates a signaling cascade leading to cell proliferation and survival. Blood 107:4807–4816. doi:10.1182/blood-2005-11-4334
Steinman L (2014) Immunology of relapse and remission in multiple sclerosis. Annu Rev Immunol 32:257–281. doi:10.1146/annurev-immunol-032713-120227
Sun HW, Bernhagen J, Bucala R, Lolis E (1996) Crystal structure at 2.6-a resolution of human macrophage migration inhibitory factor. Proc Natl Acad Sci U S A 93:5191–5196
Suzuki M, Sugimoto H, Nakagawa A, Tanaka I, Nishihira J, Sakai M (1996) Crystal structure of the macrophage migration inhibitory factor from rat liver. Nat Struct Biol 3:259–266
Vandenbark AA et al. (2013) A novel regulatory pathway for autoimmune disease: binding of partial MHC class II constructs to monocytes reduces CD74 expression and induces both specific and bystander T-cell tolerance. J Autoimmun 40:96–110. doi:10.1016/j.jaut.2012.08.004
Acknowledgments
This work was supported by the National Institutes of Health grant NS047661 and National Multiple Sclerosis Society grant RG-5068-A-6 (AAV); National Institutes of Health grants AR049610 and AI042310 (LL, RB); National Multiple Sclerosis Society grant RG5272A1/T (GB); and the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development (AAV). The contents do not represent the views of the Department of Veterans Affairs or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Drs. Vandenbark, Benedek, Meza-Romero and OHSU have a significant financial interest in Artielle ImmunoTherapeutics, Inc., a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by the OHSU and VAPHCS Conflict of Interest in Research Committees. Drs. Leng and Bucala have no conflict of interest.
Rights and permissions
About this article
Cite this article
Meza-Romero, R., Benedek, G., Leng, L. et al. Predicted structure of MIF/CD74 and RTL1000/CD74 complexes. Metab Brain Dis 31, 249–255 (2016). https://doi.org/10.1007/s11011-016-9798-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9798-x