Abstract
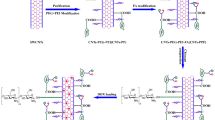
Poly(ethylene glycol) functionalized carbon nanotubes (CNTs-PEG) have attracted great research interest due to their good biocompatibility and wide applications in biomedical areas, such as drug delivery, bioimaging and photothermal therapy. The biodistribution studies are fundamental and essential to the biomedical applications and toxicological evaluations of CNTs-PEG, while isotopic labeling is the most adopted method for the quantitation of CNTs in vivo. Herein, we studied the biodistribution of hydroxyl terminated CNTs-PEG (CNTs-PEG-OH) in mice using 125I-labeling technique. The blood concentrations and accumulation levels of CNTs-PEG-OH in different organs were quantitatively analyzed by detecting the radioactivity of 125I labeled on CNTs-PEG-OH. The results indicated that CNTs-PEG-OH were quickly cleared from blood circulation and distributed to the entire body, except brain. CNTs-PEG-OH were mainly sequestrated by liver after intravenous injection. The hepatic accumulation decreased along with time elapse. Prolonging the PEG chains led to the decrease of hepatic accumulation, while the biodistribution pattern was similar. The implications to the biomedical applications and safety evaluations of CNTs-PEG are discussed.







Similar content being viewed by others
References
Schnorr JM, Swager TM (2011) Emerging applications of carbon nanotubes. Chem Mater 23:646–657
Lu F, Gu L, Meziani MJ (2009) Advances in bioapplications of carbon nanotubes. Adv Mater 21:139–152
Thakare VS, Das M, Jain AK, Patil S, Jain S (2010) Carbon nanotubes in cancer theragnosis. Nanomedicine 5:1277–1301
Vashist SK, Zheng D, Pastorin G et al (2011) Delivery of drugs and biomolecules using carbon nanotubes. Carbon 49:4077–4097
Liu Z, Chen K, Davis C et al (2008) Drug delivery with carbon nanotubes for in vivo cancer treatment. Cancer Res 68:6652–6660
Keren S, Zavaleta C, Cheng Z et al (2008) Noninvasive molecular imaging of small living subjects using Raman spectroscopy. Proc Natl Acad Sci USA 105:5844–5849
Moon HK, Lee SH, Choi HC (2009) In vivo near-infrared mediated tumor destruction by photothermal effect of carbon nanotubes. ACS Nano 3:3707–3713
Welsher K, Sherlock SP, Dai H (2011) Deep-tissue anatomical imaging of mice using carbon nanotube fluorophores in the second near-infrared window. Proc Natl Acad Sci USA 108:8943–8948
Yang S-T, Luo J, Zhou Q, Wang H (2012) Pharmacokinetics, metabolism and toxicity of carbon nanotubes for biomedical purposes. Theranostics 2:271–282
Liu J-H, Yang S-T, Wang HF, Liu Y (2010) Advances in biodistribution study and tracing methodology of carbon nanotubes. J Nanosci Nanotechnol 10:8469–8481
Yang S-T, Fernando KAS, Liu J-H et al (2008) Covalently PEGylated carbon nanotubes with stealth character in vivo. Small 4:940–944
Liu Z, Davis C, Cai W et al (2008) Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Proc Natl Acad Sci USA 105:1410–1415
Owens DE III, Peppas NA (2006) Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int J Pharm 300:93–102
Yang S-T, Wang H, Wang Y et al (2011) Removal of carbon nanotubes from aqueous environment with filter paper. Chemosphere 82:621–626
Sun Y-P, Huang W, Lin Y et al (2001) Soluble dendron-functionalized carbon nanotubes: preparation, characterization, and properties. Chem Mater 13:2864–2869
Deng X, Yang S-T, Nie H, Wang H, Liu Y (2008) A generally adoptable radiotracing method for tracking carbon nanotubes in animals. Nanotechnology 19:075101
Faure AC, Dufort S, Josserand V et al (2009) Control of the in vivo biodistribution of hybrid nanoparticles with different poly(ethylene glycol) coatings. Small 5:2565–2575
Geng Y, Dalhaimer P, Cai S et al (2007) Shape effects of filaments versus spherical particles in flow and drug delivery. Nat Nanotechnol 2:249–255
Landsiedel R, Ma-Hock L, Kroll A et al (2010) Testing metal-oxide nanomaterials for human safety. Adv Mater 22:2601–2627
Gref R, Luck M, Quellec P et al (2000) ‘Stealth’ corona-core nanoparticles surface modified by polyethylene glycol (PEG): influences of the corona (PEG chain length and surface density) and of the core composition on phagocytic uptake and plasma protein adsorption. Colloids Surf B 18:301–313
Acknowledgments
We acknowledge financial support from the China Ministry of Science and Technology (973 Project No. 2011CB933402), the China Natural Science Foundation (Nos. 21071094 and 20871010), Shanghai Municipal Education Committee (11ZZ82) and the Fundamental Research Funds for the Central Universities, Southwest University for Nationalities (No. 11NZYBS06)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10967_2012_1901_MOESM1_ESM.pdf
Supplementary material 1 (PDF 77 kb). TEM image of MWCNTs-PEG20000-OH; (2) the biodistribution of 125I-MWCNT-PEG4000-OH and Na125I in mice after intravenous injection
Rights and permissions
About this article
Cite this article
Yang, ST., Wang, YW., Liu, JH. et al. Biodistribution of multi-walled carbon nanotubes functionalized by hydroxyl terminated poly(ethylene glycol) in mice. J Radioanal Nucl Chem 295, 1181–1186 (2013). https://doi.org/10.1007/s10967-012-1901-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-012-1901-0