Abstract
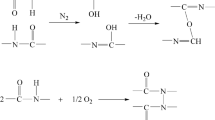
Synthesis, characterization and thermal analysis of polyaniline (PANI)/ZrO2 composite and PANI was reported in our early work. In this present, the kinetic analysis of decomposition process for these two materials was performed under non-isothermal conditions. The activation energies were calculated through Friedman and Ozawa-Flynn-Wall methods, and the possible kinetic model functions have been estimated through the multiple linear regression method. The results show that the kinetic models for the decomposition process of PANI/ZrO2 composite and PANI are all D3, and the corresponding function is ƒ(α)=1.5(1−α)2/3[1−(1-α)1/3]−1. The correlated kinetic parameters are E a=112.7±9.2 kJ mol−1, lnA=13.9 and E a=81.8±5.6 kJ mol−1, lnA=8.8 for PANI/ZrO2 composite and PANI, respectively.
Similar content being viewed by others

References
L. J. Zhang and M. X. Wan, J. Phys. Chem. B, 107 (2003) 6748.
Z. M. Zhang and M. X. Wan, Synth. Met., 132 (2003) 205.
P. S. Khiew, N. M. Huang, S. Radiman and M. S. Ahmad, Mater. Lett., 58 (2004) 516.
M. Biswas, S. S. Ray and Y. Liu, Synth. Met., 105 (1999) 99.
D. Tsocheva and L. Terlemezyan, J. Therm. Anal. Cal., 81 (2005) 3.
S. X. Wang, L. X. Sun, Z. C. Tan, F. Xu and Y. S. Li, J. Therm. Anal. Cal., 89 (2007) 609.
S. X. Wang, Z. C. Tan, Y. S. Li, L. X. Sun and T. Zhang, Thermochim. Acta, 441 (2006) 191.
H. L. Friedman, J. Polym. Sci. Part C, 6 (1963) 183.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881.
L. Jun, Thermochim. Acta, 406 (2003) 77.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, S.X., Tan, Z.C., Li, Y.S. et al. A kinetic analysis of thermal decomposition of polyaniline/ZrO2 composite. J Therm Anal Calorim 92, 483–487 (2008). https://doi.org/10.1007/s10973-007-8356-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8356-5