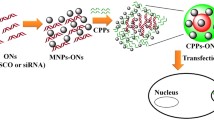
Abstract
Oligonucleotide-based gene regulation has a high potential in gene therapy, but the plasma membrane is impermeable for nucleic acid polymers and, consequently, an efficient and non-toxic transfection agent is needed for their delivery into the cell. In this study we present a novel series, NickFects, of chemically modified TP10 peptide-based delivery vectors used for the cellular delivery of single-stranded oligonucleotides. These carriers, obtained by replacement of Ile8 by threonine in stearyl-TP10 and by modifying of tyrosine and/or threonine, respectively, by phosphorylation formed 300–500 nm in size peptide:oligonucleotide nanocomplexes with negative surface charges. The highest splice-correcting effect was obtained when phosphorotiate 2′-O-methyl oligonucleotides were transduced into cells by NickFect1 (NF1) or NickFect2 (NF2). In addition, we also show how a small modification (one or two negative charges) in peptide sequence can affect its ability to deliver ONs into cells and increase their potency in the splicing redirection assay. Our studies demonstrate that NF1 and NF2 have higher transfection efficacy for oligonucleotides as compared to the most commonly used transfection agent Lipofectamine™ 2000 and lead to higher biological response in cells.





Similar content being viewed by others
Abbreviations
- 2′-OMe ON:
-
Phosphorothioate 2′-O-methyl RNA
- SCO:
-
Splice-correcting oligonucleotides
- CPPs:
-
Cell-penetrating peptides
- PBS:
-
Phosphate buffered saline
- TFA:
-
Trifluoroacetic acid
- TP10:
-
Transportan 10
- ON:
-
Oligonucleotide
- SFM:
-
Serum-free medium
- FM:
-
Serum-containing medium
- OD:
-
Optical density
- HOBt:
-
Hydroxybenzotriazole
- HBTU:
-
O-Benzotriazole-N,N,N′,N′-tetramethyl-uronium-hexafluoro-phosphate
- DIEA:
-
Diisopropylethylamine
- TIS:
-
Triisopropylsilane
References
Crooke ST (2004) Progress in antisense technology. Annu Rev Med 55:61–95
Deshayes S, Morris M, Heitz F, Divita G (2008) Delivery of proteins and nucleic acids using a non-covalent peptide-based strategy. Adv Drug Deliv Rev 60:537–547
Fattal E, Couvreur P, Dubernet C (2004) “Smart” delivery of antisense oligonucleotides by anionic pH-sensitive liposomes. Adv Drug Deliv Rev 56:931–946
Fields GB, Noble RL (1990) Solid phase peptide synthesis utilizing 9-fluorenylmethoxycarbonyl amino acids. Int J Pept Protein Res 35:161–214
Futaki S, Ohashi W, Suzuki T, Niwa M, Tanaka S, Ueda K, Harashima H, Sugiura Y (2001) Stearylated arginine-rich peptides: a new class of transfection systems. Bioconjug Chem 12:1005–1011
Gabhann FM, Annex BH, Popel AS (2010) Gene therapy from the perspective of systems biology. Curr Opin Mol Ther 12:570–577
Garcia-Blanco MA, Baraniak AP, Lasda EL (2004) Alternative splicing in disease and therapy. Nat Biotechnol 22:535–546
Kang SH, Cho MJ, Kole R (1998) Up-regulation of luciferase gene expression with antisense oligonucleotides: implications and applications in functional assay development. Biochemistry 37:6235–6239
Khalil IA, Futaki S, Niwa M, Baba Y, Kaji N, Kamiya H, Harashima H (2004) Mechanism of improved gene transfer by the N-terminal stearylation of octaarginine: enhanced cellular association by hydrophobic core formation. Gene Ther 11:636–644
Kim HJ, Ishii A, Miyata K, Lee Y, Wu S, Oba M, Nishiyama N, Kataoka NK (2010) Introduction of stearoyl moieties into a biocompatible cationic polyaspartamide derivative, PAsp(DET), with endosomal escaping function for enhanced siRNA-mediated gene knockdown. J Control Release 145:141–148
Lehto T, Abes R, Oskolkov N, Suhorutšenko J, Copolovici DM, Mäger I, Viola JR, Simonson OE, Ezzat K, Guterstam P, Eriste E, Smith CI, Lebleu B, Andaloussi El S, Langel Ü (2010) Delivery of nucleic acids with a stearylated (RxR)4 peptide using a non-covalent co-incubation strategy. J Control Release 141:42–51
Ma H, Diamond SL (2001) Nonviral gene therapy and its delivery systems. Curr Pharm Biotechnol 2:1–17
Mäe M, El Andaloussi S, Lundin P, Oskolkov N, Johansson HJ, Guterstam P, Langel Ü (2009) A stearylated CPP for delivery of splice correcting oligonucleotides using a non-covalent co-incubation strategy. J Control Release 134:221–227
Midoux P, Pichon C, Yaouanc JJ, Jaffres PA (2009) Chemical vectors for gene delivery: a current review on polymers, peptides and lipids containing histidine or imidazole as nucleic acids carriers. Br J Pharmacol 157:166–178
Morris MC, Vidal P, Chaloin L, Heitz F, Divita G (1997) A new peptide vector for efficient delivery of oligonucleotides into mammalian cells. Nucleic Acids Res 25:2730–2736
Morris MC, Deshayes S, Heitz F, Divita G (2008) Cell-penetrating peptides: from molecular mechanisms to therapeutics. Biol Cell 100:201–217
Nakamura Y, Kogure K, Futaki S, Harashima H (2007) Octaarginine-modified multifunctional envelope-type nano device for siRNA. J Control Release 119:360–367
Niidome T, Urakawa M, Takaji K, Matsuo Y, Ohmori N, Wada A, Hirayama T, Aoyagi H (1999) Influence of lipophilic groups in cationic alpha-helical peptides on their abilities to bind with DNA and deliver genes into cells. J Pept Res 54:361–367
Padari K, Koppel K, Lorents A, Hällbrink M, Mano M, Pedroso De Lima MC, Pooga M (2010) S4(13)-PV cell-penetrating peptide forms nanoparticle-like structures to gain entry into cells. Bioconjug Chem 21:774–783
Pajares MJ, Ezponda T, Catena R, Calvo A, Pio R, Montuenga LM (2007) Alternative splicing: an emerging topic in molecular and clinical oncology. Lancet Oncol 8:349–357
Pooga M, Hällbrink M, Zorko M, Langel Ü (1998) Cell penetration by transportan. FASEB J 12:67–77
Rothbard JB, Kreider E, Vandeusen CL, Wright L, Wylie BL, Wender PA (2002) Arginine-rich molecular transporters for drug delivery: role of backbone spacing in cellular uptake. J Med Chem 45:3612–3618
Said Hassane F, Saleh AF, Abes R, Gait MJ, Lebleu B (2010) Cell penetrating peptides: overview and applications to the delivery of oligonucleotides. Cell Mol Life Sci 67:715–726
Son KK, Patel DH, Tkach D, Park A (2000a) Cationic liposome and plasmid DNA complexes formed in serum-free medium under optimum transfection condition are negatively charged. Biochim Biophys Acta 1466:11–15
Son KK, Tkach D, Hall KJ (2000b) Efficient in vivo gene delivery by the negatively charged complexes of cationic liposomes and plasmid DNA. Biochim Biophys Acta 1468:6–10
Soomets U, Lindgren M, Gallet X, Hällbrink M, Elmquist A, Balaspiri L, Zorko M, Pooga M, Brasseur R, Langel Ü (2000) Deletion analogues of transportan. Biochim Biophys Acta 1467:165–176
Tonges L, Lingor P, Egle R, Dietz GP, Fahr A, Bahr M (2006) Stearylated octaarginine and artificial virus-like particles for transfection of siRNA into primary rat neurons. RNA 12:1431–1438
Wadia JS, Stan RV, Dowdy SF (2004) Transducible TAT-HA fusogenic peptide enhances escape of TAT-fusion proteins after lipid raft macropinocytosis. Nat Med 10:310–315
Zamecnik PC, Stephenson ML (1978) Inhibition of Rous sarcoma virus replication and cell transformation by a specific oligodeoxynucleotide. Proc Natl Acad Sci USA 75:280–284
Acknowledgments
The study presented in this article was supported by Swedish Research Council (VR-NT); by the Center for Biomembrane Research, Stockholm; by Cepep Eesti OÜ; by the Knut and Alice Wallenberg’s Foundation; by the EU through the European Regional Development Fund through the Center of Excellence in Chemical Biology and in Genomics, Estonia; by the targeted financing SF0180027s08 and SF0180019s11 from the Estonian Ministry of Education and Research; by the DoRa Program of The European Social Fund; by Archimedes Foundation and by the Estonian Science Foundation (ESF 8705, Mobilitas—MJD64). The authors would also like to thank J. Pae and C. Juks for assistance in peptide synthesis, S. El-Andaloussi for fruitful discussions and M. Kure for an excellent technical assistance in electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oskolkov, N., Arukuusk, P., Copolovici, DM. et al. NickFects, Phosphorylated Derivatives of Transportan 10 for Cellular Delivery of Oligonucleotides. Int J Pept Res Ther 17, 147–157 (2011). https://doi.org/10.1007/s10989-011-9252-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-011-9252-1