Abstract
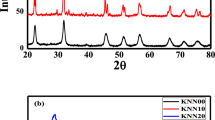
Potassium sodium niobate (KNN) ceramic powders by a variation of sol–gel method is synthesized. The metal precursors used for the KNN synthesis are potassium carbonate, sodium carbonate and niobium hydroxide, ethylene glycol are used as chelating and esterification agent, respectively. The effects of amount of oxalic acid (OA) and ethylene glycol (EG), pH value on the stability of the precursor sol were investigated. The evolution of (K0.5Na0.5)NbO3 crystal phase was also investigated by XRD, IR, SEM and TG-DTA. The results showed that stable precursor sol was formed when n(OA):n(Mn+) = 3:1, n(OA):n(EG) = 1:2 and pH value was in the range of 2.5–3.5. Xerogel was sintered in the range of 500–650 °C to prepare K6Nb10.88O30 and Na2Nb4O11 powder. Then the compound was sintered at 750 °C to produce perovskite (K0.5Na0.5)NbO3 ceramic powders. The grain size is about 100–200 nm.






Similar content being viewed by others
References
Deng YL, Liao XW, Wang HP, Cheng XM (2008) J China West Norm University 29(4):356–359
Wang K, Zhang BP, Li JF, Zhang LM (2008) J Electroceram 21:251–254
Soderlind F, Kall P-O, Helmersson U (2005) J Cryst Growth 281:468
Yang H, Lin Y, Zhu J et al (2008) Ceramics 5(2):21–28
Deng YL, Liao YW, Wang HP (2008) J China West Norm University (Natural Sciences) 29(4):356–358
Yang HB, Lin Y, Wang F (2007) Journal of Wuhan University of Technology 29(7):58–61
Camargo ER, Popa M, Kakihana M (2002) Chem Mater 14(5):2365–2368
Navale SC, Samuel V, Ravi V (2005) Bull Mater Sci 28(5):391–393
Wang B, Gao S, Shao L et al (2004) Anal Chem. Higher Education Press, Peking, pp 49–52
Li M, Du P, Ji S et al (2010) Ordnance Mater Sci Eng 33(1):13–15
Acknowledgments
Supported by the Scientific Project of Heilongjiang Province under Grant No E2007-31.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, DQ., Qin, ZC., Yang, XY. et al. Study on synthesis and evolution of sodium potassium niobate ceramic powders by an oxalic acid-based sol–gel method. J Sol-Gel Sci Technol 57, 31–35 (2011). https://doi.org/10.1007/s10971-010-2320-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-010-2320-8