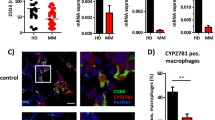
Abstract
Daratumumab (DAR) is novel human anti-CD38 IgG1, high-affinity human monoclonal antibody having broad-spectrum killing activity. The antibody is recommended to treat multiple myeloma. Recently Antibody-dependent cellular phagocytosis (ADCP) have been identified as the potential mechanism of DAR in addition to complement-dependent cytotoxicity (CDC) and antibody-dependent cell-mediated cytotoxicity (ADCC). In the present study we evaluated the effect of Daratumumab on other effector cells of multiple myeloma. Luciferase+ MM.1R GFP cells were selected for the study. For immune-compromised multiple myeloma tumour xenograft mouse model we used severe combined immunodeficient beige (SCID-beige), NOD SCID gamma (NSG) and C57Bl/6j mice. Bioluminescence imaging was carried by injecting luciferin, and in vivo confocal microscopy was done for tracing bone marrow niches. Spleen and tumours were submitted to immunophenotypic analysis. MTT assay was done for cell proliferation studies. We established tumour xenograft mouse model. It was found that DAR showed significant anti-tumour effect in tumour xenograft multiple myeloma mice. We found that DAR showed anti-tumour activity via Fc–FcγR interaction with macrophages. DAR induced phenotypic activation of macrophages in mice and resulted in ADCP of cancerous cells via interacting Fc-FcγR in vitro. The study suggested that DAR exerted anti-tumour activity in multiple myeloma by interacting with Fc-FcγR.





Similar content being viewed by others
Data availability
The data will be provided on contacting corresponding author.
References
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics. CA Cancer J Clin 62(1):10–29. https://doi.org/10.3322/caac.20138
Rajkumar SV (2011) Treatment of multiple myeloma. Nat Rev Clin Oncol. 8(8):479–91. https://doi.org/10.1038/nrclinonc.2011.63
Yang Y (2015) Cancer immunotherapy: harnessing the immune system to battle cancer. J Clin Invest 125(9):3335–3337. https://doi.org/10.1172/JCI83871
Weiner LM, Dhodapkar MV, Ferrone S (2009) Monoclonal antibodies for cancer immunotherapy. Lancet 373:1033–1040. https://doi.org/10.1007/s11033-018-4427-x
Scott AM, Wolchok JD, Old LJ (2012) Antibody therapy of cancer. Nat Rev Cancer 12:278–287. https://doi.org/10.1038/nrc3236
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DC, Oomen LA, Peipp M, Valerius T, Slootstra JW, Mutis T, Bleeker WK, Anderson KC, Lokhorst HM, van de Winkel JG, Parren PW (2011) Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol 186(3):1840–1848. https://doi.org/10.4049/jimmunol.1003032
Nimmerjahn F, Ravetch JV (2008) Fc gamma receptors as regulators of immune responses. Nat Rev Immunol 8:34–47. https://doi.org/10.1038/nri2206
Veillette A, Guo H (2013) CS1, a SLAM family receptor involved in immune regulation, is a therapeutic target in multiple myeloma. Crit Rev Oncol Hematol 88:168–177. https://doi.org/10.1016/j.critrevonc.2013.04.003
Asimakopoulos F, Kim J, Denu RA, Hope C, Jensen JL, Ollar SJ, Hebron E, Flanagan C, Callander N, Hematti P (2013) Macrophages in multiple myeloma: emerging concepts and therapeutic implications. Leuk Lymphoma 54(10):2112–2121. https://doi.org/10.3109/10428194.2013.778409
Weiskopf K, Weissman IL (2015) Macrophages are critical effectors of antibody therapies for cancer. MAbs 7:303–310. https://doi.org/10.1080/19420862.2015.1011450
Gül N, van Egmond M (2015) Antibody-dependent phagocytosis of tumor cells by macrophages: a potent effector mechanism of monoclonal antibody therapy of cancer. Can Res 75(23):5008–5013. https://doi.org/10.1158/0008-5472.CAN-15-1330
Shi Y, Fan X, Deng H, Brezski RJ, Rycyzyn M, Jordan RE, Strohl WR, Zou Q, Zhang N, An Z (2015) Trastuzumab triggers phagocytic killing of high HER2 cancer cells in vitro and in vivo by interaction with Fcγ receptors on macrophages. J Immunol 194(9):4379–4386. https://doi.org/10.4049/jimmunol.1402891
Chao MP, Alizadeh AA, Tang C, Myklebust JH, Varghese B, Gill S, Jan M, Cha AC, Chan CK, Tan BT, Park CY, Zhao F, Kohrt HE, Malumbres R, Briones J, Gascoyne RD, Lossos IS, Levy R, Weissman IL, Majeti R (2010) Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell 142(5):699–713. https://doi.org/10.1016/j.cell.2010.07.044
Overdijk MB, Verploegen S, Bögels M, van Egmond M, Lammerts van Bueren JJ, Mutis T, Groen RW, Breij E, Martens AC, Bleeker WK, Parren PW (2015) Antibody-mediated phagocytosis contributes to the anti-tumor activity of the therapeutic antibody daratumumab in lymphoma and multiple myeloma. MAbs 7(2):311–321. https://doi.org/10.1080/19420862.2015.1007813
Bezman NA, Jhatakia A, Kearney AY, Brender T, Maurer M, Henning K, Jenkins MR, Rogers AJ, Neeson PJ, Korman AJ, Robbins MD, Graziano RF (2017) PD-1 blockade enhances elotuzumab efficacy in mouse tumor models. Blood Adv 1(12):753–765. https://doi.org/10.1182/bloodadvances.2017004382
Tomayko MM, Reynolds CP (1989) Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother Pharmacol 24:148–154. https://doi.org/10.1007/BF00300234
Cassetta L, Noy R, Swierczak A, Sugano G, Smith H, Wiechmann L, Pollard JW (2016) Isolation of mouse and human tumor-associated macrophages. Adv Exp Med Biol 899:211–229. https://doi.org/10.1007/978-3-319-26666-4_12
Rossi M, Botta C, Arbitrio M, Grembiale RD, Tagliaferri P, Tassone P (2018) Mouse models of multiple myeloma: technologic platforms and perspectives. Oncotarget 9(28):20119–20133. https://doi.org/10.18632/oncotarget.24614
Matas-Céspedes A, Vidal-Crespo A, Rodriguez V, Villamor N, Delgado J, Giné E, Roca-Ho H, Menéndez P, Campo E, López-Guillermo A, Colomer D, Roué G, Wiestner A, Parren PW, Doshi P, van Bueren JL, Pérez-Galán P (2017) The human CD38 monoclonal antibody daratumumab shows antitumor activity and hampers leukemia-microenvironment interactions in chronic lymphocytic leukemia. Clin Cancer Res 23(6):1493–1505. https://doi.org/10.1158/1078-0432.CCR-15-2095
Wang C, Yu X, Cao Q, Wang Y, Zheng G, Tan TK, Zhao H, Zhao Y, Wang Y, Harris DCh (2013) Characterization of murine macrophages from bone marrow, spleen and peritoneum. BMC Immunol 14:6. https://doi.org/10.1186/1471-2172-14-6
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141:39–51. https://doi.org/10.1016/j.cell.2010.03.014
Suyanı E, Sucak GT, Akyürek N, Sahin S, Baysal NA, Yağcı M, Haznedar R (2013) Tumor-associated macrophages as a prognostic parameter in multiple myeloma. Ann Hematol 92(5):669–677. https://doi.org/10.1007/s00277-012-1652-6
Andersen MN, Abildgaard N, Maniecki MB, Møller HJ, Andersen NF (2014) Monocyte/macrophage-derived soluble CD163: a novel biomarker in multiple myeloma. Eur J Haematol 93:41–47. https://doi.org/10.1111/ejh.12296
Noy R, Pollard JW (2014) Tumor-associated macrophages: from mechanisms to therapy. Immunity 41:49–61. https://doi.org/10.1016/j.immuni.2014.06.010
Gutiérrez-González A, Martínez-Moreno M, Samaniego R, Arellano-Sánchez N, Salinas-Muñoz L, Relloso M, Valeri A, Martínez-López J, Corbí ÁL, Hidalgo A, García-Pardo Á, Teixidó J, Sánchez-Mateos P (2016) Evaluation of the potential therapeutic benefits of macrophage reprogramming in multiple myeloma. Blood 128(18):2241–2252. https://doi.org/10.1182/blood-2016-01-695395
Georgoudaki AM, Prokopec KE, Boura VF, Hellqvist E, Sohn S, Östling J, Dahan R, Harris RA, Rantalainen M, Klevebring D, Sund M, Brage SE, Fuxe J, Rolny C, Li F, Ravetch JV, Karlsson MC (2016) Reprogramming tumor-associated macrophages by antibody targeting inhibits cancer progression and metastasis. Cell Rep 15(9):2000–2011. https://doi.org/10.1016/j.celrep.2016.04.084
Jensen JL, Rakhmilevich A, Heninger E, Broman AT, Hope C, Phan F, Miyamoto S, Maroulakou I, Callander N, Hematti P, Chesi M, Bergsagel PL, Sondel P, Asimakopoulos F (2015) Tumoricidal Effects of Macrophage-Activating Immunotherapy in a Murine Model of Relapsed/Refractory Multiple Myeloma. Cancer Immunol Res 8:881–890. https://doi.org/10.1158/2326-6066.CIR-15-0025-T
Barnhart BC, Quigley M (2017) Role of Fc–FcgR interactions in the antitumor activity of therapeutic antibodies. Immunol Cell Biol 95:340–346. https://doi.org/10.1038/icb.2016.121
Roussou M, Tasidou A, Dimopoulos MA, Kastritis E, Migkou M, Christoulas D, Gavriatopoulou M, Zagouri F, Matsouka C, Anagnostou D, Terpos E (2009) Increased expression of macrophage inflammatory protein-1alpha on trephine biopsies correlates with extensive bone disease, increased angiogenesis and advanced stage in newly diagnosed patients with multiple myeloma. Leukemia 23(11):2177–2181. https://doi.org/10.1038/leu.2009.130
Zhu EF, Gai SA, Opel CF, Kwan BH, Surana R, Mihm MC, Kauke MJ, Moynihan KD, Angelini A, Williams RT, Stephan MT, Kim JS, Yaffe MB, Irvine DJ, Weiner LM, Dranoff G, Wittrup KD (2015) Synergistic innate and adaptive immune response to combination immunotherapy with anti-tumor antigen antibodies and extended serum half-life IL-2. Cancer Cell 27(4):489–501. https://doi.org/10.1016/j.ccell.2015.03.004
Viola D, Dona A, Caserta E, Troadec E, Besi F, McDonald T, Ghoda L, Gunes EG, Sanchez JF, Khalife J, Martella M, Karanes C, Htut M, Wang X, Rosenzweig M, Chowdhury A, Sborov D, Miles RR, Yazaki PJ, Ebner T, Hofmeister CC, Forman SJ, Rosen ST, Marcucci G, Shively J, Keats JJ, Krishnan A, Pichiorri F (2021) Daratumumab induces mechanisms of immune activation through CD38+ NK cell targeting. Leukemia 35(1):189–200. https://doi.org/10.1038/s41375-020-0810-4
Chen L, Han X (2015) Anti-PD-1/PD-L1 therapy of human cancer: past, present, and future. J Clin Invest 125:3384–3391. https://doi.org/10.1172/JCI80011
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, Lennon VA, Celis E, Chen L (2002) Tumor-associated B7–H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8(8):793–800. https://doi.org/10.1038/nm730
Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, Chen S, Klein AP, Pardoll DM, Topalian SL, Chen L (2012) Colocalization of inflammatory response with B7–h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med. 4(127):127ra37. https://doi.org/10.1126/scitranslmed.3003689
Schalper KA, Carvajal-Hausdorf D, McLaughlin J, Velcheti V, Chen L, Sanmamed M, Herbst RS, Rimm DL (2015) Clinical significance of PD-L1 protein expression on tumor-associated macrophages in lung cancer. J Immunother Cancer 3(Suppl 2):P415. https://doi.org/10.1186/2051-1426-3-S2-P415
Webb JR, Milne K, Kroeger DR, Nelson BH (2016) PD-L1 expression is associated with tumor-infiltrating T cells and favorable prognosis in high-grade serous ovarian cancer. Gynecol Oncol 141:293–302. https://doi.org/10.1016/j.ygyno.2016.03.008
Kim HR, Ha SJ, Hong MH, Heo SJ, Koh YW, Choi EC, Kim EK, Pyo KH, Jung I, Seo D, Choi J, Cho BC, Yoon SO (2016) PD-L1 expression on immune cells, but not on tumor cells, is a favorable prognostic factor for head and neck cancer patients. Sci Rep 6:36956. https://doi.org/10.1038/srep36956
Acknowledgements
The authors are grateful to the staff and management of Shaanxi Provincial People’s Hospital, China for providing necessary facilities. The authors express special thanks to all the funding agencies ‘Xian Foundation for Development of Science and Technology, China (Grant No. 20YXYJ0009 (11)), Science and Technology Talents Project of Shaanxi provincial of people's hospital ,China (Grant No. 2021BJ-26), SPPH incubator fund for Development of Science and Technology (Grant No. 2021YJY-19) and International Science and Technology Cooperation Projects of Shaanxi (Grant No.2022KW-14)’ for providing the necessary financial support.
Funding
This study was supported by Xian Foundation for Development of Science and Technology, China (Grant No. 20YXYJ0009 (11)).
Author information
Authors and Affiliations
Contributions
YG and YL designed the concept and study; LL, YZ, WZ and BN along with YG and YL designed the experiments and performed them as a team. All the authors contributed equally to writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The animal protocols were approved by the animal ethical review committee of Shaanxi Provincial Peoples Hospital, China (approval number SP003V). The animal studies were in accordance to Guidelines for the ethical review of laboratory animal welfare People’s Republic of China National Standard GB/T 35892–2018 guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Y., Li, L., Zheng, Y. et al. Monoclonal antibody Daratumumab promotes macrophage-mediated anti-myeloma phagocytic activity via engaging FC gamma receptor and activation of macrophages. Mol Cell Biochem 477, 2015–2024 (2022). https://doi.org/10.1007/s11010-022-04390-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04390-8