Abstract
Helicobacter pylori (H. pylori) infection is prevalent all over the world, which may even cause gastric cancer. Unfortunately, current antibiotic therapy cannot completely eradicate drug-resistant H. pylori. HF-18 (GFFKKAWRKVKKAFRRVL-NH2), a novel cationic antimicrobial peptide, showed broad antimicrobial activities, low cytotoxicity toward mammalian cells and low drug tolerance during our previous research. In this paper, to investigate the antibacterial activities and possible mechanisms underlying HF-18 against drug-resistant H. pylori, cytotoxin associated protein (CagA)-carrying recombinant E. coli BL21 was constructed. Subsequently, Zeta potential detection, N-Phenyl-1-naphthylamine uptake, propidium iodide fluorescence uptake, transmission electron microscope, qRT-PCR and Western blot assays were performed. Consequently, we found that HF-18 had potent antibacterial effects on clarithromycin- and amoxicillin- resistant H. pylori. In terms of its mode of action, HF-18 could neutralize negative charges on the surface of H. pylori, penetrate into and disrupt the integrity of bacterial membranes, resulting in the death of drug-resistant H. pylori to reduce cells infection. In addition, HF-18 inhibited the transcription of adhesion gene alpA/alpB and the expression of virulence protein CagA under sub-inhibitory concentration, and ultimately decreased bacterial virulence. Therefore, HF-18 lays a foundation for the development and utilization of new antibiotics.
Graphical Abstract





Similar content being viewed by others
Abbreviations
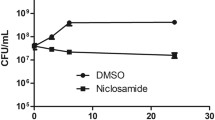
- CFU:
-
Colony forming unit
- Glu:
-
Glutamic acid
- Lys:
-
Lysine
- IPTG:
-
Isopropyl β-d-1-thiogalactopyranoside
References
Akhavan M, Foroughifar N, Pasdar H, Bekhradnia A (2019) Green synthesis, biological activity evaluation, and molecular docking studies of aryl alkylidene 2, 4-thiazolidinedione and rhodanine derivatives as antimicrobial agents. Comb Chem High Throughput Screen 22(10):716–727. https://doi.org/10.2174/1386207322666191127103122
Amieva M, Peek RM Jr (2016) Pathobiology of helicobacter pylori-induced gastric cancer. Gastroenterology 150(1):64–78. https://doi.org/10.1053/j.gastro.2015.09.004
Annunziato G (2019) Strategies to overcome antimicrobial resistance (AMR) making use of non-essential target inhibitors: a review. Int J Mol Sci. https://doi.org/10.3390/ijms20235844
Bugaytsova JA, Björnham O, Chernov YA, Gideonsson P, Henriksson S, Mendez M et al (2017) Helicobacter pylori adapts to chronic infection and gastric disease via pH-responsive BabA-mediated adherence. Cell Host Microbe 21(3):376–389. https://doi.org/10.1016/j.chom.2017.02.013
Crosio MA, Via MA, Cámara CI, Mangiarotti A, Del Pópolo MG, Wilke N (2019) Interaction of a polyarginine peptide with membranes of different mechanical properties. Biomolecules. https://doi.org/10.3390/biom9100625
De Leeuw E, Li C, Zeng P, Li C, Diepeveen-de Buin M, Lu WY et al (2010) Functional interaction of human neutrophil peptide-1 with the cell wall precursor lipid II. FEBS Lett 584(8):1543–1548. https://doi.org/10.1016/j.febslet.2010.03.004
Draper JL, Hansen LM, Bernick DL, Abedrabbo S, Underwood JG, Kong N et al (2017) Fallacy of the unique genome: sequence diversity within single helicobacter pylori strains. Mbio. https://doi.org/10.1128/mBio.02321-16
Dyer V, Brüggemann H, Sörensen M, Kühl AA, Hoffman K, Brinkmann V et al (2018) Genomic features of the helicobacter pylori strain PMSS1 and its virulence attributes as deduced from its in vivo colonisation patterns. Mol Microbiol 110(5):761–776. https://doi.org/10.1111/mmi.14123
Follmer C (2010) Ureases as a target for the treatment of gastric and urinary infections. J Clin Pathol 63(5):424–430. https://doi.org/10.1136/jcp.2009.072595
González R, Mendive-Tapia L, Pastrian MB, Albericio F, Lavilla R, Cascone O, Iannucci NB (2016) Enhanced antimicrobial activity of a peptide derived from human lysozyme by arylation of its tryptophan residues. J Pept Sci 22(2):123–128. https://doi.org/10.1002/psc.2850
Hahn AW, Jain R, Spach DH (2016) New approaches to antibiotic use and review of recently approved antimicrobial agents. Med Clin North Am 100(4):911–926. https://doi.org/10.1016/j.mcna.2016.03.012
Hatakeyama M (2014) Helicobacter pylori CagA and gastric cancer: a paradigm for hit-and-run carcinogenesis. Cell Host Microbe 15(3):306–316. https://doi.org/10.1016/j.chom.2014.02.008
Helander IM, Mattila-Sandholm T (2000) Fluorometric assessment of gram-negative bacterial permeabilization. J Appl Microbiol 88(2):213–219. https://doi.org/10.1046/j.1365-2672.2000.00971.x
Jafari E, Mahmoodi S (2021) Design, expression, and purification of a multi-epitope vaccine against helicobacter pylori based on melittin as an adjuvant. Microb Pathog 157:104970. https://doi.org/10.1016/j.micpath.2021.104970
Jiang M, Ma L, Huang Y, Wu H, Dou J, Zhou C (2020a) Antimicrobial activities of peptide Cbf-K(16) against drug-resistant helicobacter pylori infection in vitro and in vivo. Microb Pathog 138:103847. https://doi.org/10.1016/j.micpath.2019.103847
Jiang M, Yang X, Wu H, Huang Y, Dou J, Zhou C, Ma L (2020b) An active domain HF-18 derived from hagfish intestinal peptide effectively inhibited drug-resistant bacteria in vitro/vivo. Biochem Pharmacol 172:113746. https://doi.org/10.1016/j.bcp.2019.113746
Kalali B, Mejías-Luque R, Javaheri A, Gerhard M (2014) H. pylori virulence factors: influence on immune system and pathology. Mediators Inflamm 2014:426309. https://doi.org/10.1155/2014/426309
Kalenić S, Plecko V, Kresić S, Presecki V, Tripković V, Zele-Stracević L et al (1998) Helicobacter pylori: in vitro induction of resistance to azithromycin. Chemotherapy 44(1):17–20. https://doi.org/10.1159/000007085
Knutson MD, Oukka M, Koss LM, Aydemir F, Wessling-Resnick M (2005) Iron release from macrophages after erythrophagocytosis is up-regulated by ferroportin 1 overexpression and down-regulated by hepcidin. Proc Natl Acad Sci USA 102(5):1324–1328. https://doi.org/10.1073/pnas.0409409102
Lin Y, Kawai S, Sasakabe T, Nagata C, Naito M, Tanaka K et al (2021) Effects of Helicobacter pylori eradication on gastric cancer incidence in the Japanese population: a systematic evidence review. Jpn J Clin Oncol 51(7):1158–1170. https://doi.org/10.1093/jjco/hyab055
Loewen PC, Carpena X, Rovira C, Ivancich A, Perez-Luque R, Haas R et al (2004) Structure of helicobacter pylori catalase, with and without formic acid bound, at 1.6 A resolution. Biochemistry 43(11):3089–3103. https://doi.org/10.1021/bi035663i
Ma L, Wang Y, Wang M, Tian Y, Kang W, Liu H et al (2016) Effective antimicrobial activity of Cbf-14, derived from a cathelin-like domain, against penicillin-resistant bacteria. Biomaterials 87:32–45. https://doi.org/10.1016/j.biomaterials.2016.02.011
Ma L, Xie X, Liu H, Huang Y, Wu H, Jiang M et al (2020) Potent antibacterial activity of MSI-1 derived from the magainin 2 peptide against drug-resistant bacteria. Theranostics 10(3):1373–1390. https://doi.org/10.7150/thno.39157
Moosazadeh Moghaddam M, Eftekhary M, Erfanimanesh S, Hashemi A, Fallah Omrani V, Farhadihosseinabadi B et al (2018) Comparison of the antibacterial effects of a short cationic peptide and 1% silver bioactive glass against extensively drug-resistant bacteria, Pseudomonas aeruginosa and Acinetobacter baumannii, isolated from burn patients. Amino Acids 50(11):1617–1628. https://doi.org/10.1007/s00726-018-2638-z
Narayana JL, Huang HN, Wu CJ, Chen JY (2015) Epinecidin-1 antimicrobial activity: in vitro membrane lysis and In vivo efficacy against helicobacter pylori infection in a mouse model. Biomaterials 61:41–51. https://doi.org/10.1016/j.biomaterials.2015.05.014
Neshani A, Zare H, Akbari Eidgahi MR, Hooshyar Chichaklu A, Movaqar A, Ghazvini K (2019) Review of antimicrobial peptides with anti-helicobacter pylori activity. Helicobacter 24(1):e12555. https://doi.org/10.1111/hel.12555
Otvos L Jr, Ostorhazi E, Szabo D, Zumbrun SD, Miller LL, Halasohoris SA et al (2018) Synergy between proline-rich antimicrobial peptides and small molecule antibiotics against selected gram-negative pathogens in vitro and in vivo. Front Chem 6:309. https://doi.org/10.3389/fchem.2018.00309
Pan CY, Tsai TY, Su BC, Hui CF, Chen JY (2017) Study of the antimicrobial activity of tilapia piscidin 3 (TP3) and TP4 and their effects on immune functions in hybrid tilapia (Oreochromis spp.). PLoS ONE 12(1):e0169678. https://doi.org/10.1371/journal.pone.0169678
Pellicano R, Ribaldone DG, Fagoonee S, Astegiano M, Saracco GM, Mégraud F (2016) A 2016 panorama of helicobacter pylori infection: key messages for clinicians. Panminerva Med 58:304–317
Riccardi C, Nicoletti I (2006) Analysis of apoptosis by propidium iodide staining and flow cytometry. Nat Protoc 1(3):1458–1461. https://doi.org/10.1038/nprot.2006.238
Rothenbacher D, Brenner H (2003) Burden of helicobacter pylori and H. pylori-related diseases in developed countries: recent developments and future implications. Microbes Infect 5(8):693–703. https://doi.org/10.1016/s1286-4579(03)00111-4
Ryu S, Song PI, Seo CH, Cheong H, Park Y (2014) Colonization and infection of the skin by S. aureus: immune system evasion and the response to cationic antimicrobial peptides. Int J Mol Sci 15(5):8753–8772. https://doi.org/10.3390/ijms15058753
Saadat I, Higashi H, Obuse C, Umeda M, Murata-Kamiya N, Saito Y et al (2007) Helicobacter pylori CagA targets PAR1/MARK kinase to disrupt epithelial cell polarity. Nature 447(7142):330–333. https://doi.org/10.1038/nature05765
Stein M, Bagnoli F, Halenbeck R, Rappuoli R, Fantl WJ, Covacci A (2002) c-Src/Lyn kinases activate helicobacter pylori CagA through tyrosine phosphorylation of the EPIYA motifs. Mol Microbiol 43(4):971–980. https://doi.org/10.1046/j.1365-2958.2002.02781.x
Stone TA, Cole GB, Ravamehr-Lake D, Nguyen HQ, Khan F, Sharpe S, Deber CM (2019) Positive charge patterning and hydrophobicity of membrane-active antimicrobial peptides as determinants of activity, toxicity, and pharmacokinetic stability. J Med Chem 62(13):6276–6286. https://doi.org/10.1021/acs.jmedchem.9b00657
Suthisamphat N, Dechayont B, Phuaklee P, Prajuabjinda O, Vilaichone RK, Itharat A et al (2020) Anti-helicobacter pylori, anti-inflammatory, cytotoxic, and antioxidant activities of mace extracts from Myristica fragrans. Evid Based Complement Alternat Med 2020:7576818. https://doi.org/10.1155/2020/7576818
Takahashi-Kanemitsu A, Knight CT, Hatakeyama M (2020) Molecular anatomy and pathogenic actions of helicobacter pylori CagA that underpin gastric carcinogenesis. Cell Mol Immunol 17(1):50–63. https://doi.org/10.1038/s41423-019-0339-5
Tenney JH, Maack RW, Chippendale GR (1983) Rapid selection of organisms with increasing resistance on subinhibitory concentrations of norfloxacin in agar. Antimicrob Agents Chemother 23(1):188–189. https://doi.org/10.1128/aac.23.1.188
Thapa RK, Diep DB, Tønnesen HH (2020) Topical antimicrobial peptide formulations for wound healing: current developments and future prospects. Acta Biomater 103:52–67. https://doi.org/10.1016/j.actbio.2019.12.025
Theuretzbacher U, Outterson K, Engel A, Karlén A (2020) The global preclinical antibacterial pipeline. Nat Rev Microbiol 18(5):275–285. https://doi.org/10.1038/s41579-019-0288-0
Tohidpour A (2016) CagA-mediated pathogenesis of helicobacter pylori. Microb Pathog 93:44–55. https://doi.org/10.1016/j.micpath.2016.01.005
Torres MDT, Sothiselvam S, Lu TK, de la Fuente-Nunez C (2019) Peptide design principles for antimicrobial applications. J Mol Biol 431(18):3547–3567. https://doi.org/10.1016/j.jmb.2018.12.015
Wiedemann T, Loell E, Mueller S, Stoeckelhuber M, Stolte M, Haas R, Rieder G (2009) Helicobacter pylori cag-pathogenicity island-dependent early immunological response triggers later precancerous gastric changes in Mongolian gerbils. PLoS ONE 4(3):e4754. https://doi.org/10.1371/journal.pone.0004754
Wiegand I, Hilpert K, Hancock RE (2008) Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc 3(2):163–175. https://doi.org/10.1038/nprot.2007.521
Woo HJ, Yang JY, Lee MH, Kim HW, Kwon HJ, Park M et al (2020) Inhibitory effects of β-caryophyllene on helicobacter pylori Infection in vitro and in vivo. Int J Mol Sci. https://doi.org/10.3390/ijms21031008
Yang Y, Bian L, Hang X, Yan C, Huang Y, Ye F et al (2020) In vitro activity of new tetracycline analogues omadacycline and eravacycline against clinical isolates of helicobacter pylori collected in China. Diagn Microbiol Infect Dis 98(3):115129. https://doi.org/10.1016/j.diagmicrobio.2020.115129
Yeaman MR, Yount NY (2003) Mechanisms of antimicrobial peptide action and resistance. Pharmacol Rev 55(1):27–55. https://doi.org/10.1124/pr.55.1.2
Yoshida N, Yoshikawa T (2002) Effect of helicobacter pylori-mediated inflammation on nonsteroidal anti-inflammatory drugs-induced gastric mucosal injury. Keio J Med 51(Suppl 2):45–50. https://doi.org/10.2302/kjm.51.supplement2_45
Acknowledgements
This work was funded by the National Natural Science Foundation of China (Grant No. 82173863; No.81803591); National Key Research and Development Program of China (Grant No. 2018YFA0902000); the fellowship of China postdoctoral science foundation (Grant No. 2020T130723); Natural Science Foundation of Jiangsu Province of China (Grant No. BK20201327); the Basic Scientific Research Business Expense Project of China Pharmaceutical University (Grant No. 2632021ZD07)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no competing interests in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, C., Jiang, M., Ye, X. et al. Antibacterial Activities of Peptide HF-18 Against Helicobacter pylori and its Virulence Protein CagA. Int J Pept Res Ther 28, 63 (2022). https://doi.org/10.1007/s10989-022-10372-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s10989-022-10372-7