Abstract
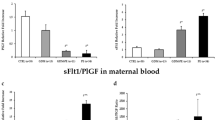
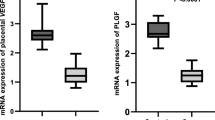
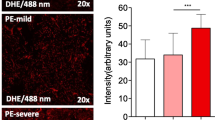
Altered placental angiogenesis is implicated in the pathophysiology of preeclampsia. We have earlier reported placental regional differences in oxidative stress markers and neurotrophins. Oxidative stress and neurotrophins are reported to regulate angiogenesis. This study aims to examine protein and mRNA levels of vascular endothelial growth factor (VEGF) and VEGF receptor 1 (VEGFR1) in four regions [central maternal (CM), central fetal (CF), peripheral maternal (PM), and peripheral fetal (PF)] of the placenta in normotensive control (NC) women (n = 51) and women with preeclampsia (PE) (n = 43) [18 delivered at term (T-PE) and 25 delivered preterm (PT-PE)]. In all groups, CF region reported highest VEGF protein levels compared to all other regions. VEGF mRNA level was higher in CF region as compared to CM region in PE group (p < 0.05). VEGF levels were lower in all regions of PE, T-PE, and PT-PE groups (p < 0.05) as compared to their respective regions in NC group. VEGFR1 levels were lower in CF (p < 0.05) and PF (p < 0.01) regions as compared to CM region only in control. However, VEGFR1 levels were higher in CF (p < 0.05) and PF (p < 0.01) regions of PT-PE group as compared to control. VEGFR1 mRNA level was higher in PM region of PE group and T-PE group (p < 0.05 for both) as compared to control. VEGF levels in the PF region were positively associated with birth weight and placental weight. This study describes placental regional changes in angiogenic factors particularly highlighting increased VEGF in CF region possibly in response to hypoxic conditions prevailing in placenta.




Similar content being viewed by others
Abbreviations
- CM:
-
Central maternal
- CF:
-
Central fetal
- PM:
-
Peripheral maternal
- PF:
-
Peripheral fetal
References
Powe CE, Levine RJ, Karumanchi SA (2011) Preeclampsia, a disease of the maternal endothelium: the role of antiangiogenic factors and implications for later cardiovascular disease. Circulation 123:2856–2869. doi:10.1161/CIRCULATIONAHA.109.853127
Musa SM, Adam I, Lutfi MF (2016) Heart rate variability and autonomic modulations in preeclampsia. PLoS ONE 11:e0152704. doi:10.1371/journal.pone.0152704
Demir R, Yaba A, Huppertz B (2010) Vasculogenesis and angiogenesis in the endometrium during menstrual cycle and implantation. Acta Histochem 112:203–214. doi:10.1016/j.acthis.2009.04.004
Demir R, Seval Y, Huppertz B (2007) Vasculogenesis and angiogenesis in the early human placenta. Acta Histochem 109:257–265. doi:10.1016/j.acthis.2007.02.008
Huppertz B, Peeters LL (2005) Vascular biology in implantation and placentation. Angiogenesis 8:157–167. doi:10.1007/s10456-005-9007-8
Kaufmann P, Mayhew TM, Charnock-Jones DS (2004) Aspects of human fetoplacental vasculogenesis and angiogenesis. II. Changes during normal pregnancy. Placenta 25:114–126. doi:10.1016/j.placenta.2003.10.009
Claesson-Welsh L (2016) VEGF receptor signal transduction—a brief update. Vasc Pharmacol 86:14–17. doi:10.1016/j.vph.2016.05.011
Roskoski R Jr (2008) VEGF receptor protein-tyrosine kinases: structure and regulation. Biochem Biophys Res Commun 375:287–291. doi:10.1016/j.bbrc.2008.07.121
Shibuya M, Claesson-Welsh L (2006) Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp Cell Res 312:549–560. doi:10.1016/j.yexcr.2005.11.012
Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L (2006) VEGF receptor signalling—in control of vascular function. Nat Rev Mol Cell Biol 7:359–371. doi:10.1038/nrm1911
Hong F, Li Y, Xu Y (2014) Decreased placental miR-126 expression and vascular endothelial growth factor levels in patients with pre-eclampsia. J Int Med Res 42:1243–1251. doi:10.1177/0300060514540627
Liu H, Li Y, Zhang J, Rao M, Liang H, Liu G (2015) The defect of both angiogenesis and lymphangiogenesis is involved in preeclampsia. Placenta 36:279–286. doi:10.1016/j.placenta.2014.12.013
Weel IC, Baergen RN, Romao-Veiga M, Borges VT, Ribeiro VR, Witkin SS, Bannwart-Castro C, Peracoli JC, De Oliveira L, Peracoli MT (2016) Association between placental lesions, cytokines and angiogenic factors in pregnant women with preeclampsia. PLoS ONE 11:e0157584. doi:10.1371/journal.pone.0157584
Kim SC, Park MJ, Joo BS, Joo JK, Suh DS, Lee KS (2012) Decreased expressions of vascular endothelial growth factor and visfatin in the placental bed of pregnancies complicated by preeclampsia. J Obstet Gynaecol Res 38:665–673. doi:10.1111/j.1447-0756.2011.01773.x
Andraweera PH, Dekker GA, Laurence JA, Roberts CT (2012) Placental expression of VEGF family mRNA in adverse pregnancy outcomes. Placenta 33:467–472. doi:10.1016/j.placenta.2012.02.013
Kweider N, Fragoulis A, Rosen C, Pecks U, Rath W, Pufe T, Wruck CJ (2011) Interplay between vascular endothelial growth factor (VEGF) and nuclear factor erythroid 2-related factor-2 (Nrf2): implications for preeclampsia. J Biol Chem 286:42863–42872. doi:10.1074/jbc.M111.286880
Lee GS, Joe YS, Kim SJ, Shin JC (2010) Cytokine-related genes and oxidation-related genes detected in preeclamptic placentas. Arch Gynecol Obstet 282:363–369. doi:10.1007/s00404-009-1222-x
Pereira RD, De Long NE, Wang RC, Yazdi FT, Holloway AC, Raha S (2015) Angiogenesis in the placenta: the role of reactive oxygen species signaling. Biomed Res Int 2015:814543. doi:10.1155/2015/814543
Burton GJ, Yung HW, Cindrova-Davies T, Charnock-Jones DS (2009) Placental endoplasmic reticulum stress and oxidative stress in the pathophysiology of unexplained intrauterine growth restriction and early onset preeclampsia. Placenta 30:43–48. doi:10.1016/j.placenta.2008.11.003
Kim Y-W, Byzova TV (2014) Oxidative stress in angiogenesis and vascular disease. Blood 123:625–631. doi:10.1182/blood-2013-09-512749
Mistry HD, Kurlak LO, Williams PJ, Ramsay MM, Symonds ME, Broughton Pipkin F (2010) Differential expression and distribution of placental glutathione peroxidases 1, 3 and 4 in normal and preeclamptic pregnancy. Placenta 31:401–408. doi:10.1016/j.placenta.2010.02.011
Dempsey EW, Luse SA (1971) Regional specializations in the syncytial trophoblast of early human placentas. J Anat 108:545–561
Matheus M, Sala MA (1989) Measurement of the villus surface area and its regional variation in the human full-term placenta. Gegenbaurs Morphol Jahrb 135:851–854
Jauniaux E, Hempstock J, Greenwold N, Burton GJ (2003) Trophoblastic oxidative stress in relation to temporal and regional differences in maternal placental blood flow in normal and abnormal early pregnancies. Am J Pathol 162:115–125. doi:10.1016/s0002-9440(10)63803-5
Jauniaux E, Jurkovic D, Campbell S (1991) In vivo investigations of the anatomy and the physiology of early human placental circulations. Ultrasound Obstet Gynecol 1:435–445. doi:10.1046/j.1469-0705.1991.01060435.x
Sahay AS, Sundrani DP, Wagh GN, Mehendale SS, Joshi SR (2015) Regional differences in the placental levels of oxidative stress markers in pre-eclampsia. Int J Gynaecol Obstet 129:213–218. doi:10.1016/j.ijgo.2015.03.001
Sahay AS, Sundrani DP, Wagh GN, Mehendale SS, Joshi SR (2015) Neurotrophin levels in different regions of the placenta and their association with birth outcome and blood pressure. Placenta 36(8):938–943. doi:10.1016/j.placenta.2015.06.006
Julio-Pieper M, Lozada P, Tapia V, Vega M, Miranda C, Vantman D, Ojeda SR, Romero C (2009) Nerve growth factor induces vascular endothelial growth factor expression in granulosa cells via a trkA receptor/mitogen-activated protein kinase-extracellularly regulated kinase 2-dependent pathway. J Clin Endocrinol Metab 94:3065–3071. doi:10.1210/jc.2009-0542
Frank P, Barrientos G, Tirado-Gonzalez I, Cohen M, Moschansky P, Peters EM, Klapp BF, Rose M, Tometten M, Blois SM (2014) Balanced levels of nerve growth factor are required for normal pregnancy progression. Reproduction 148:179–189. doi:10.1530/rep-14-0112
Sundrani D, Khot V, Pisal H, Mehendale S, Wagh G, Joshi A, Joshi S (2013) Gestation dependant changes in angiogenic factors and their associations with fetal growth measures in normotensive pregnancy. PLoS ONE 8:e54153. doi:10.1371/journal.pone.0054153
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Liu Y, Cox SR, Morita T, Kourembanas S (1995) Hypoxia regulates vascular endothelial growth factor gene expression in endothelial cells. Identification of a 5’ enhancer. Circ Res 77:638–643
Ferrara N (2004) Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev 25:581–611. doi:10.1210/er.2003-0027
Laskowska M, Laskowska K, Leszczynska-Gorzelak B, Oleszczuk J (2008) Are the maternal and umbilical VEGF-A and SVEGF-R1 altered in pregnancies complicated by preeclampsia with or without intrauterine foetal growth retardation? Preliminary communication. Med Wieku Rozwoj 12:499–506
Sundrani DP, Reddy US, Joshi AA, Mehendale SS, Chavan-Gautam PM, Hardikar AA, Chandak GR, Joshi SR (2013) Differential placental methylation and expression of VEGF, FLT-1 and KDR genes in human term and preterm preeclampsia. Clin Epigenet 5:6. doi:10.1186/1868-7083-5-6
Harris LK (2011) IFPA Gabor than award lecture: transformation of the spiral arteries in human pregnancy: key events in the remodelling timeline. Placenta 32(Suppl 2):S154–S158. doi:10.1016/j.placenta.2010.11.018
Sahay AS, Patil VV, Sundrani DP, Joshi AA, Wagh GN, Gupte SA, Joshi SR (2014) A longitudinal study of circulating angiogenic and antiangiogenic factors and AT1-AA levels in preeclampsia. Hypertens Res 37:753–758. doi:10.1038/hr.2014.71
Maynard SE, Min J-Y, Merchan J, Lim K-H, Li J, Mondal S, Libermann TA, Morgan JP, Sellke FW, Stillman IE, Epstein FH, Sukhatme VP, Karumanchi SA (2003) Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Investig 111:649–658. doi:10.1172/jci200317189
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676. doi:10.1038/nm0603-669
de Sousa Abreu R, Penalva LO, Marcotte EM, Vogel C (2009) Global signatures of protein and mRNA expression levels. Mol BioSyst 5:1512–1526. doi:10.1039/b908315d
Shchyogolev AI, Dubova EA, Pavlova KA, Lyapin VM, Sukhikh GT (2012) Expression of antiangiogenic factors in the placental structures in pre-eclampsia. Bull Exp Biol Med 154:287–291
Myatt L (2002) Role of placenta in preeclampsia. Endocrine 19:103–111. doi:10.1385/endo:19:1:103
Ulyatt C, Walker J, Ponnambalam S (2011) Hypoxia differentially regulates VEGFR1 and VEGFR2 levels and alters intracellular signaling and cell migration in endothelial cells. Biochem Biophys Res Commun 404:774–779. doi:10.1016/j.bbrc.2010.12.057
Helske S, Vuorela P, Carpen O, Hornig C, Weich H, Halmesmaki E (2001) Expression of vascular endothelial growth factor receptors 1, 2 and 3 in placentas from normal and complicated pregnancies. Mol Hum Reprod 7:205–210
Waltenberger J, Claesson-Welsh L, Siegbahn A, Shibuya M, Heldin CH (1994) Different signal transduction properties of KDR and Flt1, two receptors for vascular endothelial growth factor. J Biol Chem 269:26988–26995
Gille H, Kowalski J, Li B, LeCouter J, Moffat B, Zioncheck TF, Pelletier N, Ferrara N (2001) Analysis of biological effects and signaling properties of Flt-1 (VEGFR-1) and KDR (VEGFR-2). A reassessment using novel receptor-specific vascular endothelial growth factor mutants. J Biol Chem 276:3222–3230. doi:10.1074/jbc.M002016200
Zozzaro-Smith PE, Bushway ME, Gerber SA, Hebert D, Pressman EK, Lord EM, Miller RK, Murphy SP (2015) Whole mount immunofluorescence analysis of placentas from normotensive versus preeclamptic pregnancies. Placenta 36:1310–1317. doi:10.1016/j.placenta.2015.09.001
Munaut C, Lorquet S, Pequeux C, Coulon C, Le Goarant J, Chantraine F, Noel A, Goffin F, Tsatsaris V, Subtil D, Foidart JM (2012) Differential expression of Vegfr-2 and its soluble form in preeclampsia. PLoS ONE 7:e33475. doi:10.1371/journal.pone.0033475
Tripathi R, Rath G, Ralhan R, Saxena S, Salhan S (2009) Soluble and membranous vascular endothelial growth factor receptor-2 in pregnancies complicated by pre-eclampsia. Yonsei Med J 50:656–666. doi:10.3349/ymj.2009.50.5.656
Groten T, Gebhard N, Kreienberg R, Schleussner E, Reister F, Huppertz B (2010) Differential expression of VE-cadherin and VEGFR2 in placental syncytiotrophoblast during preeclampsia—new perspectives to explain the pathophysiology. Placenta 31:339–343. doi:10.1016/j.placenta.2010.01.014
Azimi-Nezhad M (2014) Vascular endothelial growth factor from embryonic status to cardiovascular pathology. Rep Biochem Mol Biol 2:59–69
Turner N, Grose R (2010) Fibroblast growth factor signalling: from development to cancer. Nat Rev Cancer 10:116–129. doi:10.1038/nrc2780
Tomaszewski M, Charchar FJ, Lynch MD, Padmanabhan S, Wang WY, Miller WH, Grzeszczak W, Maric C, Zukowska-Szczechowska E, Dominiczak AF (2007) Fibroblast growth factor 1 gene and hypertension: from the quantitative trait locus to positional analysis. Circulation 116:1915–1924. doi:10.1161/circulationaha.107.710293
Marwa BA, Raguema N, Zitouni H, Feten HB, Olfa K, Elfeleh R, Almawi W, Mahjoub T (2016) FGF1 and FGF2 mutations in preeclampsia and related features. Placenta 43:81–85. doi:10.1016/j.placenta.2016.05.007
Ozkan S, Vural B, Filiz S, Costur P, Dalcik H (2008) Placental expression of insulin-like growth factor-I, fibroblast growth factor-basic, and neural cell adhesion molecule in preeclampsia. J Matern Fetal Neonatal Med 21:831–838. doi:10.1080/14767050802251024
Wyatt SM, Kraus FT, Roh CR, Elchalal U, Nelson DM, Sadovsky Y (2005) The correlation between sampling site and gene expression in the term human placenta. Placenta 26:372–379. doi:10.1016/j.placenta.2004.07.003
Acknowledgements
The authors thank Indian Council of Medical Research (ICMR), India for funding this study. Author AS thanks the Department of Science and Technology (DST), Government of India for providing her the ‘INSPIRE fellowship’.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sahay, A.S., Jadhav, A.T., Sundrani, D.P. et al. VEGF and VEGFR1 levels in different regions of the normal and preeclampsia placentae. Mol Cell Biochem 438, 141–152 (2018). https://doi.org/10.1007/s11010-017-3121-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-017-3121-y